
Effect of Photoperiod on Eggs Hatchability, Growth and Survivability of Hybrid Catfish (Heterobranchus bidorsalis x. Clarias gariepinus) Larvae
*Corresponding Author(s):
Isreal AdebayoDepartment Of Fisheries And Aquaculture Management, Ekiti State University, Ado Ekiti, Nigeria
Tel:+234 8066618186,
Email:isreal.adebayo@eksu.edu.ng
Abstract
This study was conducted to determine the effects of photoperiod on egg hatchability, growth and survivability of hybrid catfish (Heterobranchus bidorsalis x. Clarias gariepinus) larvae, using hormone-induced spawning method. Eggs were stripped from two sexually matured and healthy female Clarias gariepinus of average weight of 1 kg/each and fertilized with milt from two sexually matured male Heterobranchus bidorsalis of average weight of 2 kg/each. An average of five hundred (500) eggs were introduced into each ten aquaria tanks of size 70 cm × 45cm × 40 cm/tank, using a pre-determined spoonful estimation at five photoperiod regimes: (T1) 24L : 00D (Light : Darkness); (T2) 18L : 6D; (T3) 12L : 12D; (T4) 6L : 18D and (T5) 00L : 24D in two replicates. Aquaria tanks were arranged in a flow-through system at a flow rate of 1.5 L/min with aerators to maintain good water condition. Provision of light during the night for illumination of the aquaria tanks was kept constant at 1200 1 ×, using solar panel (Mono)/inverter (Microtex) light energy. Growth and survivability of the fish larvae were monitored for six weeks. They were fed with laboratory-cultured live feed (Daphnia) to achieve maximum feed utilization. Percentage hatchability of eggs and best growth performance of fish larvae were significantly (p<0.05) highest (92.5%, 91.2 ± 0.21 mg) respectively in T5 (00L : 24D), while percentage survivability of hatchlings was significantly (p<0.05) highest (94.4%) in T3 (12L : 12D). It was observed in this study that the highest hatchability of eggs and optimum growth performance of hatchlings were under complete darkness, with reduced survivability of fish, as a result of observed cannibalism. The fish were photophobic. To achieve a balance result in terms of hatchability of eggs, growth and survivability of fish fry, it is suggested that incubation and hatching of eggs should be done under complete darkness, while rearing of fry should be under equal light and darkness exposure.
Keywords
INTRODUCTION
Artificial propagation of catfishes is carried out in hatcheries using hormonal induction method. Farmers have found this approach to be cheap, practical and highly reliable than the wild source [4]. Adequate qualitative and quantitative sexual gametes are prerequisites for a successful artificial propagation exercise, hence the need to use sexually matured and healthy breeders [5]. Parent fish must also be kept in suitable environmental conditions with good feeding to ensure quality gametes. Hybridization between Clarias gariepinus and Heterobranchus bidorsalis combines the early maturing trait in Clarias gariepinuswith fast growth trait in Heterobranchus bidorsalis to produce hybrid (hetero-clarias). The emergence of hybrid catfish, with good potentials for fast growth and high acceptability led to rapid development of catfish farming in most part of Nigeria. Production of table size hybrid catfish was widely embraced by farmers as it gave better internal rate of return on investment due to rapid growth [2].
The success of artificial propagation of fish is determined by the number of fish larva produced at a given time [6]. Photoperiod, which is the daily cycle of light and darkness, plays a major role in the hatchability of eggs, growth and survivability of fish [7]. Photoperiod has been found to have a significant effect on the survivability of fish larva by exerting a definite influence on fish metabolism, maturation, behaviour and even coloration [8]. There must be a minimum and maximum amount of exposure to light or darkness that should be allowed for certain fish larva production [7]. Batty [9] found that long exposure to light caused a decrease in hatchability of eggs, less nutrients intake and consequently poor growth rate in some freshwater fishes. Also, light or darkness has direct impact on water temperature, turbidity, pH, ammonia which may either positively or negatively affects fish larva. To this regards, it is important to determine the influence of photoperiod on water parameters and the effect on fish larva in the hatchery. It was observed that fifty percent (50%) of fish fry mortality occurred at the larva stage due to fluctuations in photoperiod [10]. If fish do not receive the correct amount of light or darkness, they can be crippled and may not develop properly [8]. Light helps larva to determine the location of its food in water through photoreception [7]. The amount of light and darkness require is species specific and must be determined to achieve maximum larva production [6]. Therefore, the objective of this study was to know the effect of varying levels of light and darkness on eggs hatchability, growth and survivability of hybrid catfish (hetero-clarias) larva under laboratory conditions.
