
Pros and Cons of Spacers in the Treatment of Late Periprosthetic Infections of the Hip
*Corresponding Author(s):
Bernd FinkDepartment Of Joint Replacement, General And Rheumatic Orthopedics, Orthopedic Clinic Markgröningeng G GmbH, University Medical Center Hamburg-Eppendorf, Kurt-Lindemann-Weg 10, 71706 Markgröningen, Germany
Tel:+49 71459153201,
Fax:+49 7145919153229
Email:b.fink@okm.de
Abstract
In late periprosthetic joint infection of the hip all foreign material has to be removed and in septic two-stage revision an antibiotic-laden spacer is an option to fill the joint gap. It preserves the function of the joint, so the patient can be mobilized and reimplantation of total hip arthroplasty is technically easier in the second stage. The spacers have an important role also in the local antibiotic therapy of the periprosthetic joint infection by releasing antibiotics. One disadvantage of spacers is the wear of cement particles which have to be removed radically via the debridement in the second stage.
Keywords
Hip arthroplasty; Periprosthetic joint infection; Spacer; Two-stage revision
INTRODUCTION
Periprosthetic joint infections as a serious complication of hip arthroplasties occur with an incidence of less than 1% after primary hip joint replacements and of around 4% after revision surgery [1-4]. When early infections occur, within 4 weeks of implantation, the implant can be left in place with a high probability of cure whereas late infections require prosthesis revision to eradicate the infection [3,4]. In such cases, one can differentiate between one-stage and two-stage revisions. One-stage revision include the removal of all foreign material and the implantation of a new prosthesis with specific antibiotics mixed in the cement for fixation of the implant components in the same operation. Prerequisites are that the pathogen or pathogens have been identified in aspirated synovial fluid or tissue biopsy and that their susceptibility to antibiotics has been determined, so that specific antibiotic mixture can be added to the bone cement and enable a specific local antibiotic therapy [4]. Two-stage revision involves an initial operation to remove all foreign materials and this is followed by an interim phase of mostly 6-12 weeks, either left as a Girdle stone situation or with the implantation of a cement spacer.
Two-stage septic revision surgery is the most common method for treating infected endoprosthesis. A general advantage of the two-stage concept is that the surgical debridement is carried out twice whereby the second operation allows for the eradication of residual organisms following the initial debridement. The cement of the spacer is not intended as a means of fixing the prosthesis so the mechanical characteristic of the cement is not of primary importance at this stage. Thus, large amounts of antibiotics can be mixed into the cement before the spacer is formed. It has been possible to achieve a survival rate using two-stage revision concepts for infected hip arthroplasties of between 90% and 100% [2,5-8].
In most two-stage revisions an antibiotic-containing spacer is usually placed in position for a certain period of time before the final prosthesis is implanted [5,6,9-11]. The function of the spacer is on the one hand to release the antibiotic into the infected bed of the prosthesis and on the other to minimize soft-tissue contractures, retain soft tissue tension and so maintain reasonable functionality until a prosthesis can be re-implanted [5].
There are many questions pertaining to both one-stage and two-stage revisions that still have to be answered and existing procedures are based more on empirical findings than on data from prospective studies with a high level of evidence. It is for this reason that the following aspects of two-stage revision have been treated very differently by different groups: the type of spacer, the type of antibiotic used in the spacer, the duration of the spacer period, the duration of systemic antibiotic treatment, aspiration before re-implantation and the type of re-implantation (cemented or cementless).
TYPE OF SPACER
Mobile spacers can be differentiated into monoblock and two-part spacers. The potential disadvantages of the monoblock spacers are spacer fracture and bone resorption [15,30]. The monoblock spacer induces bone resorption at the acetabulum because the hard cement has to articulate against the osteoporotic bone caused by the infection. This is avoided in the two-part spacer by the spacer having its own articulation surface. However, this cement-based articulation surface in the two-part spacer can lead to the release of abraded cement particles [17,31].
The femoral component of the monoblock and two-part spacers is associated with the risk of spacer fracture. This risk is particularly high when the femoral component is composed of cement alone. It is therefore recommended that the spacer consist of a metal core encased in cement, as is the case in commercially available spacers. A further risk is the potential for dislocation of the spacer out of the bone (either with or without fracture). In order to avoid this complication, it is recommended that, instead of simply inserting the spacer into the femur, the prepared spacer is fixed in position by applying cement at the metaphysis.
