
Assessment of Fish-Farms Wastewaters Synergistic Impact on a Mediterranean Non-Tidal Lagoon
*Corresponding Author(s):
Mauro LenziLagoon Ecology And Aquaculture Laboratory Lealab, World Wide Fund, Orbetello (GR), Italy
Tel:39 3331090723,
Email:lealab1@gmail.com
Abstract
Keywords
INTRODUCTION
During the period 1995 to 2015, production of farmed aquatic species reliant on feeds increased more than fourfold, from 12 to 51 million tonnes, and today, 66% of total global aquaculture production (excluding aquatic plants) is produced using exogenous feed, mostly commercially manufactured. In fact, in the same period, production of industrial aquaculture feeds increased sixfold, from 8 to 48 million tonnes. However, the proportion of fish from capture fisheries being reduced to fishmeal and fish oil has been declining in recent decades, and it is projected that a growing share of fishmeal and fish oil production will be obtained from fish processing co-products, such as fish carcasses. Furthermore, the dietary inclusion rates of fishmeal and fish oil in aquaculture feeds have also been falling, increasingly replaced by crops, especially oilseeds [1,3]. Much research is being directed into novel aquaculture feedstuffs, including seaweed and insect sources, but it is likely to be some years before these become widely available and affordable [4,5]. Despite the efforts to make aquaculture more sustainable, energetically more advantageous and environmentally friendly solutions will have to be studied and found necessary [6].
Moreover, the impact of aquaculture, in its different forms, on the host environment must be seriously considered. The coastal areas, also due to the contribution of aquaculture, have been subjected to a growing eutrophication with dramatic variations in the benthic communities, with the death of fish and enormous microalgal and macroalgal developments [7-10]. In marine finfish aquaculture, according to [11], for recently formulated feeds, 69 kg of Nitrogen (N) and 10 kg of Phosphorus (P) are released into the environment per tonne of fish produced. The same researchers also estimated that future improvements in feed production will lead in 2050 to a reduction in the Nitrogen (N) and Phosphorus (P) releases to 55 kg and 7 kg per ton of fish produced, respectively.
It is thus necessary to evaluate the impact of this practice on natural ecosystems and how the latter react to stress. A more in-depth knowledge of this issue would allow better intervention to mitigate of the consequences more effectively.
In this study, we examined the impact of wastewater from two sea bass and sea bream land-based farms, which are released into a non-tidal coastal lagoon.
Lagoon environments, despite being environments of shallow waters and poor water exchange compared to the nearby sea, have, in their community as a whole, a high resilience and a great capacity to integrate stress factors. However, since they are already perturbed environments, the stress produced by anthropic pressure, in our case the wastewater from the two fish-farms, in not easily distinguishable from the natural structural stress. This peculiarity is called estuarine quality paradox [12,13].
The aim of this study is to highlight a possible impact gradient of the wastewater of the two fish farms on the various compartments of the lagoon ecosystem, highlighting the best indicator variables of this type of impact. In the working hypothesis, the effects of this impact should decrease as the distance from the sources of waste release increases.
MATERIALS AND METHODS
The study area and the fish-farms
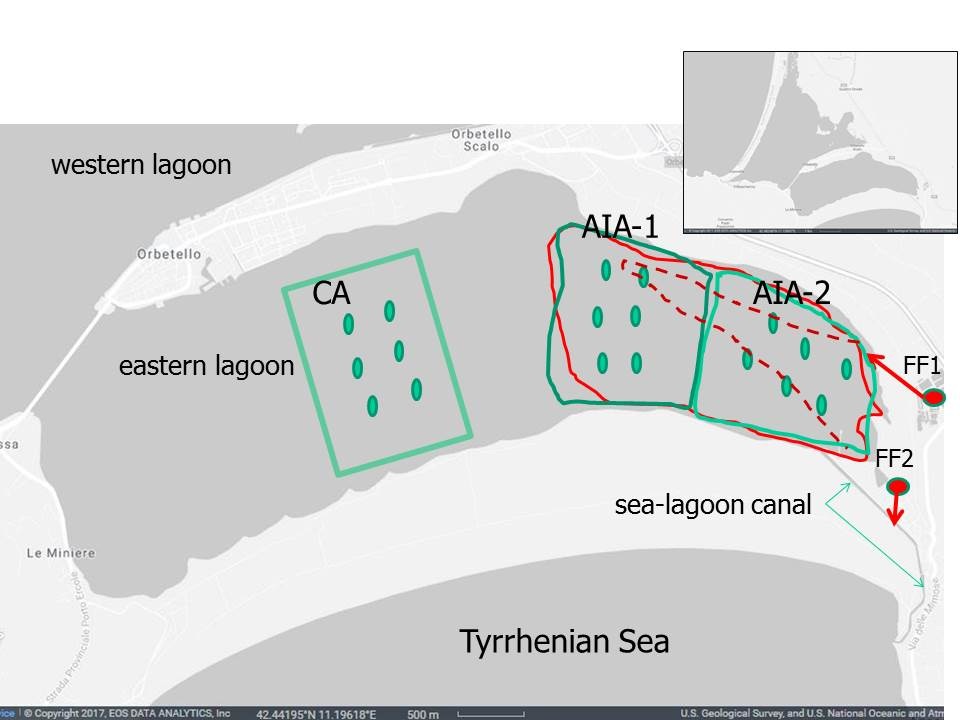
The environment is eutrophic due to fish-farm wastewaters, intermittent streams containing agricultural run-off and civil effluent, and historical input stored in sediment [14]. Due to high nutrient availability, morphology and low-water-turnover, this lagoon is subject to severe macroalgal proliferation, which can cause dystrophic crises with die-offs.
Owing to the low water renewal, sea water is pumped into the lagoon, between June and August, to promote water turnover, with input by two western pumping stations, at the mouth of the two western channels, and output by the eastern canal. This input creates a continuous one-way flow of about 13.000 L s-1, with a weak speed of 1.0-1.5 cm s-1. Although this water mass is relatively high, it follows short routes and does not allow sufficient water turnover in large part of the stagnating areas of the two basins, in shadow of this flow.