HYPOTHESIS
2. There will be no significant difference (p>0.05) in growth, survivability of hybrid catfish larvae under different photoperiod regimes.
3. Water quality will not affect the performance of the eggs hatchability, growth and survivability of the fish larvae.
MATERIALS AND METHODS
Procurement of brood stocks
Experimental set up
Hormonal induction of parent fish
Stripping, fertilization and incubation of eggs
Monitoring of water quality parameters
Determination of eggs hatchability
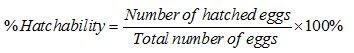
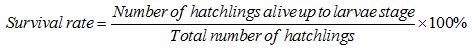
Survival rate was determined based on Jensen [12] method. The normal healthy larvae were estimated on percentage basis of dead and deformed hatchlings.
Determination of growth performance
Statistical analysis
RESULTS
|
Photoperiod Regimes (Treatments) |
Average Mean Number of Eggs Stripped and Incubated |
Average Mean Number of Hatched and Unhatched Eggs |
|
|||
|
Replicate (R1) |
Replicate (R2) |
Mean Total |
Hatched |
Unhatched |
% Hatchability |
|
|
T1 (24L : 00D) |
500 ± 0.02 |
500 ± 0.04 |
1000 ± 0.03 |
585 ± 0.01 |
415 ± 0.02 |
58.5 |
|
T2 (18L : 06D) |
500 ± 0.01 |
500 ± 0.02 |
1000 ± 0.02 |
680 ± 0.03 |
320 ± 0.01 |
68.0 |
|
T3 (12L : 12D) |
500 ± 0.00 |
500 ± 0.01 |
1000 ± 0.01 |
735 ± 0.02 |
265 ± 0.01 |
73.5 |
|
T4 (06L : 18D) |
500 ± 0.02 |
500 ± 0.00 |
1000 ± 0.02 |
860 ± 0.02 |
140 ± 0.02 |
86.0 |
|
T5 (00L : 24D) |
500 ± 0.03 |
500 ± 0.01 |
1000 ± 0.02 |
925 ± 0.01 |
75 ± 0.03 |
92.5 |
After twenty-one days of the hatching, the live, deformed and dead fish fry were estimated and the percentage mortality and survivability calculated as shown in table 2. The highest percentage mortalities were recorded in T4 and T5 (15.9%) respectively. It was observed that most of the dead fish fry were not seen may be as a result of cannibalism among the fish fry. Photoperiod regime of 12L : 12D (T3) had the highest percentage survivability (94.4%), while the least value was recorded in T5 (77.0%). The percentage of deformed fish fry was highest (13.0%) in T1 and least in T3 (4.9%)
|
Photoperiod Regimes (Treatments) |
Average Number of Hatched Eggs |
Average Number and Percentage of Live Fish Larvae |
Average Number and Percentage of Deformed Fish Larvae |
Average Number and Percentage of Dead Fish Larvae |
|
T1 (24L : 0D) |
585 ± 0.01 |
452 ± 0.01 (77.3%) |
76 ± 0.01 (13.0%) |
57 ± 0.02 (9.7%) |
|
T2 (18L : 6D) |
680 ± 0.03 |
530 ± 0.02 (77.9%) |
52 ± 0.02 (7.6%) |
98 ± 0.01 (14.4%) |
|
T3 (12L : 12D) |
735 ± 0.02 |
694 ± 0.03 (94.4%) |
36 ± 0.01 (4.9%) |
05 ± 0.00 (0.7%) |
|
T4 (6L : 18D) |
860 ± 0.02 |
676 ± 0.02 (78.6%) |
47 ± 0.02 (5.5%) |
137 ± 0.02 (15.9%) |
|
T5 (0L : 24D) |
925 ± 0.01 |
712 ± 0.01 (77.0%) |
66 ± 0.02 (7.1%) |
147 ± 0.02 (15.9%) |
The result of the physic-chemical parameters of the aquaria tanks is presented in table 3. Water temperature was within the range of 26.7-26.9°C throughout the experiment, while mean values obtained for Dissolve Oxgen (DO), pH and nitrate ranged between 6.3-6.5 mg/L, 7.5-7.9 and 0.22-0.23 mg/L respectively. There was no significant difference (p>0.05) in the values obtained for the water parameters among the treatments. Optimal water condition was maintained for the fish throughout the experiment to forestall poor results.
