
Rensheng ShouWu Protects Against Neuronal Injury by Inhibiting the Apoptosis Induced by Hypoxia and Reoxygenation
*Corresponding Author(s):
Yongming CaiDepartment Of Medical Information Engineering, Guangdong Pharmaceutical University, Guangzhou, China
Tel:+86 2039352607,
Fax:+86 2039352606
Email:cym@gdpu.edu.cn
Weijian Bei
Guangdong Province Research Centre For Chinese Integrative Medicine Against Metabolic Disease, Guangdong TCM Key Laboratory Against Metabolic Diseases, The Institute Of Chinese Medicinal Sciences, Guangdong Pharmaceutical University, Guangzhou, China
Tel:+86 2039352607,
Fax:+86 2039352606
Email:13660020495@139.com
Abstract
Rensheng Shouwu Capsule (RSSW) is an approved and patented drug of Traditional Chinese Medicine that has been used for years to treat vascular dementia and neural syndrome related to cerebral-vascular ischemia. However, the underlying mechanism of RSSW remains unclear.
Previous studies have indicated that RSSW could significantly reduce cerebral ischemic injury and improve vascular dementia. In the present study, cultured cortical neuron hypoxia/reoxygenation injury model was used to test the neuroprotection of RSSW and its mechanisms in vitro.
RSSW 0.2~5.0 μg/ml was demonstrated to increase intracellular endogenous superoxide dismutase, restore the level of mitochondrial membrane potential in hypoxia/anoxia-injured neurons in a dose-dependent manner. RSSW was also shown to inhibit neuronal apoptosis dose-dependently by suppressing the activity of caspase 3 and caspase 9 and by decreasing the level of reactive oxygen species, the leak of lactate dehydrogenase and the accumulation of malondialdehyde, in hypoxia/anoxia-injured neurons. These findings suggest that the neuroprotection of RSSW from hypoxia injuries may be related to the improvement of intracellular endogenous antioxidants, and the inhibition of the caspase 3 and caspase 9 expression, which might represent the mechanisms underlying RSSW prevention and treatment of neurodegenerative disorders and neural disorders related to cerebral-vascular ischemia.
Keywords
INTRODUCTION
Stroke and neurodegenerative disease account for the majority of public health problems in middle-aged and elderly people. These health problems have become a heavier burden over the years to families and society, as the average lifespan has continued to increase. However, combating stroke and neurodegenerative disease has proven to be an enormous challenge [1-3].
Oxidative stress has been implicated in the progression of stroke and neurodegenerative diseases, such as Alzheimer’s Disease (AD), Parkinson’s Disease (PD) and Amyotrophic Lateral Sclerosis (ALS) [4-6]. Oxidative stress is the result of an imbalance in pro-oxidant/antioxidant homeostasis that leads to the generation of toxic Reactive Oxygen Species (ROS). ROS react preferentially with certain atoms to modulate functions ranging from cell homeostasis to cell death [5].
Significant amounts of oxygen free radicals (oxidants) are generated during cerebral ischemia/reperfusion, and oxidative stress plays an important role in brain ischemic neuronal apoptosis and brain damage after stroke. Apoptosis is the main mechanism of cerebral ischemic neuronal damage. In addition to oxidizing macromolecules, leading to cell injury, oxidants are also involved in cell death/survival signal pathways and cause mitochondrial dysfunction [4].
Early signs of cell apoptosis include mitochondrial dysfunction, increased membrane permeability, and decreased membrane potential. Intracellular ROS increase dramatically and induce changes to mitochondrial membrane potential, damage to mitochondrial DNA, changes to the electron transfer chain, and disruption of mitochondrial function, eventually leading to neuronal degeneration and death [6].
Increasing endo-antioxidant function, restoring mitochondrial dysfunction, and reducing the apoptosis induced by oxidative stress are promising strategies to treat the cerebrovascular and neural disorders related to ischemic brain damage, such as ischemic stroke, vascular dementia, and neurodegenerative diseases, such as AD, PD, and others [4-10].
