
Growth Performance, Feed Utilization and Survival of Clarias gariepinus Post-larvae Fed with a Dietary Supplementation of Zingiber officinale-Allium sativum Mixture
*Corresponding Author(s):
Paulin NyadjeuDepartment Of Aquaculture, Institute Of Fisheries And Aquatic Sciences Of Yabassi, University Of Douala, Douala, Cameroon
Tel:+237 690872377,
Email:epauka@yahoo.fr
Abstract
Freshwater fish larviculture is the most sensitive step in aquaculture. It is characterized by a very high mortality rate, generally due to the mismatch between the feed used and the nutritional requirements of the larvae or fry. The present study aimed to assess the effects of dietary supplementation of the combination of garlic (Allium sativum) and ginger (Zingiber officinale) powders on growth and survival of Clarias gariepinus post-larvae reared in pond system. Thus, 1200 post-larvae were randomly divided in triplicate into four treatments. In treatment 1, post-larvae were fed with the control diet while in treatments 2 to 4, fry were fed on diet containing a mixture of equal quantity of ginger and garlic to give the final proportions of 0.5%, 1% and 1.5%, respectively for treatments 2, 3 and 4. Feeding trials were carried out for 63 days during which fry were fed daily at the rate of 10% of their body weight. Main water parameters were registered weekly. Fish sampling was done every 21 days during which fry per treatment were counted and measurements taken on a representative sample. The results obtained indicate that using the mixture of ginger and garlic as feed additive significantly improve growth performance, feed utilization and survival rate of C.gariepinus fry. With the highest rate of supplementation of 1.5%, the weight gain (7.63±0.13 g), body length growth rate (16.83±0.37% cm/d), specific growth rate (9.49±0.06% g/d), feed conversion ratio (1.29±0.17) and survival rate (84±5.13%) were significantly different compared to control by 51.77%, 24.60% 12.12% and 41.36%, respectively for the weight gain (3.68±0.12 g), body length growth rate (12.69±1.77% cm/d), specific growth rate (8.34±0.14% g/d), feed conversion ratio (2.20±0.04) and survival rate (63±6.56%). This study indicate that combination of ginger and garlic as feed additive can be very helpful to improve larvae and fry production.
Keywords
Clarias gariepinus; Garlic; Ginger; Growth; Post-larvae; Survival
INTRODUCTION
Aquaculture has been introduced in sub-Saharan Africa, particularly in Cameroon since the 1940s, on the one hand to minimize the pressure exerted by fishing communities on wild fish stocks and on the other hand to ensure permanent availability of fish for a steadily increasing population. Despite this, aquaculture production remains very low to cope with increased market demand of fish, resulting in massive imports to meet domestic demand. Local importers must therefore invest about 100 billion francs in local currency (170 million U.S dollars) each year. This represents huge financial losses and therefore an imbalance in the country's trade balance [1]. To remedy this situation, local production of available fish species should be strengthened to meet the strong market demand for fish and fish products. Among the endogenous species with high yield potential in Cameroon, there is Clarias gariepinus, which is a popular choice for local aquaculture; certainly because of its fast growth rate, omnivorous eating habits, the ability to feed on natural and complementary feeds, disease resistance, tolerance to low levels of oxygen and to overpopulation as well as pH fluctuations [2,3]. However, one of the main obstacles to the emergence of aquaculture production is lack of availability of fish seeds both quantitatively and qualitatively, as well as high quality, low-cost feeds [4-6]. Seeds production especially rearing of larvae and fry are the most sensitive steps in aquaculture production. Success of this stage is highly determined by larval as well as fry feeding and nutrition. In rural areas, Clarias gariepinus seeds production is generally carry out in rudimentary hatcheries by using the hypophyzation technic. Three to four days after hatching, the post-larvae are immediately transferred to the pond where they feed on natural feeds consists of a wide diversity of phytoplankton species (diatoms, flagellates) and zooplankton organisms (copepods, cladocerans, rotifers). However, dependence on natural feeds alone leads to a long period of larval rearing with negative consequences on survival. One of the alternative is to