Therefore, in the summer months, in the eastern basin, the pumped waters coming from the western basin flow out towards the sea, with a very low flow velocity that can be hindered by a strong wind and rising of the marine front, which determine a consequent increase in the level of lagoon waters [15].
Two land based fish-farms discharge the wastewater in the easternmost part of the eastern basin, one (FF1) near the sea-lagoon channel, the other (FF2) in the middle of the same channel (Figure 1). Therefore during the pumping period it is probable that the FF1 wastewater is carried out by the outflow towards the sea and cannot extend much beyond the discharge area, while the FF2 wastewater flows mainly towards the sea, except for the periods in which a wind of contrast raise the sea front and let the waters enter from the sea, raising the lagoon level. In the remaining nine months, according to a study that used the CORMIX hydrodynamic model [16], the FF1 discharge mixes more and is removed towards the more central areas of the east basin, while the FF2 discharge follows the tidal flow, entering the lagoon at high tide. According to this study, the influx of wastewater extended towards the innermost areas of the east basin, along an extended brush about 1.8 km (Figure 1).
The two fish-farms breed sea-bass (Dicentrarchus labrax L.) and sea-bream (Sparus aurata L.), using water with salinity varying between 15 and 30, obtained by pumping from wells on brackish aquifers, which, due to a geothermal anomaly, are at constant temperature of 18-20°C.
FF1 consists of about 42 ground tanks covered in PVC of 400-600 m3 for a total of 22.400 m3 and a stream of outgoing waters of 560 L s-1, FF2 consists of 45 ground tanks covered in PVC with dimensions and volumes similar to the previous ones, for an outgoing flow of 420 L s-1.
Overall, fish production is 700-800 tonnes a-1. Both fish farms are equipped with a system of small basins and sewage settling channels, for an extension of about 2 ha each, in which detritivorous fish (mugilids) are placed and where microalgae and macroalgae develop.
The experimental design
Three different compartments of the lagoon ecosystem have been considered: Water, sediment and macroalgae. The characterization of the three compartments took place in May 2017, in condition of incoming tide, aiming to highlight the wastewater dispersion towards the eastern lagoon centre. According to the results of May 2017, only macroalgae and sediment has been the subject of subsequent 5 campaigns: November 2017 and February, May, September, and November 2018.
Sampling and analytical determinations
In each of the 3 selected eastern lagoon areas (AIA-1, AIA-2 and CA), 6 sampling and measurement points were identified, arranged along two parallel transects (Figure 1). In May 2017, using a multi-parameter probe, Temperature (T, °C), pH, Salinity (S, psu), Dissolved Oxygen (DO, mg L-1) and Nephelometric Turbidity (NTU) were measured in duplicate in each point. Water samples were then taken in duplicate in the same points and at the exit of the two fish farm wastewaters (FF1, FF2). Water samples were stored in a light-free and refrigerated environment, and then transported to the laboratory in a few hours for analytical determinations. Samples were filtered at 0.45 µm and the following analytical determinations were conducted: Ammonium nitrogen (N-NH4+), nitrous Nitrogen (N-NO2), nitric Nitrogen (N-NO3), Total Dissolved Nitrogen (TDN), Total Dissolved Phosphorus (TDP) and Soluble Reactive Phosphorus (SRP). The analyses were conducted according to APAT IRSA-CNR [17]. Dissolved Inorganic Nitrogen (DIN=N-NHNH4++N-NO2+N-NO3), Dissolved Organic Nitrogen (DON=TDN-DIN), Dissolved Organic Phosphorus (DOP=TDP-SRP) and the atomic ratio DIN:SRP were then computed.
Sediment
To determine sedimentary content of organic Carbon (C), Nitrogen (N) and Phosphorus (P), in the same water sampling points (Figure 1), sediment samples were taken in May 2017, with a horizontal core drill, able to collect in the surface layer of the first 3-4 cm, using a 60 mL syringe. The sediment samples were transferred from the syringe into the polyethylene containers of the same volume and refrigerated and subsequently frozen pending for analysis. The samples were dried to constant weight at 75°C, and then subjected to analytical determinations. N and C were determined using an elementary analyser (CHN Thermoquest, model 1110), P according to Aspila, et al., [18]. Using the percentages of the three estimated macronutrients, the molar ratios C:N, C:P, N:P were subsequently calculated.
To determine the amount of organic matter present in the lagoon sediments as a labile fraction (LOM), sediment samples were collected in the same points, using the previously described method, in November 2017 and February, May, September and November 2018. The determination was carried out as combustion at 250°C in a muffle after reaching the constant dry weight (75°C) [19].
In February 2018, 3 samples were collected per area using the horizontal sampler, in order to define the texture of the first 3-4 cm of the sediments, in the sand, silt and clay components.
In November 2018, the detrital fraction >1 mm (cd) dried at 75°C, essentially consisting of shell debris, was considered for all sediment samples and was calculated as a percentage of the total of the sample according to the following equation: cd% = fraction>1mm*100: (fraction<1mm+fraction>1mm).
Macroalgae
In May and November 2017 and in February, May, September and November 2018, in each of the 3 areas, the number of macroalgal species present and the overall biomass were determined.
The Total Coverage (CT) of the substrate by the algal mats was estimated through Sentinel-2 satellite images obtained from the Land-Viewer site (EOS DATA ANALYTICS, USGS/NASA), and calculated through the Fiji-Image software. In the field, immediately following that of the available satellite image, the Biomass (b) was determined by collecting the plant material contained in a 60*60 cm panel lowered to 6 points per area, distributed according to the satellite images.