We use a two-part spacer where the cup-shaped acetabulum spacer is formed out of antibiotic loaded cement (with a specific mixture of antibiotics recommended by the microbiologist). The spacer stem component consists of old prosthesis stem models, monoblock devices in most cases and no longer used for primary implantations. This stem device is encased in antibiotic-supplemented cement and, just before implantation, coated in the patient's own blood in order to facilitate easier removal by decreasing the stability of the implant-cement-interface [6,31]. The two components of the spacer are connected by a metal headpiece (Figures 1 and 2) [6]. However, an analysis of synovial membranes obtained during the operation to remove the spacer and to implant the new prosthesis revealed the presence of abraded cement debris, in particular, zirconium dioxide particles [31]. So it must be concluded that all types of spacer will produce abraded cement particles and this only goes to emphasize the necessity for a radical debridement of the joint area at the time of prosthesis implantation during the second stage of the revision [31]. The use of zirconium-free spacer cement (Heraeus Medical GmbH, Wertheim, Germany) is aimed at circumventing this problem associated with abraded particles.
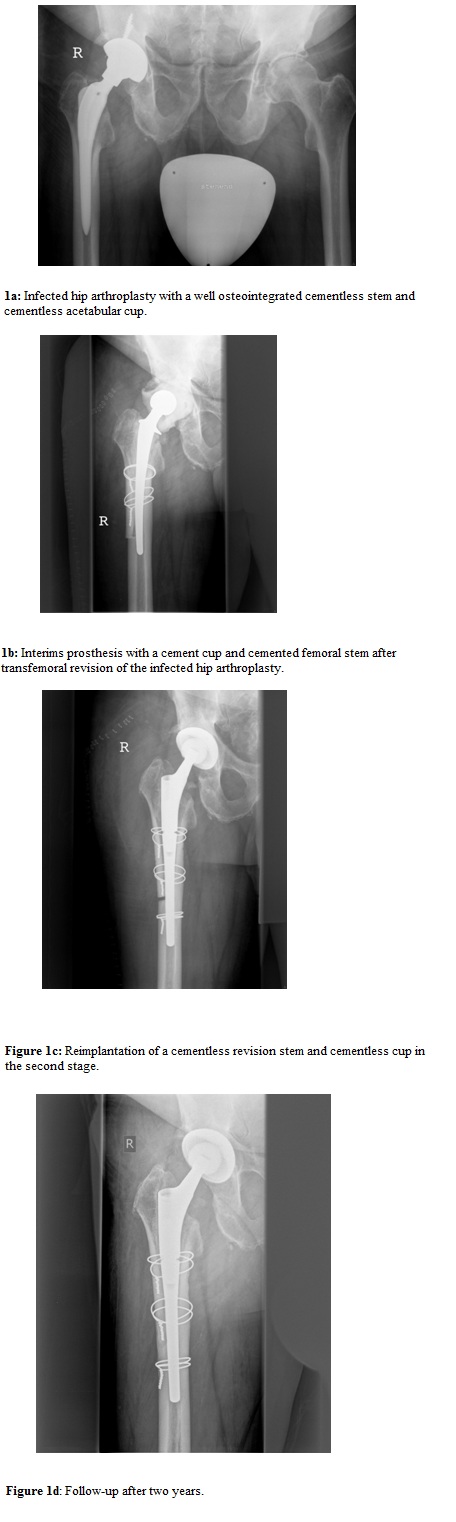
Figure 1: Two-stage revision via a transfemoral approach of an infected total hip prosthesis with a well-fixed cementless stem on the right side.
