
Use of PRP in Severe Bone Defects
*Corresponding Author(s):
Maurizio SerafiniSchool Of Medicine, University Of “Aldo Moro”Bari-Italy,- Via Caduti Sul Lavoro, 37 66100 CHIETI, Abruzzo, Italy
Email:serafinidrmaurizio@gmail.com
Abstract
Aim of the study is to demonstrate the influence of using growth factors in bone regeneration in advanced implantology cases, such as severe bone defects that involve maxillary sinus. The preliminary study of complex cases through CT scan and computerized implant software, it’s crucial for an accurate preoperative evaluation of the anatomic limitations and facilitates preoperative planning of therapeutic strategy. This two case reports present clinical and radiographic results obtained using PRP in combination with bone grafting materials which lead to a faster and predictable bone healing, allowing a time and cost effectiveness implant-prosthetic rehabilitation.
Keywords
Bone Defects; Computerized implant software; Dental implants; Grow factors; Oral rehabilitation; Platelet rich plasma
INTRODUCTION
Tissue regeneration is a crucial therapeutic aspect to achieve a complete and harmonious esthetic and functional oral rehabilitation.
The need to overcome problems such as long recovery time, high costs for both clinician and patient, cross-infection risk, led to developing new techniques based on growth factors huge potential via PRP and PRF, obtained in the office before the surgical procedure.
Bone cells homeostasis is regulated by lots of biomolecules produced by the immune system, by platelets and by cells from the osteogenic lineage. PDFG (Platelet Derived Growth Factor) is mainly involved in the mitotic activity of tissue repairing process and like β-TGF (Transforming Growth Factor-β) is involved in angiogenesis and osteogenesis, and is a chemiotactic booster for fibroblasts and other cells implicated in wound healing [1].
In-vitro and in-vivo studies have proven that the use of platelet growth factors, PRP and PRF, allows a faster healing and mineralization of the graft equal to half of the time and a bone density improvement between 15-30%, speeding mitogenesis and angiogenesis along with macrophages activation [2-4].
The amount of PDGF x million platelets is 0.06 mg, approximately 1200 PDGF molecules x single platelet.
In-vitro studies shown that growth factors like PDGF, are secreted by platelets through a degranulation process which begins within few minutes after the contact with the receiving surface.
Once degranulated, platelets are no more sources of growth factors but the tissue hypoxia between 0.3 and 5 mmHg of the site, compared to the 44-45 mmHg of the surrounding tissue, attracts monocytes and macrophages. The difference of the pressure gradients, increases the concentration of monocytes which have rapid replication and are very efficient in growth factors releasing, explaining how they lead the second phase of bone regeneration, promoting angiogenesis on the 3rd day.
In PRP platelets are not activated with calcium chloride and they are injected in the surgical site before degranulation process, the contact with the rough surface activates them and growth factors are released [5].
PRF is obtained adding calcium chloride, a platelet activator, to PRP, forming autogenous thrombin which can be mixed along with the grafting material and/or placed in the wound to help stimulate cellular proliferation and tissue healing enhancement in various treatments [6].
Long-term clinical and radiological follow-up of dental implants inserted in autologously augmented maxillary bone was proven satisfactory, with stable results after an average of 13 years [7].
MATERIALS AND METHODS
1st case report
49-years-old female patient in good health and good clinical records. Orthopantomography and dental scan (Figure 1), show severe bone loss in the alveolar palatine region of 1.6-1.7 with oro-antral communication (Figure 2).
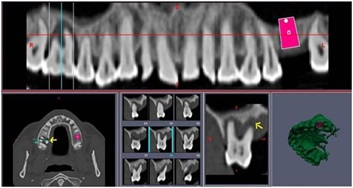 Figure 1: Dental scan
Figure 1: Dental scan
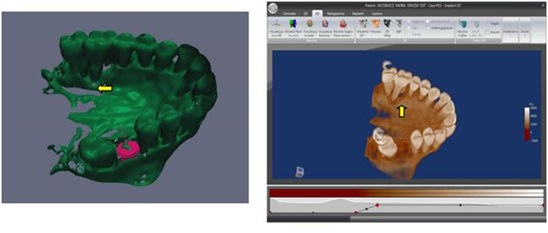 Figure 2: Oro-antral communication in region 1.6-1.7.
Figure 2: Oro-antral communication in region 1.6-1.7.
During surgery, 4 tubes of venous blood are harvested and centrifuged at 1000 rpm for 20 min, obtaining 4 ml of PRP from each tube (Figure 3).
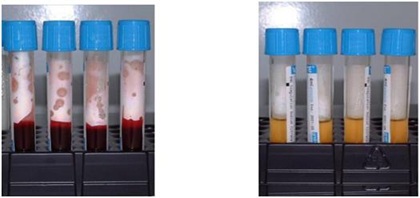 Figure 3: Venous blood sample centrifuged and transformed in P.R.P.
Figure 3: Venous blood sample centrifuged and transformed in P.R.P.
