Role of Chamomile in Cancer Treatment
*Corresponding Author(s):
Thalluri Gouri Sai KiranDepartment Of Pharmacology, Vignan Institute Of Pharmaceutical Technology, Visakhapatnam, India
Tel:+91 891 2511222,
Email:tgsaikiran@gmail.com
Abstract
Chamomile is the daisy like plants of the family Asteraceae. Mainly two species are commonly used to make herb infusions thought to have medicinal uses, they are Matricaria Chamomilla is an aromatic plant with antioxidant, anticancer, and anti-inflammatory properties. Chamaemelum nobile commonly known as Roman chamomile is a medicinal plant used for numerous diseases in traditional medicine, although its anticancer activity. Chamomile contains 0.5-3% flavonoids. The most relevant flavonoids are flavones (mostly apigenin, also luteolin) and flavonols (quercetin, isorhamnetin, myricetin, patuletin). Both in vivo and in vitro significant progress have been made in studying the chemo-preventive aspects of apigenin. Several studies have demonstrated that the anti-carcinogenic properties of apigenin occur through regulation of cellular response to oxidative stress and DNA damage, suppression of inflammation and angiogenesis, retardation of cell proliferation, and induction of autophagy and apoptosis. One of the most well recognized mechanisms of apigenin is the capability to promote cell cycle arrest and induction of apoptosis through of autophagy in several human cancer cell lines. Apigenin induces the apoptosis of colon cancer cell by inhibiting the phosphorylation of STST3 and consequently downregulates the anti apoptotic proteins Bcl-XL and Mcl-1. In this review, we discuss the details chamomile, apigenin, apoptosis, autophagy and the role of apigenin in cancer prevention via the induction of apoptosis and autophagy.
Keywords
Angiogenesis; Anti-cancer; Apigenin; Apoptosis; Autophagy; Chamomile
INTRODUCTION
Chamomile is one of the important medicinal herb native to southern and eastern Europe. It was also grown in Germany, Hungary, France, Russia, Yugoslavia and Brazil. The plants can be found in North Africa, Asia, North & South America, Australia & New Zealand. In India it was introduced during the Mughal period, now it is grown in Punjab, Uttar Pradesh, and Maharashtra, Jammu and Kashmir (Figure 1) [1]. Hungary is the main producer of the plant biomass, it also grows abundantly in poor soils and it is a source of income to the poor inhabitants of these area flowers which are exposed to Germany in the bunk for distillation of the oil [2]. In India, the plant had been cultivated in Lucknow for about 200 years and the plant was introduced in Punjab about 300 years age during the Mughal period. It was introduced in Jammu in 1957 by Handa et al. [3], the plant was introduced in the alkaline soils of Lucknow in 1964-1965 by Chandra et al. [4,5].

Figure 1: Chamaemelum nobile.
Cancer is one of the major causes of death worldwide in which deregulated proliferation of abnormal cells leads to disruption of surrounding tissues. Based on the report of the international agency for the research on cancer cases which were identified around the world in 2012, of these 7.7 million cases were men and 6.9 million women and further this number is expected to increase to 24 million by 2035 [6]. The most common cancers are lung, breast, and colorectal cancers. Breast cancer is the first leading cause of cancer death in women in 140 countries. It contributes to one fourth of all types of cancer in women. Generally, cancer has an increasing trend in developing countries due to lifestyle change such as diet change [7]. Medicinal plants have long been used in the treatment of different types of dieases due to less toxicity compared with the modern chemotherapy. Chamomile as a well reputed medicinal plant in the world was widely used for different diseases. Phytochemicals in Matricaria Chamomilla flower extract include different acids such as tartaric acid, citric acid, and succinic acid (Figure 2).

Figure 2: Kamomillasaunio (Matricaria Chamomilla).
It has other compounds such as myristin, proazolene, luteolin and coumarin derivatives as well as different flavonoids such as flavones and flavonols. Its florets contain rutin, apigenin, and free quercetin. Oxygen free radicals contribute to the pathophysiology of many diseases, including cancer and inflammation [8]. Methonolic and aqueous extracts of M. Chamomilla have the least anti-proliferative effect on normal cells while significantly affects biological ability of different cancer cells. Studies suggest that bisabolol oxide A, a compound of M. Chamomile, together with fluorouracil-5, exhibits anti-proliferative action on K562 cell line in blood cancer [9].
Many researchers have reported that M. Chamomile has pharmacological properties, including antimicrobial, anti-inflammatory, antioxidative, antispasmodic, antiviral, and sedative activities owing to the terpenoids, flavonoids (such as apigenin and luteolin), coumarins, and spiroethers in the plant. Recently, it has been studied as a therapeutic agent against aphthous stomatitis [10]. In one of the recent studies about n160 high confidence candidate apigenin target molecules were identified which were divided into three functional categories: GTPase activation, membrane transport, and mRNA metabolism/alternating splicing. Other research papers have also revealed of apigenin targets (Table 1) [11].
