
Journal of Orthopedic Research & Physiotherapy Category: Medical
Type: Research Article
The Effect of Ultra-High Frequency Electromagnetic Radiation on Reparative Osteogenesis and Angiogenesis under Transosseous Osteosynthesis
*Corresponding Author(s):
Iryanov Yu MRussian Ilizarov Scientific Center Restorative Traumatology And Orthopaedics, Kurgan, Russian Federation
Tel:+7 3522466316,
Email:irianov@mail.ru
Received Date: May 07, 2015
Accepted Date: Jul 18, 2015
Published Date: Aug 03, 2015
Abstract
Background: The use of non-medicinal facilities of correcting processes of bone tissue regeneration for various pathological conditions is one of the most urgent problems of modern medicine.
Purpose of the study: To study the efficiency of using low-intensive ultra-high frequency electromagnetic radiation in order to promote reparative osteogenesis and angiogenesis during fracture treatment under transosseous osteosynthesis making a qualitative and quantitative morphological analysis.
Materials and methods: Tibial fracture was modeled in the experiment on rats in control and experimental groups with reposition and fixation of the bone fragments. In experimental animals the fracture zone was exposed to low intensity electromagnetic radiation of ultra-high frequency. Exposure simulation was performed in the control group. The operated bones were examined using radiography, light and electronic microscopy, X-ray electron probe microanalysis.
Results: Electromagnetic radiation of ultra-high frequency in fracture treatment was proved to stimulate secretory activity and degranulation of mast cells, to produce the increase in microcirculatory bed vascular permeability, endotheliocyte migration phenotype expression, to ensure endovascular endothelial outgrowth, to activate reparative osteogenesis and angiogenesis while fracture reparation becomes one of the primary type and of a short-term duration. The content of bone tissue in the zone of fracture has been demonstrated to exceed the control values by 74%, 89% and 75%, respectively, 7, 14 and 28 days after surgery when exposed to low-intensive electromagnetic radiation of ultra-high frequency.
Conclusion: Mast cell secretory activity stimulation and endovascular angiogenesis activation is one of the therapeutic action mechanisms of ultra-high frequency electromagnetic radiation during fracture treatment.
Purpose of the study: To study the efficiency of using low-intensive ultra-high frequency electromagnetic radiation in order to promote reparative osteogenesis and angiogenesis during fracture treatment under transosseous osteosynthesis making a qualitative and quantitative morphological analysis.
Materials and methods: Tibial fracture was modeled in the experiment on rats in control and experimental groups with reposition and fixation of the bone fragments. In experimental animals the fracture zone was exposed to low intensity electromagnetic radiation of ultra-high frequency. Exposure simulation was performed in the control group. The operated bones were examined using radiography, light and electronic microscopy, X-ray electron probe microanalysis.
Results: Electromagnetic radiation of ultra-high frequency in fracture treatment was proved to stimulate secretory activity and degranulation of mast cells, to produce the increase in microcirculatory bed vascular permeability, endotheliocyte migration phenotype expression, to ensure endovascular endothelial outgrowth, to activate reparative osteogenesis and angiogenesis while fracture reparation becomes one of the primary type and of a short-term duration. The content of bone tissue in the zone of fracture has been demonstrated to exceed the control values by 74%, 89% and 75%, respectively, 7, 14 and 28 days after surgery when exposed to low-intensive electromagnetic radiation of ultra-high frequency.
Conclusion: Mast cell secretory activity stimulation and endovascular angiogenesis activation is one of the therapeutic action mechanisms of ultra-high frequency electromagnetic radiation during fracture treatment.
Keywords
Electromagnetic radiation of ultra-high frequency; Endovascular angiogenesis; Mast cells; Reparative osteogenesis; Transosseous osteosynthesis
INTRODUCTION
Advances in the field of transosseous osteosynthesis are well known [1-3]. The method is widely used for treatment of patients with limb bone defects and shortenings. The urgency of this problem is increasing due to the increase of injuries and congenital pathologies. However, this method is still rather a laborious and lengthy process, as well as it involves multistage therapeutic measures. Modern social-and-economic conditions dictate the need of introducing novel technologies in practice which allow creating more optimal conditions for reparative osteogenesis and angiogenesis. The use of non-medicinal facilities of correcting processes for various pathological conditions is one of the most urgent problems of modern medicine. The impact of Electromagnetic Radiation of Ultra-High Frequencies (UHF EMR) and low intensity (not causing the object heating) is increasingly used in clinical practice [4-7], including treating patients in traumatology and orthopaedics [4,8]. This is due to a high therapeutic effect for a wide range of diseases (action pleiotropicity), the method non-invasiveness, the lack of contraindications and allergic reactions [9,10]. Currently there is no definitive understanding of the mechanisms of UHF EMR impact on biological systems despite the marked therapeutic effect [11,12]. The influence of UHF radiation on reparative osteogenesis has been studied in separate works performed using radiologic and clinical methods [8].