After the extractions, severe bone loss and exposition of maxillary sinus membrane can be observed (Figure 4).
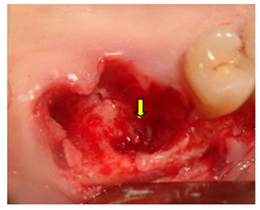 Figure 4: Bone loss and sinus membrane exposition.
Figure 4: Bone loss and sinus membrane exposition.
Two implants in region 1.6-1.7 are placed (Figure 5) after endosinus injection of liquid PRP and imbibed fibrin sponges. The large residual gap is filled with PRP activated with calcium chloride that allows platelet degranulation in order to obtain PRF (platelet-rich-fibrin), mixed with BioOss (Figure 6) and Post Surgery control (Figure 7).
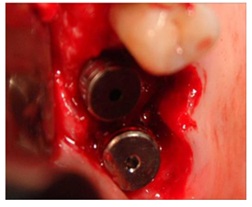 Figure 5: Two implants Placed in region 1.6-1.7.
Figure 5: Two implants Placed in region 1.6-1.7.
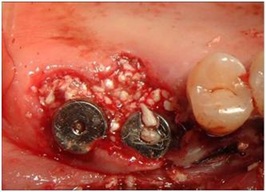 Figure 6: Bio-Oss Mixed with P.R.F.
Figure 6: Bio-Oss Mixed with P.R.F.
 Figure 7: Post Surgery control.
Figure 7: Post Surgery control.
Soft tissue healing is obtained after 15 days (Figure 8a) and after six Months (Figure 8b)
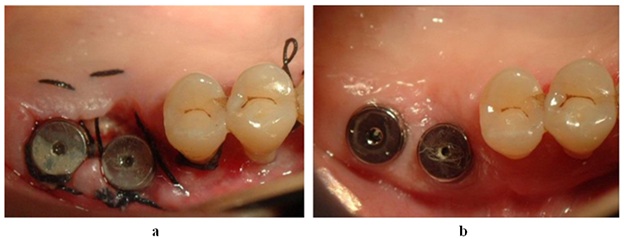 Figure 8: a) Soft tissue healing after 15 days, b) After six months.
Figure 8: a) Soft tissue healing after 15 days, b) After six months.
Tomography after six months shows radiographic integration and increased peri-implant bone density (Figure 9) and check up after two years (Figure 10).
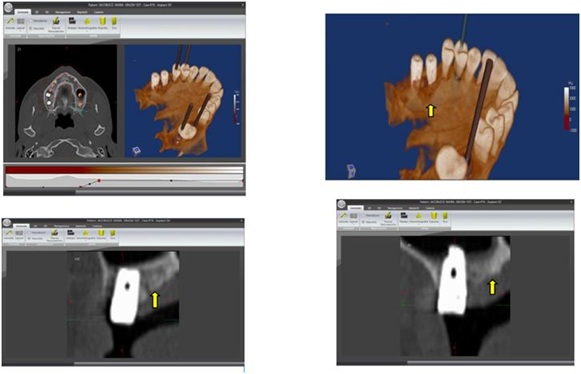 Figure 9: Tomography after six Months.
Figure 9: Tomography after six Months.
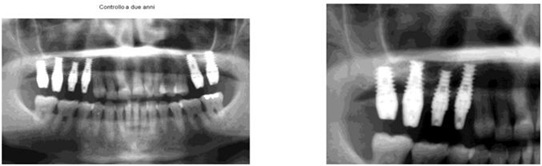 Figure 10: Two years orthopantomography checkup.
Figure 10: Two years orthopantomography checkup.
2nd case report
54-years-old male patient, who had six implants placed two months before, in substitution of a mobile prosthesis; the second phase of surgery aims to rehabilitate the upper left and right maxilla. On the right side severe bone atrophy and a hypertrophy of the sinus membrane is present, other than exposition of the palatine root of 1.6 (Figure 11).
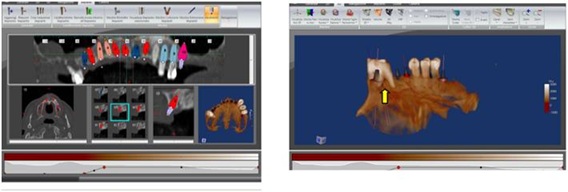 Figure 11: Dental Scan shows severe bone loss.
Figure 11: Dental Scan shows severe bone loss.
After the extraction of 1.6, the alveolus appears covered with gingival mucosa (Figure 12).
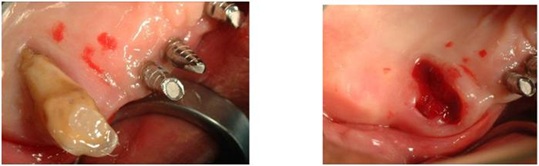 Figure 12: Extraction of 1.6 and its alveolus covered with mucosa.
Figure 12: Extraction of 1.6 and its alveolus covered with mucosa.
