
Pulmonary Embolism with Negative D-Dimer
*Corresponding Author(s):
Syed Saleh Syed SheikhDepartment Of Internal Medicine, International Islamic University, Malaysia
Email:syedsaleh86@gmail.com
Abstract
Venous thromboembolism is condition characterized by the presence of blood clot that resides in the venous system. It encompasses deep vein thrombosis which is represented by development of de novo blood clot in the deep venous system of the limbs (commonly lower limbs) and pulmonary embolism, which emerges as the preformed blood clots mobilize and occlude the pulmonary circulation. Here, we present a case of a 44 year old lady who was diagnosed with pulmonary embolism proven by CT pulmonary angiogram despite having a negative d-dimer assay.
Introduction
Venous thromboembolism (VTE) accounts for the third leading cause of death worldwide with mortality rate ranges from 9.4 to 32.3 per 100,000 population [1]. Mostly it encompasses of Pulmonary Embolism (PE) and Deep Vein Thrombosis (DVT). In these latest years, there is increase awareness of this clinical condition, especially in diagnosing PE due to its fatality. Wide availability and accessibility to Computed Tomography Pulmonary Angiogram (CTPA) as the gold standard diagnostic investigation for PE makes it more prevalent. There are few clinical probability scores developed; Wells score, revised Geneva score and YEARS clinical decision rule. These in combination with D-dimer level help to guide clinicians in diagnosing PE [2,3]
D-dimer is a fibrin degradation product and is raised in PE due to active thrombolysis process that occurs. It holds a strong position in diagnosing PE due to its high Negative Predictive Value (NPV) and sensitivity. D-dimer level can be interpretated based on age-related cut-off level or adapted to clinical probability scores as mentioned above [2-5]. However, there are certain factors that may leads to its false negative result. This case study is addressing a clinically uncommon condition of pulmonary embolism with negative D-dimer.
Case Presentation
A 44-year-old lady with a background medical history of bronchial asthma, uterine fibroid and carpal tunnel syndrome presented to orthopedic clinic for her regular follow up. On presentation, she was noted lethargic by the clinic staff and tachycardic with heart rate of 120 beats per minute. On top of that, she complained of chest tightness. She was given 6 rescue doses of MDI Salbutamol and Ipratropium Bromide with which response was seen. She was immediately sent to the emergency department. On arrival at the emergency department, she has already felt some improvement than before as shown by absence of any clinical signs of respiratory dysfunction. her breath sound was equally distributed bilaterally throughout both lungs. Her cardiovascular system was noted to be unremarkable. However, despite the improvement of her she was still noted to be tachycardic with heart rate of 130 beats per minute.
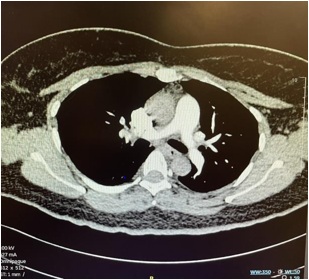
An ECG was performed showing presence of sinus tachycardia with S1Q3T3 pattern. There was absence of right bundle branch block or other evidence suggestive of pulmonary hypertension. Further history taken disclosed that she has been consuming oral contraceptive pill. The constellation of symptoms and signs contribute to a Wells clinical probability score of 4.5 for pulmonary embolism, thus, placing her at moderate risk. No hypoxia or respiratory failure noted from Arterial blood gase while her D-Dimer was < 200 ng/mL. Considering this occasion accompanied by the fact that she has been taking oral contraception, (CTPA). CTPA demonstrated a small pulmonary arterial contrast filling defect within the first-degree branch of the anterior segment of the right upper lobe artery, before its bifurcation. Transthoracic echocardiogram showed left ventricular ejection fraction of 74% without features of pulmonary hypertension and right ventricular dysfunction. Thus, based on these evidences, she was diagnosed with pulmonary embolism provoked by oral contraception. She received oral rivaroxaban 15mg twice daily for three weeks and subsequently 20mg daily for three months.