PURPOSE OF THE STUDY
To study the efficiency of using low-intensive electromagnetic radiation of ultra-high frequency in order to promote reparative osteogenesis and angiogenesis during fracture treatment under transosseous osteosynthesis with a qualitative and quantitative morphological analysis.
MATERIAL AND METHODS
The experimental work has been performed by the type of a non-randomized study. The same type of the experiment performing in the main (experimental) and the comparison (control) groups was the basic criterion of conformity. All the groups of animals were kept under the same standard conditions and feeding. We performed the experiments using 30 Wistar adult rats, males and females, of 340-390g body weight in the control group and experimental one (15 animals each). We studied the tibias of five intact rats as well. Total duration of the study was 28 days. Surgical interventions were performed according to the guidelines of the European Convention for the protection of vertebrate animals. The ethic examination was performed by the Committee on Biomedical Ethics at the Izhevsk State Medical Academy of the Russian Federation (approval form No 378 of 25.02.2014).
DETAILED MEDICAL INTERVENTION
A closed transverse non-displaced tibial fracture was modeled manually in the shaft middle third under general anesthesia, reposition and fixation of fragments performed using the device for small bone osteosynthesis developed by us [13] and special wires with slim ends [14]. The fragments were fixed to the support made of fast-hardening paste [15]. One day after surgery the animals of the experimental group underwent the impact of UHF-range EMR of low intensity using UHF-therapy device “Stella 2” (Russia). We used 40-43 gigaHz radiation frequency (wave length - 7.50-6.98 mm). The exposure applied in the pulsed mode locally to the zone of fracture for ten minutes. The distance between the radiator and the skin was one mm. UHF pulsed power at the radiator output was 10 µW/cm2, pulse frequency generation -8.6Hz, pulse duration - 1-3 µsec. The exposure sessions repeated every other day. The exposure imitation performed in the control group of animals when the device of UHF-therapy was off.
METHODS OF THE STUDY
We used five animals for each time point. X-rays made right after surgery and in the process of treatment. The animals euthanized 7, 14 and 28 days after surgery. We fixed the operated bones in 2% paraformaldehyde, glutaraldehyde solution, and also in 1% osmium tetroxide solution on phosphate buffer (?? 7.4), dehydrated and embedded in paraffin (after decalcification) and in araldite (without decalcification). Paraffin sections were stained with hematoxylin-eosin, and with picrofuchsin by Van Gieson. We made the morphological analysis and photomicrography of histological preparations using Stemi 2000-C light microscope complete with AxioCam ERc 5s digital camera and Zen blue software (Carl Zeiss MicroImaging GmbH, Germany). Bones embedded in araldite studied using INCA-200 Energy X-ray electron probe microanalyzer (Oxford Instruments Analytical, England) in the characteristic X-ray calcium radiation. Osteogenesis process activity determined by the content of bone tissue structures in the intermediate zone of a regenerated bone. The organ-specificity of newly formed cortical bone layer in the zone of fracture evaluated by the index of compactness (defined as bone tissue/non-mineralized structure content ratio). We determined the content of sodium, magnesium, sulfur, Calcium (Ca) and Phosphorus (P) in the regenerated bone. The ratio of the last two parameters (??/?) which characterized the maturity degree of bone tissue calculated. We sawed the bones of the regenerated bone zone in blocks, prepared ultra-thin sections of 70-90 nm thickness using LKB-8800 ultramicrotome (LKB, Sweden), contrasted them with uranyl acetate and lead citrate solutions, and studied using JEM-2010 (Jeol, Japan) transmission electron microscope at the accelerating voltage of 80 kV. After that we treated the blocks with 2% sodium ethyolate solution (to remove araldite from the surface), sprayed them with platinum and palladium alloy (in 1:3 ratio) in IB-6 (Eico, Japan) ion vacuum sprayer and studied using JSM-840 scanning electron microscope (Jeol, Japan) in secondary electrons at the accelerating voltage of 20 kV.