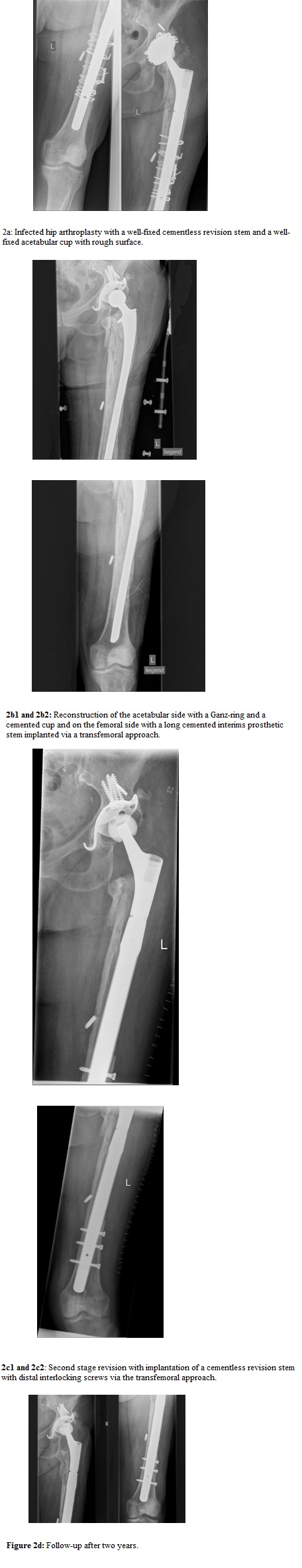
Figure 2: Combinated revision with one-stage revision of the acetabulum and two-stage revision via a transfemoral approach of the femoral stem.
A further important factor in deciding the type of spacer to be used is the amount of damage to the bone caused by the explantation of the infected prosthesis. The removal of well-fixed cemented or cementless infected femoral implants is a challenge for the surgeon. The infected prosthetic bed has to be radically debrided while sparing as much as possible the functionally important areas of bone such as the trochanter major as the attachment area for the gluteal musculature. It is for that reason that we favor the transfemoral approach for removal of well-fixed infected femoral components. This approach enables an effective debridement of the infected femoral component bed and of the osteolytic areas that are often present, while limiting any injury to the trochanter major, the vasto-gluteal muscle loop and the isthmus femoris which represents the area of fixation for the cementless revision stem implanted during the second stage. The endofemoral approach for removing a well-fixed femoral component does not always enable a reproducible debridement of the osteolytic areas and has a higher risk of femoral fracture [32-34]. The transfemoral approach avoids this risk [32-34]. However, it is important that the femoral spacer is long enough to extend beyond the boundaries of the resulting bony flap and that the whole is sufficiently stable. In this procedure, using the transfemoral approach, we favor the closure of the bony flap with cerclage wires in order to avoid migration of the flap, or its dislocation, as described by Morshed et al., [32] (Figures 1 and 2). We reopen the flap during the second stage by removing the cerclage wires so that we can carry out a second radical debridement of the prosthetic bed and ensure that the distally fixed, cementless, modular revision stem is correctly positioned in the isthmus of the femur with the fixation zone distal to the osteotomy (Figures 1 and 2). To analyse the results of the transfemoral approach for revision of infected hip prostheses, 76 septic two-stage revisions involving fixation of the bony flap in the first stage with cerclage wires and reopening of the flap at the time of re-implantation, were followed prospectively, with clinical and radiological assessment, for a mean period of 51.2±23.2 (24-118) months [33]. The rate of complete union of the bony flap after re-implantation was 98.7% and a successful outcome with no recurrence of reinfection was recorded in 93.4% of all cases. Subsidence of the stem occurred at a rate of 6.6 %, dislocation at a rate of 6.6 % and there was no aseptic loosening of the implants. The Harris Hip Score was 62.2±12.6 points with the spacer and 86.6±15.5 points two years after re-implantation of the new implant. Nine fractures (11.8 %) of the flap occurred during the operation due to osteolytic or osteoporotic weakness of the flap itself but these all healed without further intervention [33]. Our data demonstrates that the transfemoral approach is a safe method for septic revision of well-fixed cemented or cementless hip prostheses and that the use of cerclage wires for closing the osteotomy flap in the first stage does not lead to higher reinfection rates.