complete natural diet in pond with the formulated feeds containing feed additives that can enhance survival and induce faster growth of fish post-larvae as well as fry to meet the growing demand of fingerlings by grow-out fish farmers and subsequently of food fish in the market. There is a large number of feed additives commonly used to enhance growth performance in fish, among which some of antibiotics and hormones. Because they may induce harmful side effects, the use of antibiotics as feed additives to promote fish growth in aquaculture industry has been censored by government policies and consumer rights defenders owing to development of microorganism resistance and their potential deleterious effects on human health [7]. As the global trend is to return to nature, the World Health Organization encourages the use of medicinal herbs and plant products to replace or minimize the utilization of chemicals [8]. The use of natural products like medicinal plants as feed additives, may be highly beneficial to reinforce potency of feed utilization and animal productive performance [9]. Medicinal plants have different activities such as antistress, growth promotion, appetite stimulation, immunostimulation, aphrodisiac and antimicrobial properties due to the bioactive components such as alkaloids, flavanoids pigments, phenolics, terpenoids, steroids and essential oils [10]. Since many of their derivatives are known as growth stimulants agents, studies on the nutritional and pharmacological properties of these compounds have gained popularity amongst physiologists and aquaculture nutritionists in recent decades. Ginger (the powdered rhizomes of the herb Zingiber of?cinale) and Garlic (Allium sativum) are spices, in addition to contributing taste and aroma to foods, they contain bioactive compounds that are of considerable use from the point of view of food science and technology [11].
Ginger (Zingiber officinale) is an herbaceous perennial plant belonging to the family Zingiberaceae [12]. It is generally used for it safe herbal medicine, and also as spice widely used as food ingredient in many types of cuisine [13]. Ginger has been reported to be rich in mineral elements (magnesium, potassium, phosphorus, calcium and zinc), vitamins (retinol, cholecalciferol, ascorbic acid, thiamine, riboflavin, niacin, folic acid, pantothenic acid and pyridoxine) and phytochemicals such as gingerol, shogaols, zingerone, alkaloids, flavonoids, polyphenols, saponin and steroids [14-17] which have a high antioxidant activity [18]. Phamacological studies reported that ginger is anti-platelet, anti-bacterial, anti-fungal, anti-viral, anti-worm and anti-inflammatory, with effects on gastrointestinal and cardiovascular systems. It also possess anti-lipidemic, anti-hyperglycemic and anti-tumor properties and is known to be effective as an immuno-modulatory agent in human as well as animals, including fish [19,20]. Supplementing ginger in fish diets have being reported to enhance growth and survival [20-22].
Garlic (Allium sativum), a perennial bulb-forming plant belonging to the family Liliaceae, is native to Central Asia. It is commonly used in many cultures as a seasoning or spice as well as a remedy in treating many ailments such as heart diseases, headache, bites worms and tumors. Previous studies have revealed that it possess antibacterial, antiviral, antifungal activity [23], antioxidant, potent cardiovascular benefits, antihyperlipidemic, antidote (for heavy metal poisoning) immunomodulation, and significant heptoprotective role in detoxification [24-26]. It contains a variety of bioactive organosulfur compounds such as flavonoids, alkaloids, saponins, allicin (responsible for the distinctive odor) diallyl-disulfide, diallyl- trisulfide [27] and also vitamin A, C and F. Garlic has many beneficial effects on man and livestock; it has been proven to improve the health of fish [28]. Many studies have reported its beneficial effects on growth, digestibility feed intake, feed efficiency and carcass traits [29-32].
Despite the many trials of garlic and ginger on growth performance in different fish species, in our knowledge, trials on the combination of the two phyto-additve have not yet been carried out especially to promote growth in larval stage. Therefore, the present study was undertaken to investigate the effects of dietary supplementation of the combination of garlic (Allium sativum) and ginger (Zingiber officinale) on growth performance of Clarias gariepinus post-larvae and their survival rates.