METHODS OF A STATISTICAL ANALYSIS
The results of quantitative investigations were processed by the methods of variation statistics. The data are presented as the Mean (M), representativeness error (m), and the level of difference significance (p). The significance of differences in the compared parameters was calculated using Student t-test. Differences were considered significant for the level of ? < 0.05.
RESULTS
A well-pronounced diastasis was observed by X-rays in the operated bone of the animals from the control group seven days after the fracture occurrence. The ends of bone fragments acquired unclear outlines. Periosteum was thickened due to emerging the cloud-like shadows of low-contrast periosteal stratifications. We observed the formation of bone-osteoid and connective-tissue regenerated bone in the histological preparations of the periosteal zone. Fine-mesh structures of thin bone trabeculae tightly adhered to the cortical layer of the fragments were formed at the distance of 1-2mm from the fracture plane. There were areas of cartilaginous and chondroid tissue, as well as the bundles of collagen fibers oriented in parallel to the fracture plane and growing into the diastasis as ribbon-like structures in the middle part of the regenerated bone periosteal zone. As registered, enchondral osteogenesis took place. The intermediate zone of the regenerated bone was filled with mostly small islets of little-vascularized and scarring little-differentiated granulation tissue, as well as with bundles of collagen fibers. There were no structures of newly formed bone tissue in the intermediate zone. A thin layer of the trabeculae of rough-fibered bone tissue was formed along the endosteal surface of the fragments; these trabeculae represented an endosteal bone-osteoid union which did not enter the intermediate space. There were signs of peritrabecular edema around the trabeculae, as well as small cystic cavities near the cortical plate of the fragments. Fibrin clots containing erythrocytes located in the medullary canal, the foci of organized hematoma infiltrated with little-differentiated cellular elements and blood cells were revealed near the ends of bone fragments.
The regenerated bone locating all over the diameter of the fragmental ends was clearly determined by X-rays in the operated bone of the animals from the experimental group seven (7) days after surgery and three sessions of UHF-therapy.
An active process of bone formation was observed which expressed itself in mass proliferation of periosteal and endosteal cambial osteogenic cells, as well as in formation of significant in volume periosteal and endosteal bone-osteoid areas of overgrowth overlapping the diastasis. Newly formed trabeculae of reticulofibrous bone tissue anastomosed and grew towards each other from the periosteal and endosteal surface. Multiple blood vessels and cells at different stages of osteogenic differentiation located between bone trabeculae. The mast cells of spindle- and sprout-like shape at different stages of degranulation process located along blood vessels (Figures 1a and b) which had a significant number of specific cytoplasmic large granules of homogenous electron-dense and grainy structure, many of which located directly below the cytoplasmic membrane. Herewith, partial disintegration of the granules was observed with their content coming out into the extracellular medium due to exocytosis. The signs of increased permeability of the endothelial layer were registered in the vessels of microcirculatory bed localized near the mast cells as evidenced by a large number of micropinocytic vesicles, the dissociation of endothelial contacts and the emergence of interendothelial pores and slots on the luminal surface. At the same time, capillary buds as endovasal endothelial outgrowths were revealed in the lumen of vessels (Figures 1c and d).
The regenerated bone locating all over the diameter of the fragmental ends was clearly determined by X-rays in the operated bone of the animals from the experimental group seven (7) days after surgery and three sessions of UHF-therapy.
An active process of bone formation was observed which expressed itself in mass proliferation of periosteal and endosteal cambial osteogenic cells, as well as in formation of significant in volume periosteal and endosteal bone-osteoid areas of overgrowth overlapping the diastasis. Newly formed trabeculae of reticulofibrous bone tissue anastomosed and grew towards each other from the periosteal and endosteal surface. Multiple blood vessels and cells at different stages of osteogenic differentiation located between bone trabeculae. The mast cells of spindle- and sprout-like shape at different stages of degranulation process located along blood vessels (Figures 1a and b) which had a significant number of specific cytoplasmic large granules of homogenous electron-dense and grainy structure, many of which located directly below the cytoplasmic membrane. Herewith, partial disintegration of the granules was observed with their content coming out into the extracellular medium due to exocytosis. The signs of increased permeability of the endothelial layer were registered in the vessels of microcirculatory bed localized near the mast cells as evidenced by a large number of micropinocytic vesicles, the dissociation of endothelial contacts and the emergence of interendothelial pores and slots on the luminal surface. At the same time, capillary buds as endovasal endothelial outgrowths were revealed in the lumen of vessels (Figures 1c and d).