Chinese medicine plays an important role in the prevention and treatment of cerebral ischemia-reperfusion injury and neurodegenerative diseases, such as AD and PD and others [10-12]. Renshen Shouwu Capsule (RSSW) is a patented and Chinese Pharmacopoeia approved Traditional Chinese Medicine (TCM). It is widely used to treat several diseases, including neurodegenerative diseases, amnesia, insomnia, and mental decline [13]. RSSW is composed of Ginseng (Root of Ginseng-Panax ginseng C.A. Mey) and fleece-flower root (Radix Polygoni Multiflori-Polygonum multiflorum Thunb). RSSW had significant neuroprotective effects against Middle Cerebral Artery Occlusion (MCAO) and vessel occlusion caused by ischemic reperfusion (4-VO I/R) injury and a therapeutic effect on cognitive disorders in Vascular Dementia (VD) rats [14]. Our previous studies have shown that RSSW administration significantly reduced the size of the lesion of the insulted brain hemisphere and improved the neurological behavior of MCAO rats. In addition, RSSW markedly reduced an increase in brain infarct volume from an I/R-induced MCAO and reduced the cerebral water content in a dose-dependent manner. Administration of RSSW also increased pyramidal neuronal density in the hippocampus of surviving rats after transient global brain ischemia and improved the learning and memory ability of rats with 4-VO induced vascular dementia in a dose-dependent manner [14]. Although RSSW has significant neuroprotective effects against MCAO and 4-VO I/R injury and a therapeutic effect on cognitive disorders in VD rats, the mechanism underlying its neuroprotective effects remains to be elucidated. Therefore, we aimed to investigate the neuroprotective properties of RSSW against oxidative stress neuronal damage induced by hypoxia/reoxygen in primary rat cortical neurons. Edaravone is a free radical scavenger of clinical use for ischemic stroke and other neurodegenerative disorders [15]. We used edaravone as the positive agent to explore the mechanism of RSSW neuroprotection.
MATERIALS AND METHODS
Preparation of a standardized extract from Renshen Shouwu
Primary cultures of rat cortical neuronal cells
Hypoxia/reoxygenation model and cell culture treatment
Cell viability assay
Neuronal injury assays
The protective effects of RSSW on cortical neurons induced by H/R were measured by determining the amount of apoptotic cells as described by Li and Chen [4,18].
Hoechst 33258 staining for morphological analysis of apoptosis
Apoptosis assays
Measurement of SOD and MDA in neuron cultures
Determination of ROS level
Mitochondrial membrane potential (Δψm) detection
Determination of Caspase-3, 9 activity in neurons by ELISA assays
Statistical analysis
RESULTS
RSSW effect on H/R induced neuronal apoptosis
Hoechst 33258 staining and Annexin V/FITC apoptosis detection showed that 20% of neurons exposed to hypoxia/reoxigeneration suffered from apoptosis, indicating nuclear chromatin agglutination, condensation nuclear chromatin, or broken chromatin fragment with dense fluorescence. Pre-treatment with RSSW was observed to increase the cell viability and reduce apoptosis rate of neurons exposed to hypoxia/reoxygenation in a concentration-dependent manner. Edaravone also showed a similar effect to RSSW (Figure 1a, 1b and 1c). This indicated that RSSW had the ability to reduce the neuron damage and prevent neuron apoptosis induced by hypoxia/reoxygenation.
 Figure 1: Effect of RSSW on the apoptosis induced by H/R in the cortical neuron.
Figure 1: Effect of RSSW on the apoptosis induced by H/R in the cortical neuron. ##P
RSSW effect on neuron LDH leakage
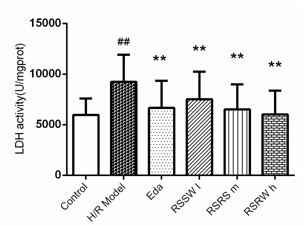 Figure 2: The Effect of RSSW on LDH leakage of the cortical neuron induced by H/R Neurons was treated as the text described. The data were expressed as the mean ±SD (Zhu et al.).