Another problem associated with spacer implantation is acetabular bone defects. These can lead to situations where a stable fixation of the cement cup or a monoblock spacer is not possible. In such cases and when the infecting organism can be identified, we carry out a one-stage revision whereby the acetabular defect is stabilized by use of the Ganz reinforcement ring, a Burch-Schneider cage or a Cup-Cage-construct into which the cup is then cemented (Figure 3). However, it is sometimes necessary to carry out a two-stage revision of the femoral component using the transfemoral approach for explantation of a septic prosthesis. In such cases, we carry out a combination of a one-stage revision of the acetabular component and a two-stage revision of the femoral component (Figure 2). We analysed 35 such cases with a follow-up of 42.2±17.2 (24-84) months. We found a successful outcome with no recurrence of reinfection in 97.1% of all cases. The Harris Hip Score was 61.2±12.8 points after the first operation and 82.4±15.7 points two years after the second operation [34].
In summary, if spacers can be used they should have a femoral and acetabular component to prevent bony acetabular erosion and should have a metallic endoskeleton on the femoral side to prevent spacer breakage [6,15-17,30].
Local antibiotics in the spacer
Our own in vivo study revealed antibiotic levels in the tissues surrounding the spacer six weeks after implantation that were higher than the minimal inhibitory concentration for the bacteria that had caused the periprosthetic infection. This was demonstrated in 14 two-stage revision septic arthroplasties using spacers containing gentamicin and clindamycin in the cement and also in cases where vancomycin was also included [38]. Ours was the first study to measure antibiotic concentrations in the tissues surrounding the spacer and thus to assess the amount of antibiotic at the site of the later implantation of the new prosthesis.
Not all antibiotics can be used for mixing into the cement because they must be available in powder form, be water-soluble and be thermostable. The most commonly used are gentamicin, clindamycin, vancomycin, tobramycin, aztreonam, ampicillin and ofloxacin [11,39,40]. Most published studies always include the same antibiotics in the cement. Some authors use vancomycin and tobramycin as local antibiotics on a regular basis because they have a broad spectrum of activity [12,25]. However, not all bacteria can be successfully treated with these agents (e.g., some gram-negative organisms). This is also the disadvantage of commercially manufactured spacers that, like the beads, only contain gentamicin or vancomycin as a single antibiotic. So this is an argument for investigating the antibiotic resistance pattern of the isolated bacteria and selecting a specific antibiotic for the treatment. Masri et al., reported a success rate of 89.7% in their retrospective study involving bacteria-specific antibiotic mixed into the cement of a PROSTALAC® spacer (DePuy Orthopaedics, Inc, Warsaw, IN) and we saw no reinfection of 36 cases with a minimum follow-up of 2 years using this concept for handmade spacers [6,26].
Different antibiotics are released at different rates from the spacers and affect each other when in combination with other antibiotics [3]. Of these antibiotics normally mixed in the cement, the use of two antibiotics results in a synergistic effect and the elution of the individual components are better than that of the single antibiotics on their own [39,41-44]. Many surgeons now use cement with gentamicin and clindamycin or gentamicin and vancomycin in combination rather than gentamicin alone because of the better antibiotic elution kinetics exhibited by the former [39,41-44]. A third antibiotic (usually vancomycin) is often added to the manufactured cement containing gentamicin and clindamycin, according to organism specificity defined by an antibiogram [6,42]. This concept enabled us and others to achieve an eradication rate of between 93.5% and 100%, which implies that an adequate level of antibiotic was available in the tissues surrounding the spacer [6,30,38]. However, in our in vivo study, the addition of vancomycin did not result in an increase in the release of the antibiotics present in Copal bone cement, namely gentamicin and clindamycin [28]. Moreover, hand-mixed cement results in a better elution of antibiotics than cement mixed under vacuum. This is because there are air bubbles in the hand-mixed cement that increase the total area of the antibiotic-eluting surface. However, the mechanical properties (resistance to breakage, for example) of the hand-mixed cement are poorer than that of the vacuum-mixed cement [45]. But, the mechanical properties of the spacer cement do not necessarily have to be equivalent to that of the cement used to fix primary endoprosthetic implants, however. Even though the reinfection rate using commercially available spacers with only one antibiotic seems not to be generally higher (Tables 1 and 2), we recommend the addition of several organism-specific antibiotics to the spacer cement because of the described differences in local antibiotic elution. Applying this concept, we were able to show that the local antibiotic concentrations 6 weeks after implantation were still above the relevant minimal inhibitory concentrations. In addition, we observed a very low rate of recurrence - at or around 0% - in the clinical setting [6,26]. Thus, the antibiotic-containing spacer not only fulfills a mechanical function it also plays an important role in the treatment of periprosthetic infections.