|
Gene Experssion |
Protein Kinases |
Transcription Factors |
Enzymes |
Membrane Proteins |
Others |
|
Cyclin D |
Ikk/IKB Kinase |
STAT-3 |
FTPase |
VCAM |
Bcl-2 |
|
Cyclin A |
JAK |
NF-Kb |
GST |
ICAM |
Bcl-xl |
|
Cyclin B1 |
Src |
AP-1 |
GSH-Px |
VEGF |
EGFR |
|
Cyclin E |
JNK |
PI-3 |
GSH-R |
EGFR |
Bax |
|
5-LOX |
HER-2 |
Egr-1 |
CAT |
MRPs |
P53 |
|
COX-1 |
Akt/PKB |
Erβ |
SOD |
MMPs |
P21 |
|
COX-2 |
PKA |
CBP |
ALT |
FAK |
P27 |
|
P53 |
PKC |
PPAR |
ALP |
IGF-1R |
Trail |
|
P21 |
P13K |
EpRE |
GGT |
VEGF |
Apc |
|
P27 |
MAPK |
Β-catenin |
LDH |
Pten |
|
|
IL-6 |
Nrf-2 |
AST |
IGF-1 |
||
|
IL-5 |
HIF-1α |
XO |
XIAP/IAP |
||
|
IL-8 |
Elk-1 |
HO |
Β-CTF |
||
|
IL-12 |
GATA-3 |
ODC |
BDNF |
||
|
IL-17 |
Cyt-P450 |
TGF-β |
|||
|
TNF-α |
Aromatase |
HO-1 |
|||
|
c-Fos |
Caspase 3,9 |
||||
|
AChE |
|||||
|
BChE |
Table 1: Molecular targets of apigenin.
AN OVERVIEW OF MOLECULAR TARGETS OF APIGENIN
In one of the recent studies about n160 high confidence candidate apigenin target molecules were identified which were divided into three functional categories: GTPase activation, membrane transport, and mRNA metabolism/alternating splicing. Other research papers have also revealed of apigenin targets, summarized in table 1 [11].
ROLE OF APIGENIN IN CANCER
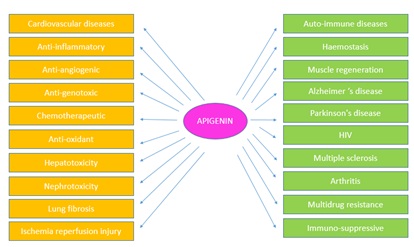
Many of the biological effects of apigenin in numerous mammalian systems in vitro as well as in vivo are related to TIS antioxidant effects and its role in scavenging free radicals. Furthermore, it exhibits anti mutagenic, anti-inflammatory, antiviral, and purgative effects. The actions of apigenin in inhibiting the cell cycle, diminishing oxidative stress, improving the efficacy of detoxification enzymes, including apoptosis, and stimulating the immune system are quite limited. One human study demonstrated that apigenin was absorbed systemically by a subject fed a diet high in parsley; this subject was found to have elevated levels of the antioxidant enzymes erythrocyte glutathione reductase and superoxide dismutase. Activities of erythrocyte catalase and glutathione peroxidase however, were found to be unchanged. Other biological effects induced by flavonoids include reduction of cell proliferation. This is apparent from another cross-sectional study conducted in Japan in which total intake of flavonoids among women was found to be inversely correlated with plasma total cholesterol and low density lipoprotein concentration, after adjustment for age, body mass index and total energy intake. The effects of flavonoids on the hematologic systems were performed, a 7 day study of 18 healthy men and women examining the effects of a daily dietary supplement providing quercetin (377±10 miles from onions) and apigenin (84±6 mg from parsley) on platelet aggregation and other hemostatic variables. They observed no significant changes in collagen or ADP induced platelet number, factor VII, plasminogen, PAI activity or fibrinogen concentrations. These inherent properties of flavonoids categorize them as a class of beneficial compounds which possess health promoting and disease preventing dietary effects (Figure 3) [12].
ACTIONS OF APIGENIN
• Apigenin induced apoptosis, activated caspases and cleaved PARP in dose and time dependent manners in U937 cells
• Exposure of U937 cells to apigenin resulted in downregulation of Bcl-2 and Mcl-1
• Exposure of U937 cells to apigenin resulted in the inactivation of Akt and pronounced increase in JNK activation
• Apigenin induced apoptosis in leukemia cells via caspase-independent inactivation of Akt and activation of JNK
• Apigenin induced apoptosis in leukemia cells via mitochondrial dependent mechanism
• Apigenin induced similar effects in other leukemia cells but not in NPBMNCs
• Inactivation of Akt is responsible for apigenin induced JNK and caspases activation and apoptosis
• Activation of JNK played an important role in apigenin-induced caspase activation and apoptosis
• Apigenin induced Mcl-1 downregulation proceeds via transcriptional and proteasome-dependent mechanisms
• Overexpression of Mcl-1 substantially diminished apigenin-induced apoptosis, caspases activation and PARP cleavage in U937 cells
• Apigenin inhibited tumor formation in xenografts of U937 human leukemia cells
• Inhibition of PI3K/Akt and mTOR signaling pathways by apigenin [13]
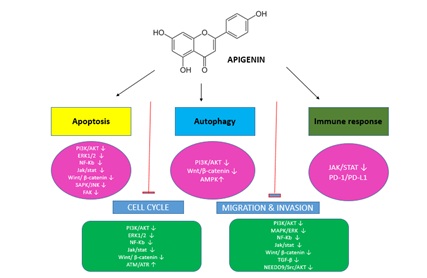
INDUCTION OF APOPTOSIS
Apoptosis is the natural process of programmed cell death. Apoptosis involves energy-dependent cascade events and different distinct morphological characteristics [14]. To date, apoptosis is induced by two core pathways: The extrinsic (Death receptor) pathway and the intrinsic (mitochondrial) pathway. Apoptosis is a critical process that allows undesirable cells to be removed under physiological conditions. Avoiding apoptosis is one of the most important characteristics of cancer cells that make them different from normal cells. Thus, triggering cancer cell apoptosis by targeting apoptotic pathways with chemotherapy reagents is a widely used strategy to treat cancer. Apigenin has been demonstrated to be an effective agent for triggering apoptosis via either the intrinsic pathway in human cancer cells.