Figure 2: The Effect of RSSW on LDH leakage of the cortical neuron induced by H/R Neurons was treated as the text described. The data were expressed as the mean ±SD (Zhu et al.).The effect of RSSW on SOD, MDA level in neurons exposed to H/R
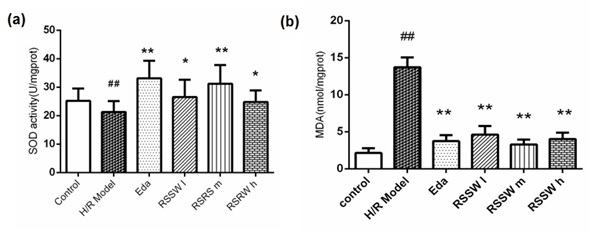 Figure 3: (a) The effect of RSSW on the activity of the intracellular SOD and (b) MDA of the rat cortical neurons exposed to H/R. Neurons were treated as the text described. The data were expressed as the mean ±SD (Zhu et al.).
Figure 3: (a) The effect of RSSW on the activity of the intracellular SOD and (b) MDA of the rat cortical neurons exposed to H/R. Neurons were treated as the text described. The data were expressed as the mean ±SD (Zhu et al.).##P<0.01, versus control group, **P<0.01?versus H/R model group.
RSSW effect on neuron intracellular ROS
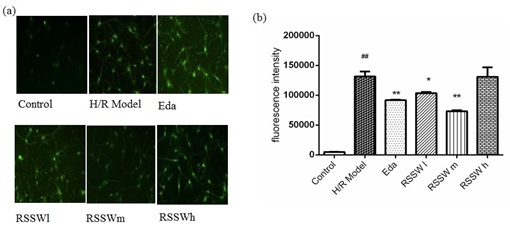 Figure 4: Effect of RSSW on ROS level in cortical neurons with hypoxia/reoxygenation injury. (a) Neurons were treated as the text described. Microscopic images of the cortical neurons staining with Dichlorofluorescein Diacetate (DCFH-DA), (b) Green Fluscence intensity (ROS level indicator) of different cortical neurons. The data were expressed as the mean ±SD (Zhu et al.).
Figure 4: Effect of RSSW on ROS level in cortical neurons with hypoxia/reoxygenation injury. (a) Neurons were treated as the text described. Microscopic images of the cortical neurons staining with Dichlorofluorescein Diacetate (DCFH-DA), (b) Green Fluscence intensity (ROS level indicator) of different cortical neurons. The data were expressed as the mean ±SD (Zhu et al.). ##P
RSSW effect on neuron mitochondrial membrane potential (Δψm)
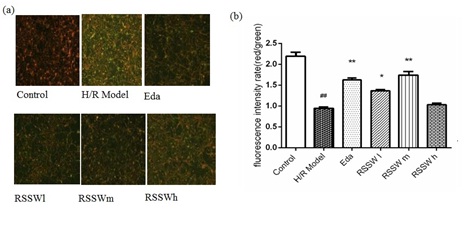 Figure 5: Effect of RSSW on mitochondrial membrane potential in neurons exposed to H/R injury ( , n=3) Neurons were treated as the text described. (a) Microscopic images of the cortical neurons staining with JC-1 MMP Assays kit, (b) red/green fluorescence intensity rate of different cortical neurons. The data were expressed as the mean ±SD (Zhu et al.).
Figure 5: Effect of RSSW on mitochondrial membrane potential in neurons exposed to H/R injury ( , n=3) Neurons were treated as the text described. (a) Microscopic images of the cortical neurons staining with JC-1 MMP Assays kit, (b) red/green fluorescence intensity rate of different cortical neurons. The data were expressed as the mean ±SD (Zhu et al.). ##P
RSSW effect on Caspase-3,9 activity in the neurons exposed to H/R
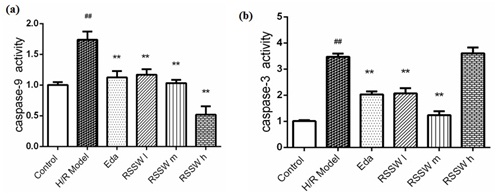 Figure 6: WSXN effect on rat cortical neuronal apoptosis protein caspase-9 and caspase-3 activity. Neurons were treated as the text described. The data were expressed as the mean ±SD, n=3 (Zhu et al.).