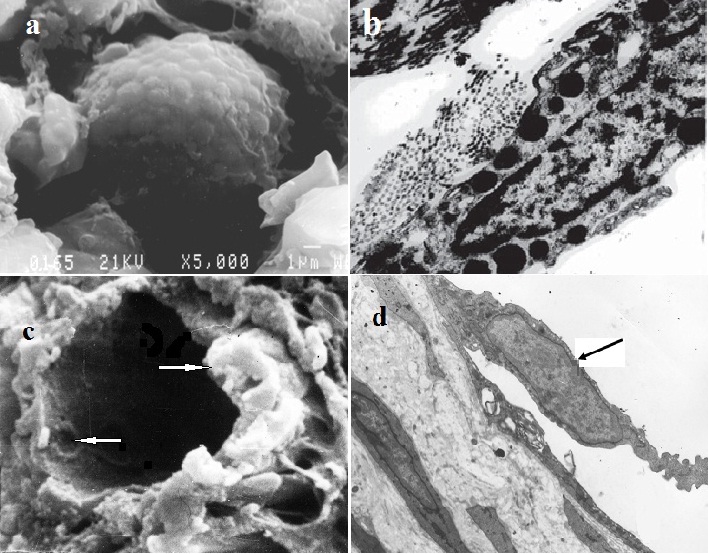
Figure 1: The areas of tibial regenerated bones seven (7) days after the fracture occurrence and performing three (3) sessions of ultra-high frequency electromagnetic radiation impact.
?,b - mast cells at the stage of degranulation: ? - scanning electron microscopy, magnification x 5000; b - an ultra-thin section, transmission electron microscopy, magnification x 8000;
c,d - endovasal endothelial outgrowths (arrows) at different stages of formation: ? - scanning electron microscopy, magnification x 3000; d - an ultra-thin section, transmission electron microscopy, magnification x 5000
The results of electron probe microanalysis (Table 1) evidenced of significant osteogenesis activation and the increase in the maturity degree of newly formed bone tissue in the regenerated bones of the experimental group animals comparing with those of the control one. It is clear from the table that bone tissue content after UHF-therapy was 172.3% and the index of compactness - 200% of the values in the animals from the control group.
| Parameters | Period of experiment, days | |||||
| 7 | 14 | 28 | ||||
| C | Exp | C | Exp | C | Exp | |
| Bone tissue, % | 18.42 ± 0.91 | 31.74 ± 1.531 | 30.45 ± 1.52 | 57.56 ± 2.111 | 45.34 ± 2.33 | 79.37 ± 3.961 |
| Index of compactness | 0.23 ± 0.01 | 0.46 ± 0.021 | 0.44 ± 0.02 | 1.36 ± 0.0721 | 0.83 ± 0.13 | 3.85 ± 0.211 |
Note1 - significant changes comparing with the parameters in animals of the control group. (p = 0.001).
The signs of the initial stage of periosteal union being formed as a result of enchondral osteogenesis appeared in the control group of animals 14 days after surgery (Figure 2a).
Hyaline and fibrous cartilaginous tissue dominated between the fragments, as well as dense fibrous connective tissue. Fracture union occurred by the secondary type with formation of mainly periosteal callus which was presented by a network of interweaving bone trabeculae of different maturity degree formed as a result of enchondral osteogenesis. Primary osteons being formed were determined.
14 days after surgery the bone fragments in the experimental group of animals connected by periosteal-and-endosteal bone structures in the form of vertical brackets. In the intermediate zone the regenerated bone represented by cancellous bone tissue tightly welded together with fragmental ends (Figure 2b). Multiple blood vessels and perivascular mast cells located in intertrabecular spaces, where reorganization of cytoplasmic granules, as well as their contents releasing to the extracellular space was observed. An important feature of mast cells in this period of the experiment consisted in the fact that they had large granules of low electron density and vacuole-like structures. Numerous endovasal endothelial outgrowths at different stages of canalization with characteristic imbricate surface microrelief formed by flat marginal areas of adjacent endotheliocytes were found in the lumens of capillaries and venules adjacent to degranulated mast cells (Figure 2c). Functionally active osteoclasts located on the periosteal and endosteal surface of the fragments, and significant stratifications of newly formed bone trabeculae surrounded by numerous large osteoblasts were observed.