The intrinsic apoptotic pathway is regulated by the Bcl-2 family of proteins, such as Bcl-2, Bcl-xL, Bcl-w and Mcl-1, which block apoptosis, while Bad, Bax, Bax, Bid and Bim trigger apoptosis [15]. Apigenin functions to upregulate pro-apoptotic proteins and/or downregulate pro survival members, there by inducing the intrinsic apoptotic pathway. In prostate cancer therapy, treatment of the androgen-refractory human prostate cancer cell lines PC-3 and DU145 with apigenin resulted in apoptosis and a reduction in cell viability caused by a decrease in Bcl-2 and Bcl-xL and an increase in the active form of the Bax protein, accompanied by dose-dependent suppression of XIAP, c-IAP1, c-IAP2 and surviving proteins [16]. In addition, in human promyelocytic leukemia HL-60 cells, apigenin reduced the mitochondrial outer membrane potential, released cytochrome c from the mitochondria into the cytosol, induced procaspase-9 processing and finally induced cell apoptosis through the intrinsic apoptotic pathway [17]. In other reports, apigenin caused cell apoptosis by changing the ratio of pro-apoptotic to pro-survival mitochondrial proteins. Apigenin increased the Bax/Bcl-2 inhibitor ABT-263 to trigger mitochondria-dependent cell apoptosis [18].
Seo et al., [19], found that apigenin neither affected the levels of Bcl-2 and Bax nor decreased the mitochondrial membrane potential in the human breast cancer BT-474 cells, but this compound induced extrinsic, caspase dependent apoptosis by upregulating the levels of cleaved caspase-8 and cleaved caspase-3 [20]. In Non-Small Cell Lung Cancer (NSCLC) cells, Chen et al., [21], showed that apigenin upregulated the levels of Death Receptor 4 (DR4) and Death Receptor 5 (DR5) in a p53-dependent manner, thereby sensitizing NSCLC cells to trail induced apoptosis. Meanwhile, apigenin triggered the intrinsic apoptotic pathway by upregulating the pro-apoptotic proteins Bad and BAX and downregulating the anti-apoptotic proteins Bcl-XL and Bcl-2 [21]. Moreover, in human keratinocytes and organotypic keratinocytes, apigenin increased UVB-induced apoptosis via both the intrinsic and extrinsic apoptotic pathways as well. Apigenin caused changes in Bax localization and in the release of cytochrome c. Over expression of the pro-survival protein Bcl-2 and the dominant-negative form of the fast-associated death domain protected against apigenin-induced apoptosis [22].
INDUCTION OF AUTOPHAGY
Autophagy, the so called type 2 non-apoptotic cell death, is characterized by the sequestration of cytoplasmic material into vacuoles for bulk degradation by lysosomal enzymes. Autophagy is a dynamic process where the cell digests its own cytoplasmic materials within lysosomes and results in the sequestration and degradation macromolecules [23]. In some cases, autophagy can serve as a cell survival pathway by providing recycled metabolic substrates and maintaining energy homeostasis during starvation, while in other settings, it can cause cell death, either in collaboration with apoptosis or as a backup mechanism. This is growing evidence that the relationship between autophagy and cancer is complex and contradictory. Autophagy triggered by apigenin was first observed in erythroleukemia TF1 cells. Apigenin treatment triggered the initiation of autophagy without apoptosis [24]. Since then, more evidences have been presented that apigenin could induce autophagy which serves as tumor suppressive or a tumor protective role under different circumstances [25,26].
Tong et al., [27] reported that apigenin exerted its chemo preventive by inducing autophagy in human keratinocytes via activation of AMPK [28]. In human breast cancer T47D and MDA-MB-231 cells, cao et al., found that the apigenin exposure triggered cell apoptosis and autophagy as evidenced by the accumulation of Acidic Vesicular Organelles (AVOs) and LC3-II, a marker of Atg5/atg7 dependent autophagy. Further, the authors found that treatment with autophagy induced by apigenin play a tumor protective role in apigenin-caused cytotoxicity [27]. Similarly, in human colon cancer HCT116 cells, Lee et al., [29] proved that apigenin concomitantly caused apoptosis and autophagy and autophagy played a cell protective role in apigenin-induced cell apoptosis as well [30].