The material collected inside the box was drained for a few minutes and weighed in field with a portable electronic scale with a sensitivity of ±0.5 g. The data obtained were transformed to the surface unit of 1 m2 (transformation factor 2.778) and expressed as kg wet weight m-2. For the determination of Standing Crops (SC), the algal mass present in a given lagoon surface at the time of detection, the following equation was applied: SC=b*CT*1000-1, where: SC is the standing crop expressed in Tonnes Wet Weight (Tww); b, the biomass expressed in kgww m-2; CT, estimated total coverage with Fiji software in m2; 1000-1, the factor for bringing the final value to tones.
On the basis of described samplings, the specific Dominance (d) per point-station and the percentage of opportunistic species (% os) on the total species observed were determined.
In May 2017, samples of the most widespread species, common to the selected areas, were collected in each of the three areas, to determine C, N, P content. These samples were washed with sea water to remove debris and other impurities, transferred into plastic bags, stored in the dark, refrigerated and transported to the laboratory in few hours. The material was then quickly washed with fresh water, dried with tissue paper, dried at 40°C in a dryer with ventilation, and then further cleaned of impurities and small animals. The sample was stored in polyethylene containers in a dry place, up to the laboratory determinations. The analysis was carried out employing the same methods as described above for the sediment on samples dried to constant weight of 75°C. The molar ratios C:N:P and C:N were subsequently calculated.
Statistical analysis
LOM and biomass data were processed by two-way ANOVA to detect significant differences between the month (Nov17-Nov18, 5 levels for LOM; May17-Nov18, 6 levels for biomass) and area (CA, AIA-1, AIA-2; 3 levels) fixed and orthogonal factors.
Cochran’s C-test was used before each analysis to check for homogeneity of variance [20], and datasets were transformed where necessary. The Student Newman Keuls (SNK) test was used for a posteriorimultiple comparisons of means.
A regression analysis was performed in order to evaluate possible correlation between cd and LOM content in the sediment, with Oct-18 data-set (6 records per area). The degree of correlation between cd content of each studied area and the LOM one was calculated and reported as the squared correlation coefficient (determination coefficient, R2).
All the statistical analysis was performed with the Statistica 10.0 software, and the critical value in all tests was P=0.05.
RESULTS
|
CA |
AIA-1 |
AIA-2 |
|
|
T |
22.32±0.13 |
23.03±0.34 |
23,92±0.95 |
|
pH |
9.15±0.22 |
9.19±0.05 |
9,06±0.17 |
|
S |
37.00±0.82 |
34.75±0.99 |
26,17±4.14 |
|
DO |
6,27±1.00 |
8.42±1.06 |
9,20±1.41 |
|
NTU |
1.58±0.43 |
0.98±0.28 |
2,88±1.10 |
|
CA |
AIA-1 |
AIA-2 |
FF1 |
FF2 |
|
|
N-NH4 |
35.63±18,01 |
27.52±22.53 |
11.86±13.34 |
142.61±14.54 |
103.68±3.46 |
|
N-NO2 |
0.27±0.45 |
0.07±0.00 |
1.15±1.55 |
20.61±0.11 |
8.46±0.12 |
|
N-NO3 |
40.38±1.32 |
45.45±3.57 |
48.06±4.28 |
127.60±0.96 |
50.18±0.18 |
|
DIN |
76.29±19.25 |
73.05±21.06 |
61.07±15.93 |
290.82±15.61 |
162.32±3.39 |
|
TDN |
848.40±186.97 |
766.25±399.08 |
685.88±175.86 |
344.36±12.79 |
654.39±2.75 |
|
DON |
772.12±190.91 |
693.20±407.22 |
624.81±172.02 |
53.54±2.82 |
492.07±0.64 |
|
TDP |
0.35±0.26 |
0.60±0.53 |
0.80±0.34 |
8.58±1.11 |
6.08±0.37 |
|
SRP |
0.07±0.08 |
0.15±0.13 |
0.42±0.27 |
7.45±0.77 |
5.04±0.02 |
|
DOP |
0.23±0.20 |
0.45±0.57 |
0.38±0.10 |
1.12±0.34 |
1.04±0.39 |
|
DIN/SRP |
2103±1067 |
1399±1272 |
285±302 |
39±2 |
32±1 |
The ANOVA analysis showed a significant effect of the area factor on the variables T, S, DO and NTU (P=0.0023, P<0.0001, P=0.0030, P=0.0012, respectively). The post hoc SNK showed significantly higher T values towards the sources (AIA-2), compared to the other two areas (CA=AIA-1), while the S values were significantly lower in AIA-2 (P<0.01). DO was significantly lower in CA than the other two areas (P<0.01) (AIA-1=AIA-2); for NTU, each area was significantly different from the other, with higher values in AIA-2 (P<0.01) and lower in AIA-1 (P<0.05).
For nutrients, ANOVA showed a significant effect of the area only for N-NO3, SRP and DIN:SRP (P=0.0072, P=0.0122, P=0.0289, respectively). With the post hoc SNK, N-NO3 values resulted significantly lower in CA than in AIA-1 (P<0.05) and AIA-2 (P<0.01), while the two AIA areas were similar. The SRP values in CA and AIA-1 were significantly lower than in AIA-2 (P<0.01, P<0.05, respectively), while the values of CA and those of AIA-1 were similar. The values of DIN:SRP were significantly higher in CA than in AIA-2 (P<0.05).
|
sand |
silt |
clay |
cd |
|
|
% |
% |
% |
% |
|
|
CA |
79.80±0.50 |
14.60±0.10 |
5.60±0.40 |
10.57±5.26 |
|
AdI-1 |
78.65±0.45 |
14.30±0.20 |
7.05±0.25 |
17.44±11.19 |
|
AdI-2 |
84.40±0.50 |
9.45±0.85 |
6.15±0.35 |
30.75±15.81 |
Table 4 shows the means (±SD) of C, N, P content and the relative molar ratios C:N, C:P and N:P for the May-17 sampling. ANOVA showed a significant effect of the area factor only on the variables C:P and N:P (P=0.0003 e P<0.0001, respectively). With the post hoc SNK, the estimated values in AIA-2 for these two variables were significantly lower (P<0.01) than those found in the other two areas (CA=AIA-1).