Summing up, there is evidence that local antibiotic release from the spacer is helpful for the local treatment of the infection and that a combination of at least two antibiotics and a mixing procedure without vacuum of the spacer cement results in higher local elution of antibiotics.
The duration of spacer period and systemic antibiotic therapy
Aspiration before re-implantation
Type of prosthesis used for reimplantation
DISCUSSION/CONCLUSION
REFERENCES
- Fitzgerald, Robert H (1995) Infected Total Hip Arthroplasty: Diagnosis and Treatment. J Am Acad Orthop Surg 3: 249-262.
- Garvin KL, Hanssen AD (1995) Infection after total hip arthroplasty. Past, present, and future. J Bone Joint Surg Am 77: 1576-1588.
- Cui Q, Mihalko WM, Shields JS, Ries M, Saleh KJ (2007) Antibiotic-Impregnated Cement Spacers for the Treatment of Infection Associated with Total Hip or Knee Arthroplasty. J Bone Joint Surg Am 89: 871-882.
- Hanssen AD, Osmon DR (2002) Evaluation of a staging system for infected hip arthroplasty. Clin Orthop Relat Res 403: 16-22.
- Burnett RS, Kelly MA, Hanssen AD, Barrack RL (2007) Technique and timing of two-stage exchange for infection in TKA. Clin Orthop Relat Res 464: 164-178.
- Fink B, Grossmann A, Fuerst M, Schafer P, Frommelt L (2009) Two-stage cementless revision of infected hip endoprostheses. Clin Orthop Relat Res 467: 1848-1858.
- Garvin KL, Evans BG, Salvati EA, Brause BD (1994) Palacos gentamicin for the treatment of deep periprosthetic hip infections. Clin Orthop Relat Res 298: 97-105.
- Lieberman JR, Callaway GH, Salvati EA, Pellicci PM, Brause BD (1994) Treatment of the infected total hip arthroplasty with a two-stage reimplantation protocol. Clin Orthop Relat Res 301: 205-212.
- Evans RP (2004) Successful treatment of total hip and knee infection with articulating antibiotic components: a modified treatment method. Clin Orthop Relat Res 427: 37-46.
- Goldman RT, Scuderi GR, Insall JN (1996) 2-stage reimplantation for infected total knee replacement. Clin Orthop Relat Res 331: 118-124.
- Hofmann AA, Goldberg TD, Tanner AM, Cook TM (2005) Ten-year experience using an articulating antibiotic cement hip spacer for the treatment of chronically infected total hip. J Arthroplasty 20: 874-879.
- Fehring TK, Calton TF, Griffin WL (1999) Cementless fixation in 2-stage reimplantation for periprosthetic sepsis. J Arthroplasty 14: 175-181.
- Haddad FS, Muirhead-Allwood SK, Manktelow AR, Bacarese-Hamilton I (2000) Two-stage uncemented revision hip arthroplasty for infection. J Bone Joint Surg Br 82: 689-694.
- Hsieh PH, Shih CH, Chang YH, Lee MS, Shih HN, et al. (2004) Two-stage revision hip arthroplasty for infection: comparison between the interim use of antibiotic-loaded cement beads and a spacer prosthesis. J Bone Joint Surg Am 86: 1989-1997.
- Leunig M, Chosa E, Speck M, Ganz R (1998) A cement spacer for two-stage revision of infected implants of the hip joint. Int Orthop 22: 209-214.
- Mitchell PA, Masri BA, Garbuz DS, Greidanus NV, Duncan CP (2003) Cementless revision for infection following total hip arthroplasty. Instr Course Lect 52: 323-330.
- Disch AC, Matziolis G, Perka C (2007) Two-stage operative strategy without local antibiotic treatment for infected hip arthroplasty: clinical and radiological outcome. Arch Orthop Trauma Surg 127: 691-697.
- McDonald DJ, Fitzgerald RH Jr, Ilstrup DM (1989) Two-stage reconstruction of a total hip arthroplasty because of infection. J Bone Joint Surg Am 71: 828-834.