Beclin-1 regulates the dynamic autophagy process via the formation of auto phagosomes [31,32]. Beclin-1 is frequently downregulated in many types of cancers, including solid Ehrlich carcinoma. Gaballah et al., [33] found that combining 5-FU with apigenin significantly increased Beclin-1 compared with the vehicle-treated control mice [33]. In addition, showed that apigenin treatment induced autophagy in macrophages as evidenced by upregulation of Beclin 1, Atg5, Atg7 and the appearance of LC3-II and autophagy inhibition by 3-MA pretreatment significantly increased apigenin-induced apoptosis, further demonstrating that the autophagy triggered by apigenin protected macrophages from apigenin-induced cytotoxicity [34]. In contrast, in human papillary thyroid carcinoma BCPAP cells, apigenin exposure resulted in autophagic cell death associated with p62 degradation and Beclin-1 accumulation and LC3 protein conversion. Interestingly, co-treatment with 3-MA significantly protected apigenin-induced cytotoxicity, indicating that apigenin-induced autophagy here is more likely to be a tumor suppressor.
Together, the role of autophagy in apigenin induced cytotoxicity depends on cancer cell types. In most reports, the apigenin-triggered autophagy functions to mediate the acquired resistance of cancer cells against cell apoptosis, evidenced as enhanced cell apoptosis induced by apigenin when in co treatment with autophagy inhibitors. Under this circumstance, the autophagy plays cyto-protective roles in apigenin-induced cytotoxicity in cancer cells. In contrast, autophagy acts as an executioner by inducing autophagic cell death in human papillary thyroid carcinoma BCPAP cells.
APIGENIN IN CANCER THERAPY
|
Tumor Type |
Cell Lines (concentration) |
Mice (dosages) |
Therapeutic Effects |
Mechanisms |
Citations |
|
Colorectal cancer |
SW480 (40 μM) |
Inhibited proliferation, invasion and migration |
Inhibited Wnt/β-catenin signaling |
[36] |
|
|
HCT116 (25 μM) |
Inhibited proliferation; autophagy; apoptosis |
Suppressed the expression of cyclin B1, Cdc2 and Cdc25c; induced PARP cleavage; induced LC3-II |
[21] |
||
|
DLD1 and SW480 (40 μM) |
20 mg/kg (athymic nude mice, intraperitoneally) |
Inhibited proliferation, invasion and migration |
Attenuated NEDD9; reduced phosphorylations of FAK, Src, and Akt |
[37] |
|
|
SW480, DLD-1, and LS174T (40 μM) |
50 mg/kg (BALB/c-nude mice, orthotopically implanted) |
Inhibited proliferation, invasion and migration |
Up-regulated TAGLN; down-regulated MMP-9 expression; decreasing phosphorylation of Akt |
[38] |
|
|
Breast cancer |
BT-474 (40 μM) |
Inhibited cell proliferation; apoptosis |
Reduced the p-JAK1, p-JAK2 and p-STAT3; up-regulated the levels of cleaved caspase-8, cleaved caspase-3 and the cleavage of PARP |
[20] |
|
|
MDA-MD-231 (40 μM) |
5, 25 mg/kg (BALB/c-nude mice, orthotopically injected) |
Cell cycle arrest |
Suppressed cyclin A, cyclin B, and CDK1; upregulated p21WAF1/CIP1; inhibited HDAC activity; induced histone H3 acetylation |
[39] |
|
|
MDA-MB-231 and T47D (40 μM) |
Inhibited cell proliferation; apoptosis |
Increased levels of caspase3, PARP cleavage and Bax/Bcl-2 ratios |
[27] |
||
|
MDA-MB-468 and 4T1 (30 μM) |
Enhanced the immune responses |
Inhibited IFN-γ-induced PD-L1 expression; inhibited STAT1 |
[40] |
||
|
SKBR3 (40 μM) |
Apoptosis |
Reduced the expression of p-JAK2 and p-STAT3; inhibited VEGF |
[19] |
||
|
MDA-MB-453 (60 μM) |
Inhibited cell proliferation; apoptosis |
Up-regulated caspase-8, caspase-3 and the cleavage of PARP; inactivation of JAK2 and STAT3 |
[29] |
||
|
Lung cancer |
H1299 and H460 (20 μM) |
Inhibited cell proliferation; apoptosis |
Suppressed GLUT1 |
[29] |
|
|
A549 (40 μM) |
Inhibited cell proliferation, migration, invasion |
Decreased the PI3K/Akt signaling pathway |
[41] |
||
|
Prostate cancer |
LNCaP (20 μM) |
Inhibited cell proliferation; apoptosis |
Decreased cyclin D1, D2 and E; upregulated WAF1/p21 |
[42] |
|
|
PC-3 and DU145 (20 μM) |
20, 50 μg/mouse/day (athymic nude mice, oral gavage) |
Cell cycle arret; apoptosis |
Suppression of XIAP, c-IAP1, c-IAP2 and survivin; decreased Bcl-xL and Bcl-2 and increase in Bax protein |
[16] |
|
|
DU145 (20 μM) |
Inhibited migration and invasion; cell cycle arrest |
Increased E-cadherin; decreased snail and vimentin |
[43] |
||
|
20 and 50 μg/mouse/day (TRAMP mice, oral gavage) |
Inhibited tumorigenesis |
Inhibited IKK activation and restored the expression of IκBα |
[44] |
||
|
PC-3 and 22Rv1 (20 μM) |
20 and 50 μg/mouse/day (athymic nude mice, oral gavage) |
Inhibited cell proliferation, invasivion |