|
CA |
AIA-1 |
AIA-2 |
|
|
%C |
4.40±2.57 |
6.48±0.28 |
5.10±2.69 |
|
%N |
0.52±0.32 |
0.73±0.04 |
0.52±0.32 |
|
%P |
0.04±0.02 |
0.06±0.01 |
0,09±0.05 |
|
C:N |
10.87±2.90 |
10.34±0.65 |
14.60±3.22 |
|
C:P |
287.18±62.53 |
280.22±23.14 |
163.48±31.82 |
|
N:P |
27.15±2.99 |
27.14±1.94 |
12.23±3.62 |
The percentages (±SD) of Labile Organic Matter (LOM) in sediment are shown in table 5, for the period from November 2017 to November 2018. ANOVA showed a significant effect of the area factor (P<0.0001), and the post hoc SNK showed lower values in AIA-2 (P<0.01) compared to the other two areas.
|
17-Nov |
18-Feb |
18-Jun |
18-Sep |
18-Nov |
||
|
LOM |
CA |
9.36±3.19 |
11.63±3.08 |
11.57±1.36 |
9.50±1.21 |
10.50±3.46 |
|
AIA-1 |
11.94±3.93 |
10.31±1.95 |
12.80±4.72 |
12.70±5.15 |
10.64±4.17 |
|
|
AIA-2 |
7.56±0.73 |
8.21±0.53 |
8.82±1.87 |
7.72±0.51 |
7.26±2.23 |
As shown in figure 2, an inverse correlation between LOM and Carbonate Debris (cd) content was found. Although the degree of correlation was R2=0.3437, the regression was significant (F=10.34; P=0.0054), indicating a significant loss of LOM at the increasing cd content in the sediment.
 Figure 2: Regression analysis performed in order to evaluate possible correlation between calcareous shell debris (cd%) and Labile Organic Matter (LOM%) content in the sediment, with November 2018 data-set (6 records per area, 18 in total).
Figure 2: Regression analysis performed in order to evaluate possible correlation between calcareous shell debris (cd%) and Labile Organic Matter (LOM%) content in the sediment, with November 2018 data-set (6 records per area, 18 in total).|
CA |
AIA-1 |
AIA-2 |
||
|
Spyridia filamentosa (Wulfen) Harvey |
R |
X |
X |
X |
|
Gracilaria gracilis (Stackhouse) Steentoft |
R |
X |
X |
X |
|
Gracilariopsis longissima (S.G. Gmelin) Steentoft, L.M. Irvine & Farnham |
R |
X |
||
|
Polysiphonia sp. |
R |
X |
X |
X |
|
Ceramium sp. |
R |
X |
X |
|
|
Alsidium corallinum C. Agardh |
R |
X |
X |
|
|
Sphaerococcus coronopifolius Stackhouse |
R |
X |
X |
|
|
Dasia ocellata (Grateloup) Harvey |
R |
X |
||
|
Ulva rigida C. Agardh |
C |
X |
||
|
Ulva prolifera O.F.Müller |
C |
X |
X |
|
|
Valonia aegagropila C. Agardh |
C |
X |
X |
X |
|
Chaetomorpha linum (O.F.Müller) Kützing |
C |
X |
X |
X |
|
Cladophora vagabunda (L.) Hoek |
C |
X |
||
|
Cystoseira barbata (Stackhouse) C. Agardh |
O |
X |
X |
|
|
Dictyota dichotoma (Hudson) J.V.Lamour. |
O |
X |
X |
|
|
n |
8 |
11 |
12 |
|
|
% s |
53.3 |
73.3 |
80 |
|
|
% os |
50 |
45.5 |
66.7 |
In table 7, the average (±SD) of the biomass (b, kg wet weight m-2), the corresponding Standing Crop (SC, in Tww), the total coverage in hectares compared to the overall surface of each area (CT, estimated using Fiji software) and the dominant species (d) are reported for each area and for each survey. The time course of SC for the three areas is shown in figure 3.
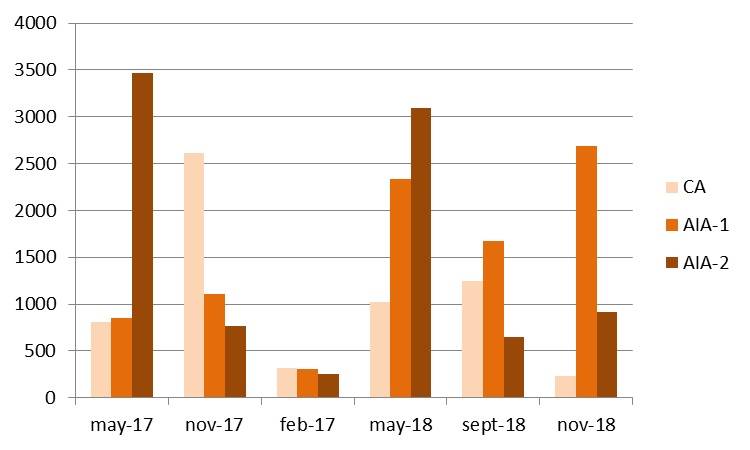 Figure 3: Seaweed standing crop in Tonnes Wet Weight (TWW), between May 2017 and November 2018, in CA, AIA-1 and AIA-2 areas, 135 hectares each.