|
Water Parameters |
Photoperiod Regimes |
||||
|
T1 (24L : 0D) |
T2 (18L : 6D) |
T3 (12L : 12D) |
T4 (6L : 18D) |
T5 (0L : 24D) |
|
|
Dissolved oxygen (mg/L) |
6.5 ± 0.22 |
6.3 ± 0.21 |
6.5 ± 0.20 |
6.5 ± 0.22 |
6.3 ± 0.21 |
|
pH |
7.5 ± 0.20 |
7.6 ± 0.15 |
7.9 ± 0.21 |
7.7 ± 0.22 |
7.6 ± 0.20 |
|
Temperature (°C) |
26.7 ± 0.21 |
26.9 ± 0.17 |
26.7 ± 0.21 |
26.5 ± 0.21 |
26.7 ± 0.20 |
|
Nitrate (mg/L) |
0.22 ± 0.002 |
0.23 ± 0.002 |
0.22 ± 0.001 |
0.23 ± 0.003 |
0.23 ± 0.001 |
The growth response of the fish larvae under the five different photoperiod regimes (T1-T5) in six weeks after hatching is presented in table 4. There were significant differences (p<0.05) in growth performance of hybrid (H. bidorsalis x. C. gariepinus) larvae. The mean weight gain value was highest in T5 (91.2 ± 0.21) mg, followed by T4 (86.0 ± 0.20) mg and least in T1 (59.0 ± 0.22) mg respectively. The instantaneous growth rate, expressed as Specific Growth Rate (SGR) was significantly highest (p<0.05) in T5 (9.16) and least in T1 (8.22).
|
Growth Parameters |
Photoperiod Regimes |
||||
|
|
T1 (24L : 0D) |
T2 (18L : 6D) |
T3 (12L : 12D) |
T4 (6L : 18D) |
T5 (0L : 24D) |
|
Initial mean weight (mg) |
1.5 ± 0.01 |
1.5 ± 0.01 |
1.5 ± 0.01 |
1.5 ± 0.01 |
1.5 ± 0.01 |
|
Final mean weight (mg) |
60.5c ± 0.20 |
63.6c ± 0.22 |
76.8b ± 0.20 |
87.5b ± 0.20 |
92.7a ± 0.20 |
|
Average weight gain (mg) |
59.0c ± 0.22 |
62.1c ± 0.20 |
75.3b ± 0.22 |
86.0a ± 0.20 |
91.2a ± 0.21 |
|
SGR |
8.22c |
8.33c |
8.75 b |
9.06b |
9.16a |
DISCUSSION
CONCLUSION
REFERENCES
- Adewumi AA, Olaleye VF (2011) Catfish culture in Nigeria: Progress, prospects and problems. African Journal of Agricultural Research 6: 1281-1285.
- Adeogun OA, Ayinla OA, Ajana AM, Ajao EA (1999) Economic impact assessment of hybrid catfish (hetero-clarias) in nigeria. Technical report of national agricultural research project (NARP). NIOMR, Lagos, Nigeria. Pg no: 17.
- Olaleye VF (2005) A review of reproduction and gamete management in the african catfish Clarias gariepinus (Burchell). Ife Journal of Science 7: 63-70.
- Oresegun A, Oguntade OR, Ayinla OA (2007) A review of catfish culture in Nigeria. Nig J Fisheries 4: 27-52.
- Phelps RP (2010) Recent advances in fish hatchery management. R Bras Zootec 39: 95-101.