Figure 6: WSXN effect on rat cortical neuronal apoptosis protein caspase-9 and caspase-3 activity. Neurons were treated as the text described. The data were expressed as the mean ±SD, n=3 (Zhu et al.). ##P
DISCUSSION
Edaravon, a free radical scavenge?has been used for the treatment of ischemic stroke and other neurodegenerative disorders. It showed a protective effect on oxidative damage of neurons, via scavenging oxygen free radicals, inhibiting lipid peroxidation, which play a vital role in the treatment of cerebral ischemia [15]. We used edaravone as the positive agent to explore the mechanism of RSSW neuroprotection. Our study found that oxidative stress stimulation by Na2S2O4 induced apoptosis of the cultured rat cortical neurons. As like edaravone, RSSW protect neurons to alleviate the apoptosis and the leakage of LDH in the insulting neurons while increasing SOD activity and the mitochondrial membrane potential of the insulting neurons, and reducing the level of reactive oxygen species and the accumulation of MDA neuronal cells, which accompanied with the inhibition of the activity of caspase 3 and caspase 9 of the neurons. This indicates that RSSW might protect neurons from hypoxia/reoxygenation-induced injuries by reducing oxidative stress, which provided a scientifical data for the RSSW clinical usage in the treatment of oxidative stress related disorder such as ischemic stroke and neurodegenerative diseases.
Oxidative stress is a major pathophysiologic mechanism in the progression of stroke and neurodegenerative diseases, such as AD, PD and ALS [4-6]. In stroke, AD, and PD, abnormal brain metabolism caused by decreased cerebral blood flow is the cause of early pathological changes in hypoxic-ischemic encephalopathy and appears in pathophysiological responses that occur mainly in neuronal apoptosis [2-5]. Several studies have indicated that cell apoptosis is closely associated with oxidative stress. Oxidative stress results in a sharp increase in intracellular ROS, which causes a change in mitochondrial decreased membrane potential, damages to mitochondrial membrane structure, damages to mitochondrial DNA and the electron transfer chain, and disruption of mitochondrial function. These effects of oxidative stress eventually lead to neuronal apoptosis, degeneration, and death [6-9,21,22]. Indeed, mitochondrial dysfunction, increased membrane permeability, and decreased membrane potential are early markers of cell apoptosis [8,9].
Ischemia and hypoxia in the brain disrupt neuronal oxidative function and produce large amounts of free radicals. SOD-level indirectly responds to the body’s ability to scavenge oxygen free radicals [4-6,21,22] and thereafter free radical scavenging can alleviate neurological injury [23].
Neuron apoptosis is regulated by active genes, such as caspases, and is reversible under certain conditions by inhibiting or blocking the expression of caspases during treatment or intervention using certain special agents in the early apoptosis stage. This phenomenon may have a protective effect on insulted cells and may be beneficial to the functional restoration of the injured and damaged neuron [21,22].
The main pathway of apoptosis contains endogenous and exogenous processes, eventually brought together in a cascade of caspase-family effector proteins, which can be used during apoptosis to induce degradation of intracellular proteins [21-24]. Hypoxic injury caused by endogenous apoptosis mainly activates BH3-only family protein and, during a series of reactions that activate caspase-9, a cascade of downstream caspase-family effector proteins become activated, finally leading to cell apoptosis [21-24]. During this process, Caspase-3 is the key caspase cascade protein that serves as the common part of many downstream effector receptor-mediated apoptosis pathways [8,9,21]. Sometimes is Caspase in other caspase. Previous studies have suggested that H/R could damage nerve cells and cause apoptosis by producing excessive reactive oxygen [21,24]. Futhermore, it was found that H/R could activate caspase-3 activity, leading to apoptosis in cultured cortical neurons [24]. Therefore, inhibiting caspase-3 and caspase-9 activity can reduce the H/R neuronal damage that produces apoptosis, thereby increasing neuronal survival and improving neurological function.
Our study showed that RSSW could decrease the activity of caspase-3, and caspase-9 and inhibit cell apoposis in a dose-dependent manner. Although at high doses caspase-3 vitality was increased, overall our data indicated that RSSW protected neurons from apoptosis via inhibiting the activity of caspase-3 and caspase-9 and improving mitochondrial function.