Figure 3: Seaweed standing crop in Tonnes Wet Weight (TWW), between May 2017 and November 2018, in CA, AIA-1 and AIA-2 areas, 135 hectares each.|
CA |
AIA-1 |
AIA-2 |
||
|
17-May |
b |
1,04±0,86 |
0,93±0.62 |
3,15±1,85 |
|
CT |
0.58 |
0.68 |
0.82 |
|
|
SC |
808 |
848 |
3468 |
|
|
d |
s, G>V |
S, G, E |
G>E, CH |
|
|
17-Nov |
b |
2,25±1,56 |
1,44±1.61 |
0.86±0.62 |
|
CT |
0.68 |
0.57 |
0.66 |
|
|
SC |
2617 |
1104 |
768 |
|
|
d |
a, V |
a, G, S |
S, a, CH |
|
|
18-Feb |
b |
0,60±0,88 |
0,41±0,32 |
0,37±0,39 |
|
CT |
0.4 |
0.55 |
0.51 |
|
|
SC |
322 |
306 |
257 |
|
|
d |
a, V |
G, S |
G |
|
|
18-May |
b |
1.89±0.69 |
3,14±1,02 |
4,51±2,53 |
|
CT |
0.49 |
0.51 |
0.77 |
|
|
SC |
1021 |
2331 |
3098 |
|
|
d |
a>S |
S>G, Cb |
G>>S |
|
|
18-Sep |
b |
2.31±1.23 |
2.25±1.61 |
0.94±0.36 |
|
CT |
0.4 |
0.55 |
0.51 |
|
|
SC |
1245 |
1672 |
647 |
|
|
d |
A |
S |
S, G |
|
|
18-Nov |
b |
0.67±0.25 |
3.06±1.99 |
0.88±0.16 |
|
CT |
0.26 |
0.65 |
0.77 |
|
|
SC |
234 |
2688 |
913 |
|
|
d |
a>CH>V |
S>a>G |
G, CH |
The pattern of biomass observed in the 3 areas, in the various sampling months, indicates that there were no significant biomass variations for CA, while AIA-1 showed significant changes between February 2018 and May 2018 and November 2018 (P<0.05); AIA-2, on the other hand, confirmed a significant variation in the biomass between the two spring months and the remaining months (May-17≠Nov-17, Feb-18, Set-18, Nov-18, P<0.05; May-18≠Nov-17, Feb-18, Set-18, Nov-18, P<0.01; the other months did not show significant differences between them).
The dominant vegetation was constituted by Gracilariopsis longissima>>Chaetomorpha linum>Ulva prolifera in AIA-2. Elsewhere, a winter carpet of low thickness prevailed, consisting mainly of a mixture of Gracilaria gracilis and Spyridia filamentosa, alternating, in the other seasons, a higher biomass with high dominance of Alsidium corallinum in CA, and Sphaerococcus coronopifolius in AIA-1 (Table 7).
In table 8, number of analysed samples (n), means (±SD) of C, N, P content in dried matter of C. linum, U. prolifera and Gracilaria spp., and the relative molar ratios C:N:P are reported, for each area in the May 2017 survey.
|
n |
C |
N |
P |
C:N |
C:P |
N:P |
||
|
CH |
CA |
6 |
25.73±2.34 |
1.06±0.39 |
0.050±0.020 |
33.11±13.99 |
1402±488 |
44±8 |
|
AIA-1 |
2 |
22.83±2.27 |
0.67±0.03 |
0.048±0.011 |
39.77±5.70 |
1260±169 |
32±9 |
|
|
AIA-2 |
6 |
24.87±3.14 |
1.44±0.26 |
0.140±0.060 |
21.03±5.97 |
546±221 |
26±6 |
|
|
G |
CA |
2 |
39.81±3.59 |
2.65±0.53 |
0.110±0.013 |
17.72±1.95 |
933±22 |
53±5 |
|
AIA-1 |
2 |
35.83±0.91 |
1.86±0.18 |
0.076±0.003 |
22.57±2.70 |
1214±83 |
54±3 |
|
|
AIA-2 |
7 |
31.09±4.74 |
1.76±0.35 |
0.121±0.045 |
21.37±6.15 |
712±180 |
34±9 |
|
|
U |
CA |
1 |
19.19 |
1.02 |
0.06 |
22.06 |
828 |
38 |
|
AIA-1 |
3 |
22.77±1.94 |
0.96±0.43 |
0.062±0.012 |
30.53±9.92 |
969±115 |
34±8 |
|
|
AIA-2 |
4 |
22.96±3.84 |
1.60±0.62 |
0.094±0.027 |
17.98±4.41 |
616±143 |
31±3 |
The sample numbers allowed only for C. linum a macronutrients content comparison by Student T-test, between the areas CA and AIA-2. There were no significant differences for the carbon (P=0.6329) and nitrogen (P=0.0917) content, while a significantly higher phosphorus content was found in AIA-2, compared to CA (P=0.0108).
C. linum and Gracilaria sp. showed similar C:N:P ratios in CA and in AIA-1, while lower values were found in AIA-2. The same result was found, though less markedly, for U. prolifera. Lowest C:N ratio were found in AIA-2 for C. linum and U. prolifera. Values of the N:P ratio around 30, indicating P-limitation [21,22], were observed in CA for C. linum, more markedly in CA and AIA-1 for Gracilaria sp. and slightly in CA for U. prolifera.
Carbon content remained substantially stable for the various species with the variation of the area, and Gracilaria sp. had the highest content. N values were discordant, higher in AIA-2 for C. linum and U. prolifera, but lower for the nitrophilous Gracilaria sp., while for the first two the lowest values were estimated in AIA-1. P was markedly higher in all samples collected in AIA-2.
DISCUSSION
Water
Some significant variations, however, are highlighted for salinity and temperature. The two fish farm wastewaters lower the salinity (significant changes in AIA-2 compared to the other two areas), despite the proximity of a marine mouth, thereby contributing to countering summer evaporation and mitigating the temperature, counteracting excessive lowering in winter and raising in summer. This result, on the one hand highlights the poor marine turnover in the 9 months in which there is the natural flow of the tides, on the other it suggests that the mitigation of salinity conditions could affect macro and microalgal growth, favoring it even in extreme seasons.
The fact that DO was found to be significantly lower in CA than in AIA areas in May 2017, could be the consequence of the significant increase in the presence of plant biomass in AIA-2, but, as in the case of NTU, which resulted significantly among all areas, many possible factors might have affected this result.