- Zheng W (2009) Review, culture of catfish in China. Aquaculture 75: 35-44.
- Pena R, Dumas S, Saldivar-Lucio R, Garcia G, Trasvina A, et al. (2004) The effect of light intensity on first feeding of the Spotted sand bass paralabrax maculatofasciatus (Steindachner) larvae. Aquaculture Research 35: 345-349.
- Puvanendran V, Brown J (2002) Foraging, growth and survival of atlantic cod larvae in different light intensities and photoperiods. Aquaculture 214: 131-151.
- Batty R (1987) Effect of light intensity on activity and food-searching of larval herring, Clupea harengus: A laboratory study. Marine biology 94: 323-327.
- Puvanendran V, Brown JA (1998) Effect of light intensity on the forging and growth of atlantic cod larva: Inter-population differences? Mar Ecol Prog Ser 167: 207-214.
- Aluko PO, Ali MH (2001) Production of eight types of fast growing in-tergeneric hybrids from four clariid species. Journal of Aquaculture in the Tropics 16: 139-147.
- Jensen AL (1996) Berverton and holt life history invariants results from optimal trade-off of reproduction and survival. Canadian Journal of Fisheries and Aquatic Sciences 53: 820-822.
- Olvera-Novoa MA, Campros GS, Sabido GM, Martinez-Palacious CA (1990) The use of alfalfa leaf protein concentrates as a protein source in diets of tilapia (Oreochromis mossambicus). Aquaculture 90: 291-302.
- Duncan DB (1955) Multiple ranges and multiple t-tests. Biometrics 1: 1-42.
- Luiz GG, Haluko M, Joao BKF (2012) Effect of photoperiod on reproduction of siamese fighting fish (Betta splendens). R Bras Zootec 41: 821-826.
- Watanabe WO, Woolridge CA, Daniels HV (2006) Progress toward year-round spawning of southern flounder brood stock by manipulation of photoperiod and temperature. Journal of the World Aquaculture Society 37: 256-272.
- Okwiri B (2015) Effect of tank background colour on the hatchability of O. Niloticus eggs and survival of fry. International Journal of Fisheries and Aquatic Studies 2: 81-86.
- Wasiu AO, Ofelia GO (2014) Embryonic and larval developmental stages of African catfish Heterobranchus bidorsalis (Geoffroy Saint Hilaire, 1809) (Teleostei, Clariidae). Springerplus 3: 677.
- Downing G, Litvak MK (2000) The effect of photoperiod, tank colour and light intensity on growth of larval haddock. Aquaculture International 7: 369-382.
- Bromage N, Porter M, Randall C (2001) The environmental regulation of maturation in farmed finfish with special reference to the role of photoperiod and melatonin. Aquaculture.
- Appelbaum S, Kamler E (2000) Survival, growth, metabolism and behaviour of Clarias gariepinus(Burchell) early stages under different light conditions. Aquacultural Engineering 22: 269-287.
- El-Sayed AFM, Kawanna M (2007) Effects of photoperiod on growth and spawning efficiency of Nile tilapia (Oreochromis niloticus L.) brood stock in a recycling system. Aquaculture Research 38: 1242-1247.
- Boeuf G, Le Bail PY (1999) Does light have an influence on fish growth? Aquaculture 177: 129-152.
- Adewolu MA, Adeniji CA, Adejobi AB (2008) Feed utilization, growth and survival of Clarias gariepinus (Burchell 1822) fingerlings cultured under different photoperiods. Aquaculture 283: 64-67.
- Kiyono M, Hirano R (1981) Effects of light on feeding and growth of black porgy mylio macrocephalus(Basilewsky), post larvae and juveniles. Cons Int Explo Mer 178: 334-336.
- Olaniyi W, Omitogun O (2013) Stages in the early and larval development of the african catfishClarias gariepinus (Teleostei, Clariidae). Zygote 22: 314-330.
Citation: Adebayo I (2018) Effect of Photoperiod on Eggs Hatchability, Growth and Survivability of Hybrid Catfish (Heterobranchus bidorsalis x. Clarias gariepinus) Larvae. J Aquac Fisheries 2: 007.
Copyright: © 2018 Isreal Adebayo, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