Inactivation of IKKα; suppressed NF-?B/p65 activation |
[45] |
|
|
PC3-M and LNCaP C4-2B (25 μM) |
Inhibited cell proliferation and metastases |
Inhibited the Smad2/3 and Src/FAK/Akt pathways |
[46] |
||
|
PC3 (25 μM) |
Apoptosis; cell cycle arrest; suppressed stem cell migration |
Increased p21 and p27; upregulated caspases-8, -3 and TNF-α; downregulation of PI3K/Akt and NF-κB signaling |
[47] |
||
|
Melanoma |
A375, C8161 (40 μM) |
Inhibited proliferation and invasion; apoptosis; cell cycle arrest |
Activation of cleaved caspase-3 and cleaved PARP; decreased ERK1/2 proteins, p-AKT and p-Mtor |
[48] |
|
|
A2058, A375 (20 μM) |
Inhibited metastasis |
Inhibited the phosphorylation of FAK/ERK1/2 |
[49] |
||
|
A375, G361 (20 μM) |
150 mg/kg (C57BL/6 mice, oral gavage) |
Inhibited metastasis |
Suppressed STAT3 phosphorylation; down-regulated MMP-2, MMP-9, VEGF and Twist1 |
[50] |
|
|
Leukemia |
HL60 (60 μM) |
Apoptosis |
Activation of caspase-9 and caspase-3 |
[17] |
|
|
HL60 (50 μM); TF1 (30 μM) |
Cell cycle arrest |
Inhibited JAK/STAT pathway |
[51] |
||
|
U937 (40 μM) |
20, 40 mg/kg (athymic nude mice, intraperitoneally) |
Apoptosis |
Inactivation of Akt; activation of JNK; downregulated Mcl-1 and Bcl-2 |
[52] |
|
|
Ovarian cancer |
A2780 (20, 40 μM) |
5 mg/kg (BALB/c nude mice, intraperitoneally) |
Inhibited adhesion, migration and invasion |
Inhibited FAK expression |
[53] |
|
SKOV3 (20, 40 μM) |
Inhibited the self-renewal capacity |
Downregulated Gli1; inhibition of CK2α |
[54] |
||
|
Glioblastoma |
GL-15 (50 μM) |
Inhibited angiogenic |
Reduced TGF-b1 production |
[55] |
|
|
U87MG and U373MG (25 μM) |
Inhibited self-renewal capacity |
Blocked the activation of c-Met signaling |
[56] |
||
|
Renal cell carcinoma |
ACHN, 786-0, and Caki-1 (20 μM) |
30 mg/kg (BALB/c-nude mice intraperitoneally) |
Cell cycle arrest |
p53 accumulation; modulated ATM signalling |
[57] |
|
Adenoid cystic carcinoma |
ACC-2 (40 μM) |
Inhibited proliferation; apoptosis |
Suppressed the expression of GLUT-1 |
[58] |
|
|
Papillary thyroid carcinoma |
BCPAP (25 μM) |
Cell cycle arrest; autophagy |
Down-regulation of Cdc25C expression |
[59] |
|
|
Oral squamous cell carcinoma |
SCC-25, HaCaT (100 μM) |
Inhibited proliferation; apoptosis |
Decreased expression of cyclin D-1 and E; inactivation of CDK1 |
[60] |
|
|
Pancreatic cancer |
Murine Panc02 (20 μM) |
25 mg/kg (female C57BL/6N mice, intraperitoneally) |
Maintain T cell homeostasis |
Stabilizing Ikaros expression |
[61] |
|
Mesothelioma |
Malignant mesothelioma (MM) cells (50 μM) |
20 mg/kg (C57BL/6 mice, oral gavage) |
Apoptosis |
Inhibited AKT and c-Jun phosphorylation, and inhibited NF-κB nuclear translocation |
[62] |
|
Osteosarcoma |
U2OS and MG63 (50 μg/ml) |
Inhibited proliferation and invasion |
Inactivated Wnt/β-catenin signaling |
[63] |
|
|
Head and neck squamous cell carcinoma |
HSC-3, HN-8, and HN- 30 (40 μM) |
Suppressed cancer stem cell marker expression |
Downregulated the stem cell markers of CD44,NANOG, and CD105, and abolished the hypoxia-induced increase |
[64] |
|
|
Cervical cancer |
HeLa (40 μM) |
Inhibited cell self-renewal capacity |
Downregulation of CK2α expression |
[65] |
Table 2: Effects of Apigenin Treatment on Cancer Cells.
CONCLUSION
Recent studies considerably support the notion that a diet rich in chamomile plant flavones is associated with a number of health benefits, including a reduction of the risk of developing certain cancers. Integration of dietary modification rich in chamomile flavones might be a comprehensive chemo preventive strategy for the high-risk individual that may have an impact in the neoplastic transformation. Since apigenin is one of the most bioactive plant flavones and is widely distributed in common fruits, beverages and vegetables, its consumption through diet is highly recommended. Based on the studies provided apigenin affects several critical pathways and /or targets which are associated with several health disorders including cancer. Further research is required before apigenin could be brought to the clinical trials. In addition, apigenin has been demonstrated to help in improving cardiovascular conditions, stimulate the immune system and provide some protection against cancer. Establishing whether or not therapeutic effects of apigenin are beneficial to patients will require research and generation of scientific evidence. However, based on the above highlighted findings apigenin has potential for further investigation and development and development as a cancer chemo preventive and/or therapeutic agent.