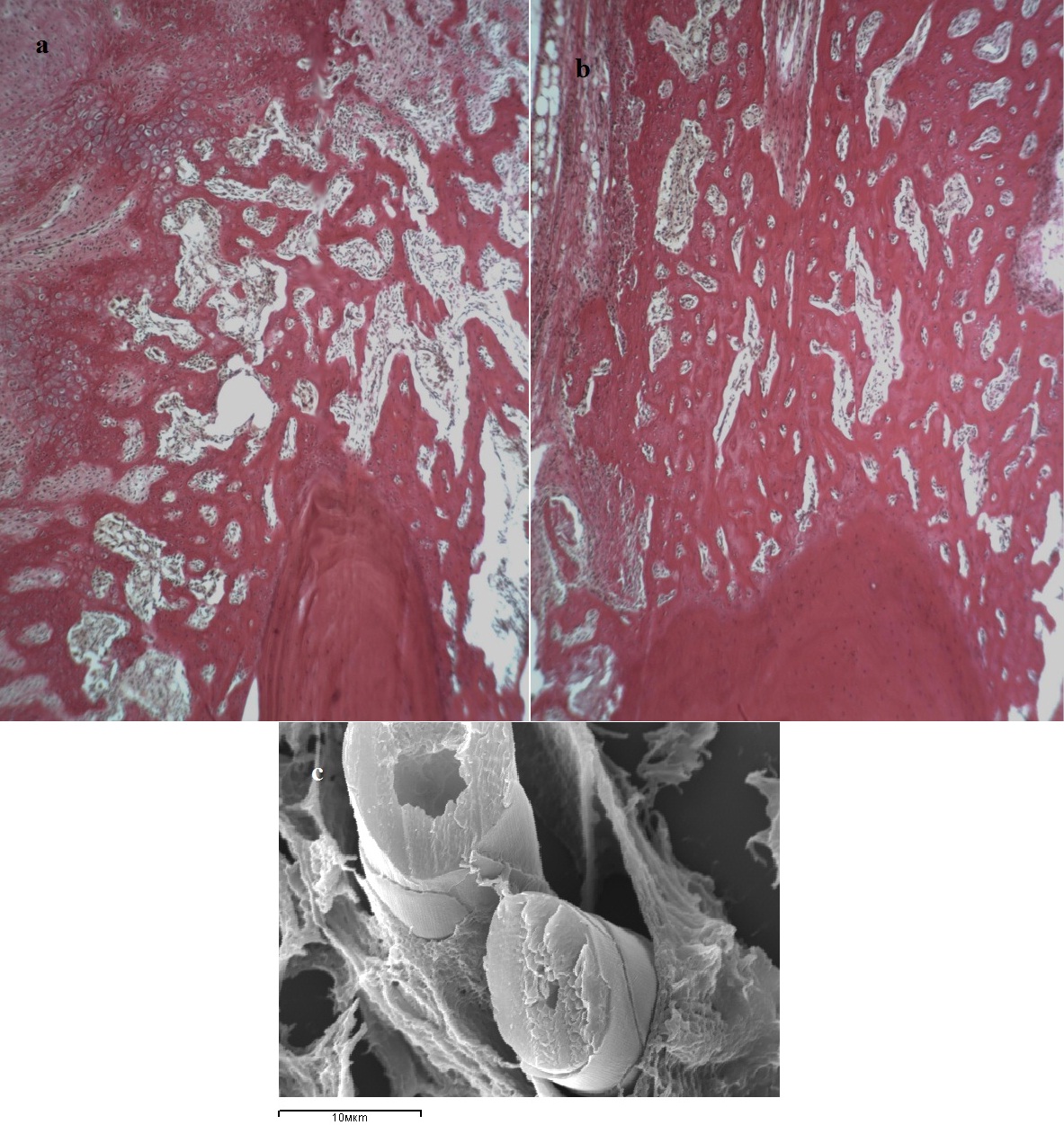
Figure 2: Structure of the regenerated bones in the zone of tibial fracture 14 days after surgery.
As electron probe microanalysis revealed, bone tissue content in the regenerated bones was 189% and the index of compactness - 309% of the values in the animals from the control group 14 days after fracture occurrence and performing six (6) sessions of UHF-therapy (see table 1).