Discussion
Venous thromboembolism (VTE) accounts for the third leading cause of death worldwide, with its mortality rate ranges from 9.4 to 32.3 per 100,000 population [1]. This encompasses pulmonary embolism and deep vein thrombosis. There is increase awareness among clinicians in diagnosing PE due to its fatality. Together with wide availability of computed tomography pulmonary angiogram (CTPA); the gold standard diagnostic investigation for PE, its usage has blossom exponentially [6]. There are few clinical prediction scoring rules developed to guide clinicians; Wells score, revised Geneva score and YEARS clinical decision rule. These clinical probability scorings, in combination with D-dimer testing help to avoid overuse of CTPA in diagnosing PE [2,3].
D-dimer is a fibrin degradation product produced through fibrinolysis process. This process happens as a sequalae of thrombosis that occurs in PE. Its usage helps to exclude PE in low clinical probability population given its high Negative Predictive Value (NPV) and diagnostic sensitivity. D-dimer can be interpretated based on age- related cut-off level or adapted to clinical probability using the aforementioned scoring rules [2]. YEAR’s clinical decision rule or algorithm was published in 2017 which entails three clinical items (hemoptysis, clinical signs of DVT, and PE as the most likely diagnosis) plus D-dimer level. According to this, PE is excluded in the absence of clinical item with D-dimer level less than 1000ng/mL or at least one clinical item with D-dimer level less than 500ng/mL [4]. As for Wells, Geneva and revised Geneva rules, their usage in excluding PE with negative D-dimer (less than 500 ng/mL) have been extensively analyzed and established [3,5].
Pulmonary embolism manifestations are heterogenous, with commonly reported symptoms being dyspnea, chest pain, syncope, and cough [7]. In individual with pre-existing pulmonary disease as in this case, worsening dyspnea can be the only symptom to suggest PE and presence of tachycardia occurs in estimated 40% of cases with PE [2]. These, together with electrocardiogram (ECG) changes of prominent S wave in lead I, and presence of Q wave and T wave inversion (S1Q3T3) in lead III, led to the suspicion of PE after ruling out other causes of the signs and symptoms in spite of no hypoxemia recorded in the arterial blood gas. According to available literatures, up to 40% of PE patients have absence of hypoxemia [2,7] Risk factors for developing PE have become a common knowledge to most clinicians. In this case, the noticeable risk factor was consumption of oral contraceptive pill.
Based on available validated clinical probability scores, this case fell into the category of moderate risk for PE, both by Wells and revised Geneva Score and low risk according to YEARS score. Taking into consideration of the negative D-dimer value of less than 200ng/mL, adapted to clinical probability scores (Wells, revised Geneva score and YEARS algorithm), there was no role for proceeding further with CTPA to investigate for PE. However, due to persistent sign and symptom and high clinical suspicion, CTPA was performed and proved the presence of PE. Pulmonary embolism with negative D-dimer is uncommon but there are few reported cases and retrospective studies looking into this unique condition [8-11].
Timing of blood sampling for D-dimer testing may lead to false negative D-dimer; eg: blood sampling taken after receiving anti-coagulant, sampling done too early during early formation of clot or when the clot has organized (approximately later than seven days) [9]. Published literature reported that, D-dimer level falls approximately 25% after 24-hour of receiving anti-coagulant and this leads to reduction in its sensitivity from 95.6% to 89.4% [12]. Presence of heterophilic antibodies, autoantibodies, and cross- reactive substances may also leads to false negative D-dimer. This can be tackled by using heterophilic antibody blocking reagent and dilution test [13]. A hypothesis on different D-dimer assays may affect D-dimer level is addressed by a post-hoc analysis of YEARS study. It analyzes the differences in median D-dimer level captured by different assays, by which the difference is statistically significant. However, the NPV are comparable between these different assays (99.5% to 100%) in low clinical probability group [14]. This is further supported by systematic review and meta-analysis that shows even less sensitive D-dimer assay in combination with clinical prediction scores is strong enough to rule out PE in moderate risk clinical probability group [5].