REFERENCES
- Ivens GM (1979) Stinking mayweed. NZJ Agric 138: 21-23.
- Svab J (1979) New aspects of cultivating chamomile. Herba Polonica 25: 35-39.
- Handa KL, Chopra IC, Abrol BK (1957) Introduction of some of the important exotic aromatic plants in Jammu and Kashmir. Indian Perfumer 1: 42-49.
- Chandra V (1973) Cultivation of plants for perfumery industry at Lucknow. Indian Perfumer 16: 40-44.
- Chandra V, Singh A, Kapoor LD (1968) Experimental cultivation of some essential oil bearing plants in saline soils, Matricariachamomilla L. Perfum Essent Oil Rec 59: 871.
- Bray F, Ren JS, Masuyer E, Ferlay J (2013) Global estimates of cancer prevalence for 27 sites in the adult population in 2008. Int J Cancer 132: 1133-1145.
- Harirchi I, Kolahdoozan S, Karbakhsh M, Chegini N, Mohseni SM, et al. (2011) Twenty years of breast cancer in Iran: Downstaging without a formal screening program. Ann Oncol 22: 93-97.
- Nikseresht M, Kamali AM, Rahimi HR, Delaviz H, Toori MA, et al. (2017) The Hydroalcoholic Extract of Matricaria Chamomilla suppresses migration and invasion of human breast cancer MDA-MB-468 and MCF-7 cell lines. Pharmacognosy Res 9: 87-95.
- Ogata Ikeda I, Seo H, Kawanai T, Hashimoto E, Oyama Y (2011) Cytotoxic action of bisabololoxide A of German chamomile on human leukemia K562 cells in combination with 5-fluorouracil. Phytomedicine 18: 362-365.
- Park EH, Bae WY, Eom SJ, Kim KT, Paik HD (2017) Improved antioxidative and cytotoxic activities of chamomile (Matricaria Chamomilla) florets fermented by Lactobacillus plantarum KCCM 11613P*. J Zhejiang Univ Sci B 18: 152-160.
- Ali F, Rahul, Naz F, Jyoti S, Siddique YH (2017) Health functionality of apigenin: A review. International Journal of Food Properties 20: 1197-1238.
- Shukla S, Gupta S (2010) Apigenin: A promising molecule for cancer prevention. Pharm Res 27: 962-978.
- Budhraja A, Gao N, Zhang Z, Son YO, Cheng S et al. (2012) Apigenin induces apoptosis in human leukemia cells and exhibits anti-leukemic activity in vivo. Molecular Cancer Therapeutics 11: 132-142.
- Elmore S (2007) Apoptosis: A review of programmed cell death. Toxicol Pathol 35: 495-516.
- Vela L, Marzo I (2015) Bcl-2 family of proteins as drug targets for cancer chemotherapy: The long way of BH3 mimetics from bench to bedside. Curr Opin Pharmacol 23: 74-81.
- Shukla S, Fu P, Gupta S (2014) Apigenin induces apoptosis by targeting inhibitor of apoptosis proteins and Ku70-Bax interaction in prostate cancer. Apoptosis 19: 883-894.
- Wang IK, Lin Shiau SY, Lin JK (1999) Induction of apoptosis by apigenin and related flavonoids through cytochrome c release and activation of caspase-9 and caspase-3 in leukaemia HL-60 cells. Eur J Cancer 35: 1517-1525.
- Shao H, Jing K, Mahmoud E, Huang H, Fang X, et al. (2013) Apigenin sensitizes colon cancer cells to antitumor activity of ABT-263. Mol Cancer Ther 12: 2640-2650.
- Seo HS, Ku JM, Choi HS, Woo JK, Jang BH, et al. (2015) Apigenin induces caspase-dependent apoptosis by inhibiting signal transducer and activator of transcription 3 signaling in HER2-overexpressing SKBR3 breast cancer cells. Mol Med Rep 12: 2977-2984.
- Seo HS, Jo JK, Ku JM, Choi HS, Choi YK, et al. (2015) Induction of caspase-dependent extrinsic apoptosis by apigenin through inhibition of signal transducer and activator of transcription 3 (STAT3) signalling in HER2-overexpressing BT-474 breast cancer cells. Biosci Rep 35: 00276.
- Chen M, Wang X, Zha D, Cai F, Zhang W, et al. (2016) Apigenin potentiates TRAIL therapy of non-small cell lung cancer via upregulating DR4/DR5 expression in a p53-dependent manner. Sci Rep 6: 35468.
- Abu Yousif AO, Smith KA, Getsios S, Green KJ, Van Dross RT, et al. (2008) Enhancement of UVB-induced apoptosis by apigenin in human keratinocytes and organotypic keratinocyte cultures. Cancer Res 68: 3057-3065.