The formation of a new cortical bone layer noted in the both groups of animals 28 days after surgery (Figure 3). The intermediate regenerated bone was clearly defined in the animals from the control group, bone thickening was observed in the zone of injury due to remaining periosteal stratifications of 1.5-2mm which compactized and united the ends of fragments as a fusiform coupler (Figure 3?). The regenerated bone in the intermediate zone is represented by spongy and compact bone tightly adhered to the cortical layer of bone fragments. There were foci of forming primary osteons deprived of ordered structure and orientation.
? - an animal from the control group - enchondral osteogenesis; b - an animal from the experimental group - direct angiogenic (intramembranous) osteogenesis. a, b - paraffin sections, staining by Van Gieson, lens 20, eyepiece 5, magnification x 100. ? - an animal from the experimental group: endovasal endothelial outgrowths (arrows) at different stages of canalization, scanning electron microscopy, magnification x 6000
As electron probe microanalysis revealed, bone tissue content in the regenerated bones was 189% and the index of compactness - 309% of the values in the animals from the control group 14 days after fracture occurrence and performing six (6) sessions of UHF-therapy (see table 1).
The formation of a new cortical bone layer noted in the both groups of animals 28 days after surgery (Figure 3). The intermediate regenerated bone was clearly defined in the animals from the control group, bone thickening was observed in the zone of injury due to remaining periosteal stratifications of 1.5-2mm which compactized and united the ends of fragments as a fusiform coupler (Figure 3?). The regenerated bone in the intermediate zone is represented by spongy and compact bone tightly adhered to the cortical layer of bone fragments. There were foci of forming primary osteons deprived of ordered structure and orientation.

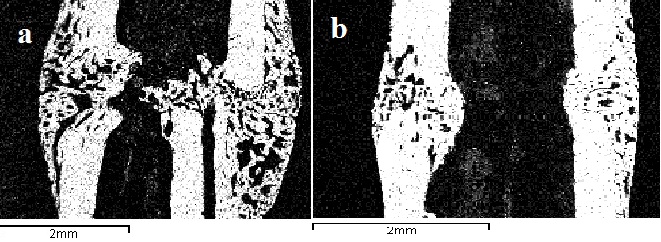
Figure 3: Zone of tibial fracture 28 days after surgery.
a - an animal from the control group; b - an animal from the experimental group; the maps of X-ray electron probe microanalysis, the image in the characteristic X-ray calcium radiation, magnification x 20
In this period of the experiment calcium and phosphorus content in the regenerated bones of the animals from the control group reached 81.04% and 91.27%, respectively, of the level of the values in the shaft cortical layer of intact animals (Table 2).
Table 2: The content of osteotropic chemical elements in the intermediate zone of the regenerated bone in the control and experimental groups of animals 28 days after surgery and in cortical diaphysis of intact animals (M ± m, %).
Full contact of bone fragments in the zone of fracture was observed in experimental animals 28 days after surgery and 12 sessions of UHF-therapy. Complete periosteal, intermediate, and endosteal bone union was revealed throughout the width of the fragments (Figure 3b). Fracture healing occurred by the type of primary one. The ends of the fragments were connected by the secondary osteons of lamellar bone tissue of different maturity degree with compactization scenes. The expanded osteon canals were observed with growing new blood vessels into them. The content of bone tissue in regenerated bones almost 2-fold, and the index of compactness - more than 4-fold exceeded the similar parameters in the control group of animals (Table 1). The newly formed cortical layer did not practically differ in the degree of mineralization from the shaft cortical layer of intact animals. Calcium content in it was 95.17%, and that of phosphorus - 96.03% of the level of the parameters of cortical diaphysis of intact animals (Table 2).
In this period of the experiment calcium and phosphorus content in the regenerated bones of the animals from the control group reached 81.04% and 91.27%, respectively, of the level of the values in the shaft cortical layer of intact animals (Table 2).
| Parameters | Control | Experiment | Cortical diaphysis |
| Sodium | 0.41 ± 0.02 | 0.35 ± 0.02 | 0.30 ± 0.02 |
| Magnesium | 0.32 ± 0.01 | 0.28 ± 0.01 | 0.22 ± 0.01 |
| Phosphorus (?) | 11.51 ± 0.62 | 12.14 ± 0.72 | 12.61 ± 0.72 |
| Sulfur | 0.23 ± 0.01 | 0.30 ± 0.02 | 0.34 ± 0.02 |
| Calcium (??) | 20.82 ± 1.31 | 25.67 ± 1.421 | 26.92 ± 1.42 |
| ??/? | 1.81 ± 0.05 | 2.11 ± 0.06 | 2.13 ± 0.06 |
Note 1 - significant changes comparing with the parameters in animals of the control group (p<0.05).