Tailoring back to this case, D-dimer level was taken prior to initiation of anti- coagulant. However, it is unfeasible to ascertain whether the sampling was performed too soon before clot formation. Assay used to measure D-dimer level was Dialab D- Dimertest Latex, a rapid agglutination assay with serial dilution technique. It is tested to have no interference with potential substances with test specificity of 95.3%. As proven by CTPA coupled with the consumption of oral contraceptive pill as part of the risk factors for PE, this patient was diagnosed as pulmonary embolism. She was treated with oral anticoagulant for three months.
Conclusion
This case study represents the uncommon condition of PE with negative D- dimer. Though uncommon, more cases have been detected due to increase in clinical suspicion of PE and easy accessibility to CTPA. Factors that may lead to negative D- dimer value include timing of D-dimer sampling, and the probability of interference by cross-reactive substances and autoantibodies during testing of which, these factors can be avoided and minimized. It is important to remember, the decision to further investigate for PE lies not solely on D-dimer level adapted to clinical probability scores, but rather also involves the high clinical suspicion by treating clinicians.
References
- Wendelboe AM, Raskob GE (2016) Global Burden of Thrombosis: Epidemiologic Aspects. Circ Res 118: 1340-1347.
- Konstantinides SV, Meyer G, Becattini C, Bueno H, Geersing GJ, et al. (2020) 2019 ESC Guidelines for the diagnosis and management of acute pulmonary embolism developed in collaboration with the European respiratory society (ERS). Eur Heart J 41: 543-603.
- Guo DJ, Zhao C, Zou YD, Huang XH, Hu JM, et al. (2015) Values of the wells and revised Geneva scores combined with D-dimer in diagnosing elderly pulmonary embolism patients. Chin Med J (Engl) 128: 1052-1057.
- Van der Hulle T, Cheung WY, Kooij S, Beenen LFM, van Bemmel T, et al. (2017) Simplified diagnostic management of suspected pulmonary embolism (the YEARS study): a prospective, multicentre, cohort study. Lancet (London, England) 390: 289-297.
- Ceriani E, Combescure C, Le Gal G, Nendaz M, Perneger T, et al. (2010) Clinical prediction rules for pulmonary embolism: A systematic review and meta-analysis. J Thromb Haemost 8: 957- 970.
- Wiener RS, Schwartz LM, Woloshin S (2013) When a test is too good: How CT pulmonary angiograms find pulmonary emboli that do not need to be found. BMJ 347: f3368.
- Morrone D, Morone V (2018) Acute Pulmonary Embolism: Focus on the Clinical Picture. Korean Circ J 48: 365.
- Hannoodee H, Khanam V, Taheri Abkouh D, Akyuz K, I Kulairi Z (2022) Acute Pulmonary Embolism In A Patient With A Normal D-Dimer. Chest 162: A1141.
- Patel K, Patel R, Miller R, Trivedi K (2020) A Diagnostic Dilemma: Normal D-Dimer In A Patient With Extensive Pulmonary Embolism. Chest 158: A2119- A2120.
- Dhananjaya M, Meti K, Parakh R (2018) D-dimer negative pulmonary embolism. Int J Adv Med 5: 429.
- Sikora-Skrabaka M, Skrabaka D, Ruggeri P, Caramori G, Skoczynski S, et al. (2019) D-dimer value in the diagnosis of pulmonary embolism -may it exclude only?. J Thorac Dis 11: 664-672.
- Couturaud F, Kearon C, Bates SM, Ginsberg JS (2002) Decrease in sensitivity of D-dimer for acute venous thromboembolism after starting anticoagulant therapy. Blood Coagul Fibrinolysis. 13: 241-246.
- Zhang XY, Zhang XX, Xu JL, Huang TY, Wu Y (2020) Identification of and solution for false D-dimer results. J Clin Lab Anal 34: e23216.
- Hamer HM, Stroobants AK, Bavalia R, Ponjee GAE, Klok FA (2021) Diagnostic accuracy of four different D- dimer assays: A post-hoc analysis of the YEARS study. Thromb Res 201: 18-22.
Citation: Sheikh SSS, Jamhuri NS (2023) Pulmonary Embolism with Negative D-Dimer. J Pulm Med Respir Res 9: 076.
Copyright: © 2023 Syed Saleh Syed Sheikh, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