- Korolchuk VI, Rubinsztein DC (2011) Regulation of autophagy by lysosomal positioning. Autophagy 7: 927-928.
- Chaabane W, User SD, El Gazzah M, Jaksik R, Sajjadi E, et al. (2013) Autophagy, apoptosis, mitoptosis and necrosis: Interdependence between those pathways and effects on cancer. Arch Immunol Ther Exp (Warsz) 61: 43-58.
- Ruela-de-Sousa RR, Fuhler GM, Blom N, Ferreira CV, Aoyama H, et al. (2010) Cytotoxicity of apigenin on leukemia cell lines: Implications for prevention and therapy. Cell Death Dis 1: 19.
- Salmani JMM, Zhang XP, Jacob JA, Chen BA (2017) Apigenin’s anticancer properties and molecular mechanisms of action: Recent advances and future prospectives. Chin J Nat Med 15: 321-329.
- Tong X, Smith KA, Pelling JC (2012) Apigenin, a chemo preventive bioflavonoid, induces AMP-activated protein kinase activation in human keratinocytes. MolCarcinog 51: 268-279.
- Sung B, Chung HY, Kim ND (2016) Role of apigenin in cancer prevention via the induction of apoptosis and autophagy. J Cancer Prev 21: 216-226.
- Lee YM, Lee G, Oh TI, Kim BM, Shim DW, et al. (2016) Inhibition of glutamine utilization sensitizes lung cancer cells to apigenin-induced apoptosis resulting from metabolic and oxidative stress. Int J Oncol 48: 399-408.
- Cao X, Liu B, Cao W, Zhang W, Zhang F, et al. (2013) Autophagy inhibition enhances apigenin-induced apoptosis in human breast cancer cells. Chin J Cancer Res 25: 212-222.
- Jung YY, Lee YK, Koo JS (2016) The potential of Beclin 1 as a therapeutic target for the treatment of breast cancer. Expert OpinTher Targets 20: 167-178.
- Wirawan E, Lippens S, Vanden Berghe T, Romagnoli A, Fimia GM, et al. (2012) Beclin1: A role in membrane dynamics and beyond. Autophagy 8: 6-17.
- Gaballah HH, Gaber RA, Mohamed DA (2017) Apigenin potentiates the antitumor activity of 5-FU on solid Ehrlich carcinoma: Crosstalk between apoptotic and JNK-mediated autophagic cell death platforms. Toxicol Appl Pharmacol 316: 27-35.
- Wang Q, Zeng P, Liu Y, Wen G, Fu X, et al. (2015) Inhibition of autophagy ameliorates atherogenic inflammation by augmenting apigenin-induced macrophage apoptosis. Int Immunopharmacol 27: 24-31.
- Yan X, Qi M, Li P, Zhan Y, Shao H (2017) Apigenin in cancer therapy: Anti-cancer effects and mechanisms of action. Cell & Bioscience 7: 50.
- Xu M, Wang S, Song YU, Yao J, Huang K, et al. (2016) Apigenin suppresses colorectal cancer cell proliferation, migration and invasion via inhibition of the Wnt/β-catenin signaling pathway. Oncol Lett 11: 3075-3080.
- Dai J, Van Wie PG, Fai LY, Kim D, Wang L, et al. (2016) Downregulation of NEDD9 by apigenin suppresses migration, invasion, and metastasis of colorectal cancer cells. Toxicol Appl Pharmacol 311: 106-112.
- Chunhua L, Donglan L, Xiuqiong F, Lihua Z, Qin F, et al. (2013) Apigenin up-regulates transgelin and inhibits invasion and migration of colorectal cancer through decreased phosphorylation of AKT. J Nutr Biochem 24: 1766-1775.
- Tseng TH, Chien MH, Lin WL, Wen YC, Chow JM, et al. (2017) Inhibition of MDA-MB-231 breast cancer cell proliferation and tumor growth by apigenin through induction of G2/M arrest and histone H3 acetylation-mediated p21WAF1/CIP1 expression. Environ Toxicol 32: 434-444.
- Coombs MR, Harrison ME, Hoskin DW (2016) Apigenin inhibits the inducible expression of programmed death ligand 1 by human and mouse mammary carcinoma cells. Cancer Lett 380: 424-433.
- Seo HS, Ku JM, Choi HS, Woo JK, Jang BH, et al. (2014) Induction of caspase-dependent apoptosis by apigenin by inhibiting STAT3 signaling in HER2-overexpressing MDA-MB-453 breast cancer cells. Anticancer Res 34: 2869-2882.
- Zhou Z, Tang M, Liu Y, Zhang Z, Lu R, et al. (2017) Apigenin inhibits cell proliferation, migration, and invasion by targeting Akt in the A549 human lung cancer cell line. Anticancer Drugs 28: 446-456.
- Gupta S, Afaq F, Mukhtar H (2002) Involvement of nuclear factor-kappa B, Bax and Bcl-2 in induction of cell cycle arrest and apoptosis by apigenin in human prostate carcinoma cells. Oncogene 21: 3727-3738.
- Zhu Y, Wu J, Li S, Wang X, Liang Z, et al. (2015) Apigenin inhibits migration and invasion via modulation of epithelial mesenchymal transition in prostate cancer. Mol Med Rep 11: 1004-1008.