A gradient was detected for N-NO3 and SRP, with significant increase towards AIA-2, especially for the last variable. The increase in P in AIA-2, closer to the fish farms discharge, is the cause of the significant reduction in the DIN:SRP molar ratio in that area compared to the other two.
As can be seen from table 2, N-NH4+, N-NOx and DON conveyed with fish-farms wastewater were 42%, 37% and 16% for FF1, and 16%, 9% and 75% for FF2, respectively. These decidedly different percentages between the two fish-farms, although they breed the same species at the same densities, could be due to the different conformation of the settling basins and canals and to their dynamics, so it is possible that uptake or release processes and decomposition of plant masses take place at different times and quantities. Among the inorganic chemical species, N-NH4+ is normally dominant in fish-farm wastewater, since it is the major excretion product of nitrogen from fish [24], however, in the three areas it showed large fluctuations in data sets and the averages are paradoxically decreasing towards the source. On the contrary, the oxidized form prevailed in the area closest to the wastewater. This could be due to an intense nitrification favored by the high DO values produced by macroalgae, moreover, the same macroalgae that in AIA-2 are present with Gracilaria-Gracilariopsis dominance, could have subtracted particularly ammonium at spring temperatures [25].
Giordani, et al. [26], have hypothesized that the environmental quality value for the variable DIN is inversely proportional to the value of the variable itself. DIN values greater than 100 µM, correspond to the lowest quality (score=0), while values of DIN=0 µM to the highest quality (100). The same Authors suggested the best quality (100) is found at SRP=0 µM and the worst (0) at values >6 µM. Viaroli, et al. [23] suggested that, for transition environments, the optimal condition for good productivity is in the range from 0 to 20 µM of DIN, although other Authors considered 20 µM the critical threshold for the coastal lagoons for this variable [27,28]. Therefore, the concentrations of DIN in water found in the present study indicate scarce environmental quality in all three areas examined, including the control area. In fact, the average values of DIN ranged from 61 to 76 µM, while SRP values were lower than 1 µM.
DIN abundance in all the examined areas resulted in very high values of the DIN:SRP atomic ratio, which progressively decreases moving closer to the source. This indicates P-limitation in all the three areas [21,22], including AIA-2, the nearest to the nutrient input of fish farm wastewaters, while only for the wastewater of the two fish farms DIN:SRP reached the lowest values (39 for FF1 and 32 for FF2). However, it is clear that high values of the DIN:SRP ratio in AIA-1 and AIA-2, do not express a real limitation of P, but a relative deficiency of P, an imbalance of abundance, since N concentrations were decidedly greater than P, which is not lacking.
DON, in all the areas, and DOP, to a lesser extent, were the most abundant components of dissolved nitrogen and phosphorus in the water column, varying between 87% and 91% of TDN and between 46% and 79% of TDP. High values of DON and DOP have been reported in other studies of this lagoon [29-31], and are probably a characteristic of eutrophic environments with algal blooms, due to the release of cellular exudates and the presence of macromolecules that come from cell lysis, from the decomposition of plant masses and from bacterial extracellular enzymatic activities, in an environment that has a relatively modest water mass and abundant primary production. Different DON content in fish farms wastewater could be due to processes occurring in the settling basins.
Sediment
A significant difference was found for LOM between all the three areas, with unexpected higher values in CA and AIA-1 than in AIA-2. Higher values of LOM in areas more distant from the nutrient source, and the variability of nutrient data are probably due to the granulometric differences of the sediments of the sampling areas (Table 3). In fact, there is a significant inverse correlation between organic matter and Calcareous Detritus (cd) produced by shells (Figure 2), whose relative deficiency was probably the cause of the highest LOM values in CA and AIA-1. Sediments with sandy dominance and high quantities of coarse shelled debris retain the organic components to a lesser extent and favor interstitial oxygen penetration enhancing nitrification/denitrification processes [32]. This was the case of AIA-2, the area with the lowest LOM content, while the highest LOM values in AIA-1 and in CA were probably due to a relative lower content of sand. Sand was lower in AIA-1 than in CA, but the first had higher values of shell debris. These results did not allow identifying the fish farms input as a source of LOM that seems mainly due to the lagoon intrinsic dynamics.
Phosphorus in sediments is present as insoluble orthophosphate adsorbed by other mineral components [33,34]. It is probable the high variability in P accumulation, that was found within a same area and among the three areas, could be also attributable to the variable granulometry of the sediment.
N:P molar ratio in CA and AIA-1 sediments was similar to the average value of 30 estimated by Atkinson and Smith [21] in thalli of different macroalgal species, which would suggest that it derives directly from the decay of masses macroalgal, as substantially confirm the molar ratios N:P in the algal thalli, a little more P-limited (Table 8). In AIA-2, the values were decidedly lower than 30, indicating, although without statistical support, a lower relative presence of N, which reflects the N:P values of the thalli, but where this value is due to a greater accumulation of P.
Macroalgae
In this lagoon basin, two marine species are commonly found, Alsidium corallinum e Sphaerococcus coronopifolius, which seem to well tolerate the eutrophic conditions, degenerating to a large extent during the hot season, but developing shortly afterwards. Although thalli may occasionally be found everywhere, they have two distinct areas in which development is dominant: A. corallinum, in CA and widens a little towards the westernmost part of the eastern basin; S. coronopifolius, essentially in AIA-1. Therefore, the three areas of this study are characterized by dominance of different three algal species. However, the presence of the sea-lagoon communication channel in AIA-2 further could confuse the picture because typically marine species can be found in AIA-2 and AIA-1 occasionally conveyed with the waters of the incoming tide, as is undoubtedly the case of the Rhodophyta Dasia ocellata observed in AIA-1 in May 2018.