Once removed, the prolapsed Schneiderian membrane can be seen from the alveolus (Figure 13).
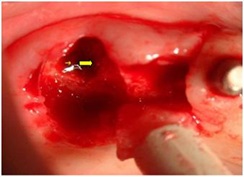 Figure 13: Schneiderian membrane prolapsed.
Figure 13: Schneiderian membrane prolapsed.
Follows the placement of the implants after imbibition of the surgical site with PRP (Figure 14).
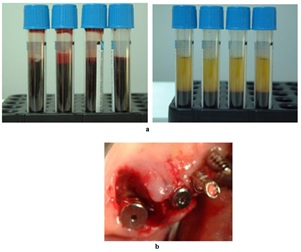 Figure 14: a) 6-7 Blood sample transformed in P.R.P, b) Bone gap around implant.
Figure 14: a) 6-7 Blood sample transformed in P.R.P, b) Bone gap around implant.
The severe palatal gap between implant and palatal mucosa (Figure 14) is filled with PRP activated by calcium chloride that allows platelet degranulation in order to obtain PRF (platelet-rich-fibrin), mixed with Bio-Oss (Figure 15).
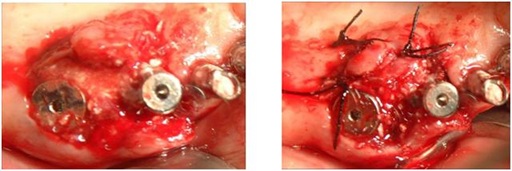 Figure 15: Bio-Oss mixed with P.R.F.
Figure 15: Bio-Oss mixed with P.R.F.
A crestal sinus lift is performed on both sides using growth factors, which are crucial for the healing process and obtain adequate bone and restore the patient normal and complete occlusion (Figure 16).
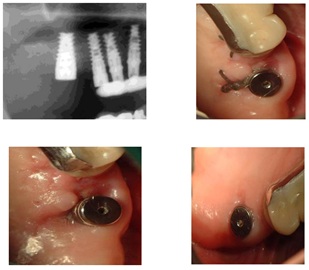 Figure 16: Tissue healing after crestal sinus lift.
Figure 16: Tissue healing after crestal sinus lift.
5 year follow up is shown in Figure 17.
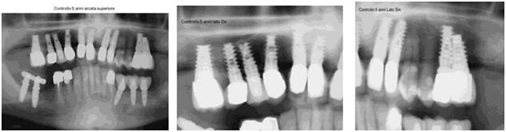 Figure 17: 5 years follow up.
Figure 17: 5 years follow up.
DISCUSSION
Complicated clinical cases usually have long healing period which can be stressful for patients. Shortening this time is important for functional, esthetic and social well-being of the patients. Modern dentistry, thanks to CT scan and its derived programs with the use of modern regeneration techniques using growth factors combined with alloplastic materials or auto-homo-eterologous bone grafting, is able to solve critical anatomical situations. Rehabilitations with the use of PRP is on the increase because of its reduced cost, biocompatibility, autologous nature and ready acceptance by the body providing safe and natural healing.
REFERENCES
- Roberts AB, Sporn MB, Assoian RK, Smith JM, Roche NS, et al. (1986) Transforming growth factor type beta: rapid induction of fibrosis and angiogenesis in vivo and stimulation of collagen formation in vitro. Proc Natl Acad Sci USA 83 : 4167-4171.
- Marx RE, Carlson ER, Eichstaedt RM, Schimmele SR, Strauss JE, et al. (1998) Platelet Rich plasma: growth factor enhancement for bone grafts. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 85: 638-641.
- Kim J, Ha Y, Kang NH (2017) Effects of Growth Factors From Platelet-Rich Fibrin on the Bone Regeneration. J Craniofac Surg 28: 860-865.
- Shah R, Triveni MG, Thomas R, Mehta DS (2017) An Update on the Protocols and Biologic Actions of Platelet Rich Fibrin in Dentistry. Eur J Prosthodont Restor Dent 25: 64-72.
- Marx RE (2004) Platelet-Rich Plasma: Evidence to support its use. J Oral Maxillofac Surg 62: 489-496.
- Marx RE, Carg AK (2005) Dental and craniofacial applications of platelet-rich plasma. Quintessence books (Pg no: 168), Chicago, USA.
- Attia S, Narberhaus C, Schaaf H, Streckbein P, Pons-Kühnemann J, et al. (2020) Long-Term Influence of Platelet-Rich Plasma (PRP) on Dental Implants after Maxillary Augmentation: Retrospective Clinical and Radiological Outcomes of a Randomized controlled Clinical Trial. J Clin Med 9: 355.
Citation: Inchingolo F, Contaldo M, Domenico MD, Serpico R, Palma GD, et al. (2020) Use of PRP in Severe Bone Defects. J Stem Cell Res Dev Ther 6: 052.
Copyright: © 2020 Francesco Inchingolo, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