- Colyer RA, Capello WN (1994) Surgical treatment of the infected hip implant: Two-stage reimplantation with a one-month interval. Clin Orthop Relat Res 298.
- Younger AS, Duncan CP, Masri BA, McGraw RW (1997) The outcome of two-stage arthroplasty using a custom-made interval spacer to treat the infected hip. J Arthroplasty 12: 615-623.
- Hsieh PH, Chen LH, Chen CH, Lee MS, Yang WE, et al. (2004) Two-stage revision hip arthroplasty for infection with a custom-made, antibiotic-loaded, cement prosthesis as an interim spacer. J Trauma 56: 1247-1252.
- Wilson MG, Dorr LD (1989) Reimplantation of infected total hip arthroplasties in the absence of antibiotic cement. J Arthroplasty 4: 263-269.
- Nestor BJ, Hanssen AD, R. Ferrer-Gonzalez R, Fitzgerald RH Jr (1994) The use of porous prostheses in delayed reconstruction of total hip replacements that have failed because of infection. J Bone Joint Surg Am 76: 349-359.
- Koo KH, Yang JW, Cho SH, Song HR, Park HB, et al. (2001) Impregnation of vancomycin, gentamicin, and cefotaxime in a cement spacer for two-stage cementless reconstruction in infected total hip arthroplasty. J Arthroplasty 16: 882-892.
- Kraay MJ, Goldberg VM, Fitzgerald SJ, Salata MJ (2005) Cementless two-staged total hip arthroplasty for deep periprosthetic infection. Clin Orthop Relat Res 441: 243-249.
- Masri BA, Panagiotopoulos KP, Greidanus NV, Garbuz DS, Duncan CP (2007) Cementless two-stage exchange arthroplasty for infection after total hip arthroplasty. J Arthroplasty 22: 72-78.
- Yamamoto K, Miyagawa N, Masaoka T, Katori Y, Shishido T, et al. (2009) Cement spacer loaded with antibiotics for infected implants of the hip joint. J Arthroplasty 24: 83-89.
- Berend KR, Lombardi AV Jr, Morris MJ, Bergeson AG, Adams JB, et al. (2013) Two-stage treatment of hip periprosthetic joint infection is associated with a high rate of infection control but high mortality. Clin Orthop Relat Res 471: 510-518.
- Camurcu Y, Sofu H, Buyuk AF, Gursu S, Kaygusuz MA, et al. (2015) Two-stage cementless revision total hip arthroplasty for infected primary hip arthroplasties. J Arthroplasty 30: 1597-1601.
- Hsieh PH, Shih CH, Chang YH, Lee MS, Yang WE, et al. (2005) Treatment of deep infection of the hip associated with massive bone loss: two-stage revision with an antibiotic-loaded interim cement prosthesis followed by reconstruction with allograft. J Bone Joint Surg Br 87: 770-775.
- Fink B, Rechtenbach A, Buchner H, Vogt S, Hahn M (2011) Articulating spacers used in two-stage revision of infected hip and knee prostheses abrade with time. Clin Orthop Relat Res 469: 1095-1102.
- Morshed S, Huffman GR, Ries MD (2005) Extended trochanteric osteotomy for 2-stage revision of infected total hip arthroplasty. J Arthroplasty 20: 294-301.
- Fink B, Oremek D (2016) The Transfemoral Approach for Removal of Well-Fixed Femoral Stems in 2-Stage Septic Hip Revision. J Arthroplasty 31: 1065-1071.
- Fink B, Schlumberger M, Oremek D (2017) Single-stage Acetabular Revision During Two-stage THA Revision for Infection is Effective in Selected Patients. Clin Orthop Relat Res 475: 2063-2070.
- Masri BA, Duncan CP, Beauchamp CP (1998) Long-term elution of antibiotics from bone-cement: an in vivo study using the prosthesis of antibiotic-loaded acrylic cement (PROSTALAC) system. J Arthroplasty 13: 331-338.
- Hsieh PH, Chang YH, Chen SH, Ueng SW, Shih CH (2006) High concentration and bioactivity of vancomycin and aztreonam eluted from Simplex cement spacers in two-stage revision of infected hip implants: a study of 46 patients at an average follow-up of 107 days. J Orthop Res 24: 1615-1621.