- Shukla S, Shankar E, Fu P, MacLennan GT, Gupta S (2015) Suppression of NF-κB and NF-κB-Regulated gene expression by apigenin through IκBα and IKK pathway in TRAMP mice. PLoS One 10: 0138710.
- Shukla S, Kanwal R, Shankar E, Datt M, Chance MR, et al. (2015) Apigenin blocks IKKα activation and suppresses prostate cancer progression. Oncotarget 6: 31216-31232.
- Mirzoeva S, Franzen CA, Pelling JC (2014) Apigenin inhibits TGF-β-induced VEGF expression in human prostate carcinoma cells via a Smad2/3-and Src-dependent mechanism. Mol Carcinog 53: 598-609.
- Erdogan S, Doganlar O, Doganlar ZB, Serttas R, Turkekul K, et al. (2016) The flavonoid apigenin reduces prostate cancer CD44(+) stem cell survival and migration through PI3K/Akt/NF-kappaB signaling. Life Sci 162: 77-86.
- Zhao G, Han X, Cheng W, Ni J, Zhang Y, et al. (2017) Apigenin inhibits proliferation and invasion, and induces apoptosis and cell cycle arrest in human melanoma cells. Oncol Rep 37: 2277-2285.
- Hasnat MA, Pervin M, Lim JH, Lim BO (2015) Apigenin attenuates melanoma cell migration by inducing anoikis through integrin and focal adhesion kinase inhibition. Molecules 20: 21157-21166.
- Cao HH, Chu JH, Kwan HY, Su T, Yu H, et al. (2016) Inhibition of the STAT3 signaling pathway contributes to apigenin-mediated anti-metastatic effect in melanoma. Sci Rep 6: 21731.
- Budhraja A, Gao N, Zhang Z, Son YO, Cheng S, et al. (2012) Apigenin induces apoptosis in human leukemia cells and exhibits anti-leukemic activity in vivo. Mol Cancer Ther 11: 132-142.
- Hu XW, Meng D, Fang J (2008) Apigenin inhibited migration and invasion of human ovarian cancer A2780 cells through focal adhesion kinase. Carcinogenesis 29: 2369-2376.
- Tang AQ, Cao XC, Tian L, He L, Liu F (2015) Apigenin inhibits the self-renewal capacity of human ovarian cancer SKOV3-derived sphere-forming cells. Mol Med Rep 11: 2221-2226.
- Freitas S, Costa S, Azevedo C, Carvalho G, Freire S, et al., (2011) Flavonoids inhibit angiogenic cytokine production by human glioma cells. Phytother Res 25: 916-921.
- Kim B, Jung N, Lee S, Sohng JK, Jung HJ (2016) Apigenin inhibits cancer stem cell-like phenotypes in human glioblastoma cells via suppression of c-Met signaling. Phytother Res 30: 1833-1840.
- Meng S, Zhu Y, Li JF, Wang X, Liang Z, et al. (2017) Apigenin inhibits renal cell carcinoma cell proliferation. Oncotarget 8: 19834-19842.
- Fang J, Bao YY, Zhou SH, Fan J (2015) Apigenin inhibits the proliferation of adenoid cystic carcinoma via suppression of glucose transporter-1. Mol Med Rep 12: 6461-6466.
- Zhang L, Cheng X, Gao Y, Zheng J, Xu Q, et al. (2015) Apigenin induces autophagic cell death in human papillary thyroid carcinoma BCPAP cells. Food Funct 6: 3464-3472.
- Maggioni D, Garavello W, Rigolio R, Pignataro L, Gaini R, et al. (2013) Apigenin impairs oral squamous cell carcinoma growth in vitro inducing cell cycle arrest and apoptosis. Int J Oncol 43: 1675-1682.
- Nelson N, Szekeres K, Iclozan C, Rivera IO, McGill A, et al. (2017) Apigenin: Selective CK2 inhibitor increases Ikaros expression and improves T cell homeostasis and function in murine pancreatic cancer. PLoS One 12: 0170197.
- Masuelli L, Benvenuto M, Mattera R, Di Stefano E, Zago E, et al. (2017) in vitro and in vivo anti-tumoral effects of the flavonoid apigenin in malignant mesothelioma. Front Pharmacol 8: 373.
- Liu X, Li L, Lv L, Chen D, Shen L, et al. (2015) Apigenin inhibits the proliferation and invasion of osteosarcoma cells by suppressing the Wnt/β-catenin signaling pathway. Oncol Rep 34: 1035-1041.
- Ketkaew Y, Osathanon T, Pavasant P, Sooampon S (2017) Apigenin inhibited hypoxia induced stem cell marker expression in a head and neck squamous cell carcinoma cell line. Arch Oral Biol 74: 69-74.
- Liu J, Cao XC, Xiao Q, Quan MF (2015) Apigenin inhibits HeLa sphere-forming cells through inactivation of casein kinase 2α. Mol Med Rep 11: 665-669.
Citation: Thalluri GSK, Srinu P (2018) Role of Chamomile in Cancer Treatment. J Pathol Clin Med Res 1: 001.
Copyright: © 2018 Thalluri Gouri Sai Kiran, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.