The present study shows for the first time that RSSW significantly inhibits neuronal apoptosis, suppresses oxidative stress through inhibition of caspase 3 and 9, and protects neurons against hypoxic neuronal injury.
We previous reported that administration of RSSW (50, 100, and 200 mg/kg) has significant neuroprotective effects against MCAO and 4-VOI/R injury and a therapeutic effect on cognitive disorders in VD rats. In this study, 0.2~1 μg/ml of RSSW contains approximately 3.34 ng/g of total ginsenoside (including 1.74 ng/g Rb1 and 0.58 ng/g Rg1) and 5.42 ng/g of stilbene glucoside. This concentration is consistent with the actual concentration in the rat brain after administration of 100 mg/kg RSSW (unpublished data). It was reported that ginsenoside (the active ingredient of Panax ginseng) and stilbene glucoside (the active ingredient of fleece-flower root) were effective at protecting neurons from oxidative stress-induced neurotoxicitye at the concentration of 1~5 μg/ml [14,17-21]. This might indicate that RSSW showed neuroprotection via improving hypoxia injury through its active ingredients of ginsenoside and stilbene glucoside, which have been shown to have antioxidant properties, such as scavenging oxygen free radicals. It was noted in our experiment that a higher concentration (more than 5 μg/ml) of RSSW did not lead to greater neuroprotection when studying the reduction of ROS and MDA, inhibition of caspase 3 activity, apoptosis ratio to lower level, or increase of mitochondrial membrane potential. Our finding is consistent with other reports on the neuroprotective effects of some agents with antioxidant properties in oxidative stress models, which might contribute to their ability to produce free radicals that induce apoptosis [25]. The underlying cause remains to be further elucidated. It might also suggest that the dosage of RSSW and other antioxidants should be controled properly in the clinic use as not to give excessive dose to avoid the reverse effect and waste the herbs resources.
In conclusion, this study’s findings suggest that the neuroprotection of RSSW from hypoxia injuries may be related to its ability to increase intracellular endogenous antioxidant SOD, reduce ROS, improve mitochondrial membrane potential, and decrease caspase 3 and caspase 9 activity in neurons exposed to oxidative stress. These processes might represent the mechanisms underlying the use of RSSW in the prevention and treatment of neural disorders related to cerebrovascular ischemia and some neurodegenerative diseases, such as PD and AD.
ACKNOWLEDGMENT
This study was supported by grants from the Natural Sciences Funds, Republic of China No.81473588,2014, the Guangdong Province Science and Technology New Drug R & D Key Project No. 2013A022100041 and the Natural Science Foundation of Guangdong Province NO.2014A030313585.
REFERENCES
- Go AS, Mozaffarian D, Roger VL, Benjamin EJ, Berry JD, et al. (2013) Heart disease and stroke statistics--2013 update: a report from the American Heart Association, Circulation127: 6-245.
- Ward A, Arrighi HM, Michels S, Cedarbaum JM (2012) Mild cognitive impairment: disparity of incidence and prevalence estimates. Alzheimers Dement 8: 14-21.
- Crizzle AM, Classen S, Uc EY (2012) Parkinson disease and driving: an evidence-based review. Neurology 79: 2067-2074.
- Chen H, Yoshioka H, Kim GS, Jung JE, Okami N, et al. (2011) Oxidative stress in ischemic brain damage: mechanisms of cell death and potential molecular targets for neuroprotection. Antioxid Redox Signal 14: 1505-1517.
- Barnham KJ, Masters CL, Bush AI (2004) Neurodegenerative diseases and oxidative stress. Nat Rev Drug Discov 3: 205-214.
- Orrenius S, Gogvadze V, Zhivotovsky B (2007) Mitochondrial oxidative stress: implications for cell death. Annu Rev Pharmacol Toxicol 47: 143-183.
- Klein JA, Ackerman SL (2003) Oxidative stress, cell cycle, and neurodegeneration. J Clin Invest 111: 785-793.
- Andersen JK (2004) Oxidative stress in neurodegeneration: cause or consequence? Nat Med 10: 18-25.