MATERIALS AND METHODS
Study area
The study was carried out in the technical facilities of a private farm named “Common Initiative Group-Aquaculture Integrated of West (CIG-AIW)” in the West Region of Cameroon. The farm has the essential of the aquaculture installations such as earthen ponds, concrete tanks and plastic tanks, working space, a functional hatchery and constant water supply for induced breeding of this fish species.
Experimental fish and housing
The experimental fish larvae were obtained by artificial reproduction using the hypophyzation technic. Briefly, ripe, sexually mature female and male catfish were obtained from one of the brood stock ponds of the CIG-AIW’s farm. The females were injected with the pituitary gland extract and then kept in separate tanks with well aerated water at 24°C. The females were stripped after 18 hrs and the obtained eggs fertilized using the milt from the male. The fertilized eggs were incubated in the aerated incubation tanks for a period of 24 hrs at 28°C. After hatching, the larvae were transferred to the hatchling tanks until complete absorption of the yolk sac. On the fourth day after hatching, a total of 1200 post-larvae were randomly distributed in twelve hapas (0.5×0.5×1 m, L: W: H) made up of mosquito net cloth. These hapas were suspended in an earthen pond of 200 m2 as described by Nyadjeu et al, [33]. The pond water was previously fertilized with the chicken manure at the rate of 0.5 kg per m2. Before the start of the experience, the fry were allowed for acclimation for five days during which, they fed on natural feeds made up of both zooplanktons and phytoplanktons as the starter feed so as to obtain uniform size. At the end of acclimatization period, ten post-larvae were collected randomly from each hapa and weighed using a sensitive electronic weighing balance (Model pocket scale ) on which a zero weight wet filter paper was placed. This was repeated five times, and the average of the results of the five sets were taken as the initial weight of the individual larvae in the hapa. Five sets of five post-larvae each was measured using a transparent millimeter calibrated ruler and a magnifying hand lens. The average lengths of each of the sets were taken and the mean of the various set was taken as initial length of each post-larva in the hapa. Post-larvae or fry in hapas were grouped in triplicate into four treatments and fed four experimental diets containing different concentrations of the mixture Z.officinale-A.sativum powders as listed below.
• Treatment 1 (T0): 0% officinale-0% A.sativum
• Treatment 2 (ZO-AS: 0.5%): 0.25% officinale-0.25% A.sativum
• Treatment 3 (ZO-AS: 1%): 0.5% officinale-0.5% A.sativum
• Treatment 4 (ZO-AS: 1.5%): 0.75% officinale-0.75% A.sativum
Collection and processing of the feed additives
Fresh ginger and fresh garlic bulbs were purchased from a local market. After removing the dry skin of both fresh garlic and ginger, they were cut into small pieces and then dried in the shade. The dried ginger and garlic were then crushed finely using a household grinder and sieved using a household sieve to obtain 500 g of each of the powders which were kept in dry containers until formulation and preparation of experimental diets.
Formulation and preparation of experimental diets
Four isonitrogenous diets were formulated using Pearson square method to contain 40% crude protein. Ginger (Z.officinale (ZO)) and garlic (A.sativum (AS)) in powdered form were added to the formulated fish diets at 0% (T0) as control diet, 0.25% each one (ZO-AS: 0.5%), 0.5% each one (ZO-AS: 1.0%) and 0.75% each one (ZO-AS: 1.5%). Feed ingredients used for the experimental diet include: Ginger, garlic, fish meal, soybean cake, Peanut cake, maize meal, wheat bran, Methionine, Lysine, premix, vitamin C, bone meal and palm oil (Table 1). The dried and grinded ingredients of each diet were weighed and mixed thoroughly in a bowl for 10 min, palm oil and warm water were then added slowly along to the mixture and mixed manually for 20 others minutes to achieve a proper consistency. The resulting mixture was pelletized (2 mm) using an electric pelleting machine. The wet pellets were sun-dried, packaged into airtight containers and stored at room temperature to be crumbled before being used as a growth and finishing diet. Ten grams (10 g) of the Formulated diet samples were analyzed following the procedures of A.O.A.C [34]. Moisture was analyzed by drying the sample in an air convection oven at 105°C overnight. Crude protein was analyzed by the Kjeldahl method after acid digestion (% crude protein = % nitrogen × 6.25) while crude lipid was determined by extraction with petroleum ether using the Soxhlet method. The ash content in the diet was analyzed by combustion of samples in a muffle furnace at 550°C for 12 h (Table 1).