Full contact of bone fragments in the zone of fracture was observed in experimental animals 28 days after surgery and 12 sessions of UHF-therapy. Complete periosteal, intermediate, and endosteal bone union was revealed throughout the width of the fragments (Figure 3b). Fracture healing occurred by the type of primary one. The ends of the fragments were connected by the secondary osteons of lamellar bone tissue of different maturity degree with compactization scenes. The expanded osteon canals were observed with growing new blood vessels into them. The content of bone tissue in regenerated bones almost 2-fold, and the index of compactness - more than 4-fold exceeded the similar parameters in the control group of animals (Table 1). The newly formed cortical layer did not practically differ in the degree of mineralization from the shaft cortical layer of intact animals. Calcium content in it was 95.17%, and that of phosphorus - 96.03% of the level of the parameters of cortical diaphysis of intact animals (Table 2).
DISCUSSION
UHF EMR power is completely absorbed in skin surface layers, herewith, maximum specific absorption is localized at the depth of 0.7mm [11,12]. Only skin structures such as keratinocytes, fibroblasts, mast cells (mastocytes), cells of microcirculatory bed and peripheral blood, structures of nerve endings, are subjected to direct radiation impact [6-8]. Thus, the revealed by us stimulating effect of UHF EMR on reparative osteogenesis and angiogenesis is not direct but mediated, and it implemented with involvement of the complex systems of autocrine, paracrine, neuroendocrine, and immune regulation. In this case, the primary cell target appears to be mast cells the secretory activity stimulation of which is an important enhancing mechanism in organism’s system response to the impact of low-intensive UHF EMR, that is confirmed by the literature data [12]. Mast cells are the elements of the diffuse neuroendocrine system that are involved in many physiological responses excreting some biologically active substances and mediators into the extracellular medium, such as histamine, heparin, serotonin, various proteases, leukotrienes ?4, D4, ?4, prostaglandin D, tryptase, collagenase [16,17]. Degranulation of mast cells stimulates α2-macroglobulin secretion by macrophages, and α2-macroglobulin being a radioprotector and humoral factor of endogenous anticancer control [12], initiates a response of the microcirculatory bed vessels to UHF EMR, causes prolonged dilatation of capillaries and endothelium-dependent vasodilatation. Herewith, the expression of endotheliocyte migration phenotype is promoted, as well as formation of the endothelial outgrowths in vascular lumens, which form capillary sprouts spreading along the running of “maternal” vessels over long distances meeting no resistance of perivascular tissue structures. This way of vascular development and growth (endovascular angiogenesis) was first discovered by one of the authors of the present work (Iryanov Yu M) during the healing of bone wounds [18,19]. Endovasal angiogenesis is one of regeneration angiogenesis types, and it provides accelerated regeneration and oriented growth of newly formed blood capillaries. Paracrine activity of fibroblasts after the exposure to low intensive UHF EMR also promotes angiogenesis due to the secretion of some Growth Factors of Endothelium of the Vessels (VEGF) activating endothelial progenitor cells, and that of the main Growth Factor of Fibroblasts (bFGF) which accelerates the growth and migration of endotheliocytes [3].
CONCLUSION
As it has been found, the sessions of UHF EMR impact on the zone of the regenerated bone being formed after fracture healing stimulate the secretory activity and degranulation of mast cells, induce increasing the permeability of the microcirculatory bed vessels, expression of endotheliocyte migration phenotype and activation of endovasal angiogenesis. The process of reparative osteogenesis under the impact of UHF EMR occurred much more actively than that in the control group. This manifested itself in the earlier formation of the regenerated bone and bone union, the acceleration of reorganization processes and the compactization of newly formed bone tissue, the increase in its maturity degree, the marked activation of osteoblasts and osteoclasts. The intermediate regenerated bone was formed even seven (7) days after surgery and the sessions of exposure to UHF EMR, union of fragments was observed after 14 days, and complete periosteal, intermediate and endosteal bone union was determined after 28 days. Fracture healing occurred by the type of primary one. The findings reveal the possible mechanism of UHF EMR impact at the level of the whole organism, as well as they prove the efficiency of using UHF-therapy for fracture treatment in clinical practice.