- Park CB, Larsson NG (2011) Mitochondrial DNA mutations in disease and aging. J Cell Biol 193: 809-818.
- Shang XH, Xu XY (2013) Advance of experimental studies on protective effect of traditional Chinese medicines and their extracts on cerebral ischemia. China Journal of Chinese Materia Medica 38: 1109-1115.
- Liu DS, Zhou YH, Liang ES, Li W, Lin WW, et al. (2013) Neuroprotective effects of the Chinese Yi-Qi-Bu-Shen recipe extract on injury of rat hippocampal neurons induced by hypoxia/reoxygenation. J Ethnopharmacol 145: 168-174.
- Zhao J, Zhao Y, Zheng W, Lu Y, Feng G, et al. (2008) Neuroprotective effect of curcumin on transient focal cerebral ischemia in rats. Brain Res 1229: 224-232.
- Pharmacopoeia of the People’s Republic of China (2015), (Vol. 1), Chemical Industry Press, Beijing. Pg no: 468.
- Wan L, Cheng YF, Luo ZY, Guo HB, Gu QL, et al. (2015) Neuroprotection, learning & memory improvement of a standardized extract from Renshen Shuowu against neuronal injury and vascular dementia in brain ischemic rats. J Ethnopharmacol 165: 118-126.
- Lee BJ, Egi Y, Van Leyen K, Lo EH, Aria K (2010) Edaravone, a free radical scavenger, protects components of the neurovascular unit against oxidative stress in vitro. Brain res 1307: 22-27.
- Bei WJ, Peng WL, Zang LQ, Xie ZY, Hu DH, et al. (2007) Neuroprotective effects of a standardized extract of Diospyros kaki leaves on MCA Otransient focal cerebral ischemic rats and cultured neurons injured by glutamate or hypoxia. Plant a Medica 73: 636-643.
- Wei J, Fang WR, Sha L, Han D, Zhang R, et al. (2013) XQ-1H Suppresses Neutrophils Infiltration and Oxidative Stress Induced by Cerebral Ischemia Injury Both In Vivo and In Vitro. Neurochem Res.
- Li XB, Li Y, Chen JZ, Sun J, Li XF, et al. (2010) Tetrahydroxystilbene glucoside attenuates MPP+-induced apoptosis in PC12 cells by inhibiting ROS generation and modulating JNK activation. Neurosci Lett 483: 1-5.
- Xu S, He M, Zhong M, Li L, Lu Y, et al. (2015) The neuroprotective effects of taurine against nickel by reducing oxidative stress and maintaining mitochondrial function in cortical neurons. Neurosci Lett 590: 52-57.
- Sun FL, Zhang L, Zhang RY, Li L (2011) Tetrahydroxystilbene glucoside protects human neuroblastoma SH-SY5Y cells against MPP+-induced cytotoxicity. Eur J Pharmacol 660: 283-290.
- Warner DS, Sheng H, Batinic-Haberle I (2004) Oxidants, antioxidants and the ischemic brain. J Exp Biol 207: 3221-3231.
- Lewén A, Matz P, Chan PH (2000) Free radical pathways in CNS injury. J Neurotrauma 17: 871-890.
- Martinvalet D, Zhu P, Lieberman J (2005) Granzyme A induces caspase-independent mitochondrial damage, a required first step for apoptosis. Immunity 22: 355-370.
- Ueda H, Fujita R, Yashida A, Matsunaga H, Ueda M (2007) Identification of prothymosin-alpha1, the necrosis-apoptosis switch molecule under the in vitro ischemia condition. Neuroscience Research 58: 6.
- Bei W, Peng W, Ma Y, Xu A (2005) Flavonoids from the leaves of Diospyros kaki reduce hydrogen peroxide-induced injury of NG108-15 cells. Life Sci 76: 1975-1988.
Citation: Zhu J, Wan L, Zhu Y, Yang X, Cai Y et al. (2016) Rensheng ShouWu Protects Against Neuronal Injury by Inhibiting the Apoptosis Induced by Hypoxia and Reoxygenation. J Alzheimers Neurodegener Dis 2: 008.
Copyright: © 2016 Jing Zhu, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