- Bertazzoni Minelli E, Benini A, Magnan B, Bartolozzi P (2004) Release of gentamicin and vancomycin from temporary human hip spacers in two-stage revision of infected arthroplasty. J Antimicrob Chemother 53: 329-334.
- Fink B, Vogt S, Reinsch M, Buchner H (2011) Sufficient release of antibiotic by a spacer 6 weeks after implantation in two-stage revision of infected hip prostheses. Clin Orthop Relat Res 469: 3141-3147.
- Anagnostakos K, Kelm J, Regitz T, Schmitt E, Jung W (2005) In vitro evaluation of antibiotic release from and bacteria growth inhibition by antibiotic-loaded acrylic bone cement spacers. J Biomed Mater Res B Appl Biomater 72: 373-378.
- Hoff SF, Fitzgerald RH Jr, Kelly PJ (1981) The depot administration of penicillin G and gentamicin in acrylic bone cement. J Bone Joint Surg Am 63: 798-804.
- Baleani M, Persson C, Zolezzi C, Andollina A, Borrelli AM, et al. (2008) Biological and biomechanical effects of vancomycin and meropenem in acrylic bone cement. J Arthroplasty 23: 1232-1238.
- Ensing GT, van Horn JR, van der Mei HC, Busscher HJ, Neut D (2008) Copal bone cement is more effective in preventing biofilm formation than Palacos R-G. Clin Orthop Relat Res 466: 1492-1498.
- Penner MJ, Masri BA, Duncan CP (1996) Elution characteristics of vancomycin and tobramycin combined in acrylic bone-cement. J Arthroplasty 11: 939-944.
- Simpson PM, Dall GF, Breusch SJ, Heisel C (2005) [In vitro elution and mechanical properties of antibiotic-loaded SmartSet HV and Palacos R acrylic bone cements]. Orthopade 34: 1255-1262.
- Hanssen AD, Spangehl MJ (2005) Practical applications of antibiotic-loaded bone cement for treatment of infected joint replacements. Clin Orthop Relat Res 427: 79-85.
- Mont MA, Waldman BJ, Hungerford DS (2000) Evaluation of preoperative cultures before second-stage reimplantation of a total knee prosthesis complicated by infection. A comparison-group study. J Bone Joint Surg Am 82: 1552-1557.
- Schafer P, Fink B, Sandow D, Margull A, Berger I, et al. (2008) Prolonged bacterial culture to identify late periprosthetic joint infection: a promising strategy. Clin Infect Dis 47: 1403-1409.
- Preininger B, Janz V, von Roth P, Trampuz A, Perka CF, et al. (2017) Inadequacy of Joint Aspiration for Detection of Persistent Periprosthetic Infection During Two-Stage Septic Revision Knee Surgery. Orthopedics 40: 231-234.
- Hoell S, Moeller A, Gosheger G, Hardes J, Dieckmann R, et al. (2016) Two-stage revision arthroplasty for periprosthetic joint infections: What is the value of cultures and white cell count in synovial fluid and CRP in serum before second stage reimplantation? Arch Orhtp Trauma Surg. 136: 447-452.
- Frangiamore SJ, Siqueira MB, Saleh A, Daly T, Higuera CA, et al. (2016) Synovial cytokines and the MSIS criteria are not useful for determing infection resolution after periprosthetic joint infection explantation. Clin Orthop Relat Res 474: 1630-1639.
- Fink B, Fuerst M, Hahn M, Thybaud L, Sieber HP, et al. (2005) [Principles of fixation of the cementless modular revision stem Revitan]. Unfallchirurg 108: 1029-1032, 1034-1037.
- Wirtz DC, Niethard FU (1997) [Etiology, diagnosis and therapy of aseptic hip prosthesis loosening--a status assessment]. Z Orthop Ihre Grenzgeb 135: 270-280.
Citation: Fink B, Sevelda F, Frommelt L (2017) Pros and Cons of Spacers in the Treatment of Late Periprosthetic Infections of the Hip. J Infect Non Infect Dis 3: 026.
Copyright: © 2017 Bernd Fink, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