This horizontal distribution certainly cannot be random, also because it is stable over time, as we have observed over the years (unpublished data). Many factors may have affected this distribution: nutrients, salinity, sediment grain size, LOM accumulation, light radiation, hydrodynamism. Certainly, the nutrient intakes of fish farms may have contributed, as in part the N and P accumulations in thalli suggest, however the high nutritional availability can constitute a limit for many marine algal species, for which the optimal conditions are normally oligotrophy or mesotrophy, and they cannot tolerate lagoon eutrophy or hypertrophy due to continuous external nutrient inputs. Therefore, a relative nutritional abundance could be one of the factors that prevent the development of A. corallinum and S. coronopifolius in AIA-2. However, these two species could be further confined to their respective areas of dominance by salinity values. In fact, in CA there is salinity values similar to those of the near sea and quite stable, and in AIA-1 the values are a little lower and equally stable; it is in AIA-2 that the salinity values are the lowest, with greater variability than the other two areas, which is also accompanied by a greater thermal rise (Table 1). This combination of factors, to which the fish-farm wastewaters contribute to a great extent, could favor the G. longissima competition in AIA-2 but cause its summer decay, while the other two species find more favorable conditions in the other areas and result to survive to summer criticality.
C. linum physiologically tends to P-limitation, so as soon as phosphorus becomes available it accumulates this nutrient and then uses it when needed [36], therefore this behavior would explain its relative abundance in AIA-2, compared to the other two areas, although less competitive than G. longissima. Although the records were relatively few, both G. longissima and U. prolifera showed the highest value of P among all the samples analysed in AIA-2, highlighting a predisposition to eutrophication of these species.
CONCLUSION
In a non-tidal lagoon, water column and sediment can present large variations of environmental variables, both chemical-physical and nutritional, in a very complex dynamic, influenced by many factors: The wind thrust on the lagoon water masses; the tides, however weak; the sea front (sea level that lowers or rises following the wind direction, favoring the outflow of the lagoon waters towards the sea, or preventing it); the stratification of water masses with different thermal and salt characteristics; the sediment resuspension by wind and human activities; the natural predisposition to eutrophication; sedimentary texture; bioturbation by the infauna and fish schools. All these factors produce a complex picture and can hinder the source of impact whose environmental impact is to be established.
In the case under examination, the heterogeneous data sets did not allow, for all the considered variables, a clear assessment of the environmental impact of the two fish-farm wastewaters in the area for impact assessment. For the dissolved nutrients in the water column, only N-NO3 and SRP were significantly higher, and DIN:SRP atomic ratio significantly lower, in the area closest to wastewaters discharges, compared to control area. For the sediments, among all the variables, only C:P and N:P showed significant values in AIA-2 to support a nutrient impact. By contrast, LOM had significantly lower values in AIA-2, contrary to expectations. It was clear that the presence of detrital organic matter is strongly influenced by sedimentary texture, which sees in AIA-2 greater presence of sand and limestone detritus and shell >1 mm, which favor both oxidation and removal.
Macroalgae, on the other hand, provided clearer information than water and sediments, both in terms of biomass, higher in the area closest to wastewaters discharges than in the other two areas, and in terms of the number of opportunistic species, more present in AIA-2. A different horizontal stratification of macroalgal species was highlighted in this study, with the dominance of G. longissima in AIA-2, S. coronopifolius in AIA-1 and A. corallinum in CA, although for these last two species it was not possible to establish if and to what extent their location depends on the influence of fish-farm wastewater. In a previous study concerning the influence of wastewater from urban treatment plant in the western basin of the same lagoon, a clearer horizontal stratification of nitrophilous and P demanding species was observed near the nutrient input.
Also for the tissue content of the C. linum thalli, it was possible to highlight a significant difference due to the higher levels of P in the thalli collected in AIA-2 compared to those collected in CA, and also the few records of the other two algal species have shown the same behavior; this suggests macroalgae can better highlight the contributions of P, compared to sediment variables, due to the differences in texture. Eventually, the incidence of fish farm wastewaters on macroalgae seemed to occur predominantly in AIA-2 area.
However, even for submerged vegetation there can be numerous factors that can confuse, mask or amplify the extent of the impact. For example, even in other areas of the same lagoon, the wind can bring the pleustophytic algal masses away from the area of influence and accumulate in different areas; therefore it is necessary to take these factors into account.
This study suggests that the variables that can better and more quickly highlight a eutrophic anthropogenic impact in an already eutrophic, low-turnover and shallow water environment, such as lagoons or estuaries, are those related to macroalgal vegetation. The water and sediment variables require many sampling points, relatively long times and a complex study plan, with a strong uncertainty on the final result. On the contrary, the picture provided by the qualitative-quantitative distribution of macroalgae and their content in tissue macronutrients can quickly provide a reliable assessment of the extent of anthropogenic impact.
ACKNOWLEDGMENT
REFERENCES
- FAO (2018) The State of World Fisheries and Aquaculture 2018. FAO, Rome, Italy.
- Bostock J, McAndrew B, Richards R, Jauncey K, Telfer T, et al. (2010) Aquaculture: Global status and trends. Philos Trans R Soc Lond B Biol Sci 365: 2897-2912.
- Tacon AGJ, Hasan MR, Metian M (2011) Demand and supply of feed ingredients for farmed fish and crustaceans. FAO, Rome, Italy.
- FAO (2012) The State of World Fisheries and Aquaculture 2012. FAO, Rome, Italy.
- Troell M, Naylor RL, Metian M, Beveridge M, Tyedmers PH, et al. (2014) Does aquaculture add resilience to the global food system? Proc Natl Acad Sci U S A 111: 13257–13263.
- Lenzi M (2013) The future of aquaculture. Journal of Aquaculture Research & Development 4: 106.
- Hauxwell J, Valiela I (2004) Effects of nutrient loading on shallow seagrass-dominated coastal systems: Patterns and processes. In: Nielsen SL, Banta GT, Pedersen MF (Eds). Estuarine Nutrient Cycling: The Influence of Primary Producers Pg no: 59-92.
- Charlier RH, Morand P, Finkl CW, Thys A (2006) Green tides on the Brittany coasts. Environmental Research Engineering and Management 3: 52-59.