REFERENCES
- Ilizarov GA, Iryanov YM (1991) Osteogenesis under conditions of tensile stress. Bulletin of Experimental Biology and Medicine 111: 194-196.
- Shevtsov VI, Iryanov YM (1995) Osteogenesis and angiogenesis during distraction osteosynthesis. Bulletin of Experimental Biology and Medicine 120: 95-99.
- Iryanov YM, Iryanova TY (2003) Reparative bone formation at the lengthening of limbs under transosseous distraction osteosynthesis. Morphology 3: 83-86.
- Betsky OV, Lebedeva HH, Kotrovsky TI (2005) Application of the use of low-intensity millimeter waves in medicine (retrospective review). Millimeter Waves Biol Med 2: 23-39.
- Alekseev SI, Radzievsky AA, Szabo I, Ziskin MC (2005) Local heating of human skin by millimeter waves: effect of blood flow. Bioelectromagnetics 26: 489-501.
- Lushnikov KV, Shumilina YV, Yakushina VS, Gapev AB, Sadonikov VB, et al. (2004) Effect of low-intensity electromagnetic radiation of extremely high frequency on inflammation. Bull Exp Biol 4: 412-415.
- Belyaev I Ya (2005) Non-thermal biological effects of microwaves. Microwave Review 11: 13-29.
- Kamenev YF (1999) The use of electromagnetic radiation in traumatology and orthopedics. Millimeter waves in biology and medicine 2: 20-25.
- Pletnev SD (2000) The use of millimeter band electromagnetic waves in clinical oncology. Crit Rew Biomed Engineering 29: 573-588.
- Walters TJ, Ryan KL, Nelson DA, Blick DW, Mason PA (2004) Effects of blood flow on skin heating induced by millimeter wave irradiation in humans. Health Phys 86: 115-120.
- Gapeev AB, Sokolov PA, Chemeris NK (2000) Study of the absorption of electromagnetic radiation of extremely high frequency in the skin of rats of dosimetry using different methods and approaches. Biophysics 4: 759-768.
- Popov VI, Rogachevski? VV, Gapeev AB, Khramov RN, Fesenko EE (2001) Degranulation of skin mast cells caused by high frequency electromagnetic irradiation of low intensity. Biofizika 46: 1096-1102.
- Iryanov YM, Naumov EA, Iryanova TY; FGBI “RISC” RTO of the RF Ministry of Health. Device for osteosynthesis of small bones. Russia patent 113651. 2012 February 12.
- Iryanov YM, Dyuryagina OV, Nakoskin AN, Iryanova TY, Naumov EA; FGBI “RISC” RTO Academy of the GA Ilizarov Russian Ministry of Health. Spoke for osteosynthesis. Russian patent 87899. 2009 October 27.
- Iryanov YM, Dyuryagina OV, Nakoskin AN, Iryanova TY; FGBI “RISC” RTO Academy of the GA Ilizarov Russian Ministry of Health. Support device for transosseous osteosynthesis. Russia patent 879000. 2009 October 27.
- Andersson CK, Mori M, Bjermer L, Löfdahl CG, Erjefält JS (2010) Alterations in lung mast cell populations in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med 121: 206-217.
- Bradding P (2009) Human lung mast cell heterogeneity. Thorax 64: 278-280.
- Iryanov YM, Dyuryagina OV (2014) Effect of the local chamber of granulation tissue formed in the medullary cavity on reparative osteogenesis. Bull Exp Biol 1: 121-125.
- Iryanov YM, Kiryanov NA, Popkov AV (2014) Fracture healing in terms of intramedullary injection needles coated with hydroxyapatite. Bulletin of the Russian Academy of Medical Sciences 7-8: 127-132.
Citation: Iryanov YM, Sazonova NV, Kiryanov NA (2015) The Effect of Ultra-High Frequency Electromagnetic Radiation on Reparative Osteogenesis and Angiogenesis under Transosseous Osteosynthesis. J Orthop Res Physiother 1: 010.
Copyright: © 2015 Iryanov Yu M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

Journal Highlights
© 2026, Copyrights Herald Scholarly Open Access. All Rights Reserved!