- Liu D, Keesing JK, Xing Q, Shi P (2009) World’s largest macroalgal bloom caused by expansion of seaweed aquaculture in China. Mar Pollut Bull 58: 888-895.
- Liu F, Pang S, Chopin T, Gao S, Shan T, et al. (2013) Understanding the recurrent large-scale green tide in the Yellow Sea: Temporal and spatial correlations between multiple geographical, aquacultural and biological factors. Mar Environ Res 83: 38-47.
- Bouwman AF, Beusen, Overbeek AHW, Bureau CC, Pawlowski DP, et al. (2013) Hindcasts and future projections of global inland and coastal nitrogen and phosphorus loads due to finfish aquaculture. Reviews in Fisheries Science 21: 112-156.
- Dauvin JC (2007) Paradox of estuarine quality: Benthic indicators and indices, consensus or debate for the future. Mar Pollut Bull 55: 271-281.
- Elliott M, Quintino V (2007) The estuarine quality paradox, environmental homeostasis and the difficulty of detecting anthropogenic stress in naturally stressed areas. Mar Pollut Bul 54: 640-645.
- Lenzi M, Palmieri R, Porrello S (2003) Restoration of the eutrophic Orbetello lagoon (Tyrrhenian Sea, Italy): Water quality management. Mar Pollut Bull 46: 1540-1548.
- Aminti PL, Cappietti L, Tecchi MG, Venturini A (2003) Hydrodynamics of lagoons, a case study: The Orbetello lagoon. MEDCOAST03 - Sixth International Conference on the Mediterranean Coastal Environment, Florence, Italy.
- Franchi E, Ferrara G, Birardi F, Volterrani M, Mocenni C (2009) Qualitàglobaledeiprodottiittici di acquacoltura. Valutazionedeirefluiittici in relazionedegliobiettivi di qualitàdellanuovadirettivaeuropea in materia di tuteladelleacque e le relazioni con la qualità del prodotto finale. 248 pp.
- APAT, IRSA-CNR (2003) MetodiAnalitici per le Acque. ISBN 88-448-0083-7, 1: 1153.
- Aspila KI, Agemiam H, Chau ASY(1976) A semiautomatic method for the determination of inorganic, organic and total phosphate in sediments. Analyst 101: 187-197.
- Loh PS (2005) An Assessment of the Contribution of Terrestrial Organic Matter to Total Organic Matter in Sediments in Scottish Sea Lochs. UHI Millenium Institute 350.
- Underwood AJ (1997) Experiments in ecology. Their logical design and interpretation using analysis of variance. Cambridge University Press, Cambridge, UK 12: 410-411.
- Atkinson MJ, Smith SV (1983) C:N:P ratios of benthic marine plants. Limnology and Oceanography 28: 568-574.
- Wheeler PA, Bjornsater BR (1992) Seasonal fluctuations in tissue nitrogen, phosphorus, and n:p for five macroalgal species common to the Pacific Northwest coast. Journal of Phycology 28: 1-6.
- Viaroli P, Bartoli M, Giordani G, Naldi M, Orfanidis S, et al. (2008) Community shifts, alternative stable states, biogeochemical controls and feedbacks in eutrophic coastal lagoons: A brief overview. Aquatic Conservation 18: 105-117.
- Handy RD, Poxton MG (1993) Nitrogen pollution in mariculture: Toxicity and excretion of nitrogenous compounds by marine fish. In Reviews in Fish Biology and Fisheries 3: 205-241.
- Smit AJ (2002) Nitrogen Uptake by Gracilariagracilis (Rhodophyta): Adaptations to a Temporally Variable Nitrogen Environment. Botanica Marina 45: 196-209
- Giordani G, Zaldivar JM, Viaroli P (2009) Simple tools for assessing water quality and trophic status in transitional water ecosystems. Ecological Indicators 9: 82-991.
- Souchu P, Ximenes MC, Lauret M, Vaquer A, Dutriex E (2000) Mise a` jour d’indicateurs du niveau d’eutrophisation des milieux lagunaires méditerranéens, Ifremer-Creocean-Université´ Montpellier II, 412.
- EPA Environmental protection Agency (2005) National Coastal Condition, Report II. Report of US. Environmental Protection Agency, 286.
- http://www.xivcongresso.societaitalianaecologia.org/articles/.
- Lenzi M, Finoia MG, Persia E, Comandi S, Gargiulo V, et al. (2005) Biogeochemical effects of disturbance in shallow water sediment by macroalgae harvesting boats. Mar Pollut Bull 50: 512-519.
- Lenzi M, Salvaterra G, Gennaro P, Mercatali I, Persia E, et al. (2015) A new approach to macroalgal bloom control in eutrophic, shallow-water, coastal areas. J Environ Manage 150: 456-465.
- Revsbech NP, Sorensen J, Blackburn TH, Lomholt JP (1980) Distribution of oxygen in marine sediments measured with microelectrodes. Limnology and Oceanography 25: 403-411.
- Golterman HL (2001) Phosphate release from anoxic sediments or ‘what did Mortimer really write?’ Hydrobiologia 450: 99-106.
- Golterman HL (1995) The role of the ironhydroxide-phosphate-sulphide system in the phosphate exchange between sediments and overlying water. Hydrobiologia 297: 43-54.
- Lenzi M, Persiano M, Gennaro P, Rubegni F (2016) Wind mitigating action on effects of eutrophication in coastal eutrophic water bodies. International Journal of Marine Science and Ocean Technology 3: 14-20
- Lavery PS, McComb AJ (1991) The nutritional eco-physiology of Chaetomorpha linum and Ulva rigidain Peel Inlet, Western Australia. Botanica Marina 34: 251-260
Citation: Lenzi M, Gennaro P, Franchi E, Marsili L (2019) Assessment of Fish-Farms Wastewaters Synergistic Impact on a Mediterranean Non-Tidal Lagoon. J Aquac Fisheries 3: 021.
Copyright: © 2019 Mauro Lenzi, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