|
Ingredients |
Control |
ZO-AS: 0.5% |
ZO-AS: 1.0% |
ZO-AS: 1.5% |
|
Ginger |
// |
0.25 |
0.5 |
0.75 |
|
Garlic |
// |
0.25 |
0.5 |
0.75 |
|
Fish Meal |
37 |
37 |
37 |
37 |
|
Soybean cake |
17.5 |
17.3 |
17.3 |
17.3 |
|
Peanut cake |
17.5 |
17.2 |
17.2 |
17.2 |
|
Maize meal |
11.5 |
11.5 |
11.5 |
11.3 |
|
Wheat bran |
11.5 |
11.5 |
11.5 |
11.2 |
|
Methionine |
0.75 |
0.75 |
0.5 |
0.5 |
|
Lysine |
0.75 |
0.75 |
0.5 |
0.5 |
|
Premix 10% |
1 |
1 |
1 |
1 |
|
Vitamin C |
0.5 |
0.5 |
0.5 |
0.5 |
|
Bone meal |
1 |
1 |
1 |
1 |
|
Palm oil |
1 |
1 |
1 |
1 |
|
Proximate Composition (%) |
||||
|
Protein |
40.49 |
41.46 |
42.11 |
42.44 |
|
Lipid |
9 |
8.1 |
8.02 |
8.3 |
|
Ash |
9.3 |
9.5 |
9.4 |
8.8 |
|
Moisture |
7 |
8 |
8 |
8 |
|
Dry matter |
93 |
92 |
92 |
92 |
|
Energy (kcal/100g) |
269.12 |
267.26 |
269.66 |
273.62 |
Table 1: Formulations and proximate composition of experimental diets (g/100 g dry weight).
Experimental design
At the end of the acclimation period, the 1,200 post-larvae (weight: 19.32±1.06 mg and length: 7.98±0.9 mm) previously randomly divided into three replicates of 100 post-larvae each in 12 hapas, and fed with natural feeds from the fertilization of the pond, were subjected to four feeding treatments. In treatment 1 or control (T0), the post-larvae were fed with a normal feed, while in treatments 2 (ZO-AS: 0.5%), 3 (ZO-AS: 1.0%) and 4 (ZO-AS: 1.5%), they were fed with a normal feed supplemented with a mixture of equal quantity of ginger and garlic powders to obtain final proportions of 0.5%, 1% and 1.5%; respectively for treatments 2, 3 and 4. During the first 21 days of the experiment, fry were hand-fed four times a day (09:30, 12:30, 15:30 and 18:30 hours) follow by a frequency of three meals a day (10:30, 14:30 and 18:30 hours) for the rest of the trial period. Fry were fed by smearing the crumbled feed on the surface of the water in the hapa follow by a brief observation. Fry were fed at a rate of 10% of their body weight per day for 63 consecutive days. Every 21 days, the total population of each hapa was transferred to a beaker containing pre-weighed water to determine the biomass, then counted to determine the survival rate. After that, the individual weight and length were recorded on a sample of 20 fry per replicate. The fish fry were starved for 12 hours before sampling and 12 hours after sampling to limit the stress and mortality associated with handling.
Water quality management
Physico-chemical parameters of the water in each hapa such as temperature (T°C) was measured using a Maximum-minimum thermometer; transparency, using a Secchi disk, Dissolved Oxygen (D.O), using JBL Test Kits, pH, nitrate (NO3-) and nitrite (NO2-), using Test strips (JBL Easy Test 6 in 1) (Table 2). Water physico-chemical parameters were recorded weekly before feeding.
|
Parameters |
Rearing period (days) |
|
|||
|
0 |
21 |
42 |
63 |
Optimum values [35,36] |
|
|
T°C (°C) |
23.5±0.1 |
23.3±0.08 |
23.1±0.20 |
23.6±0.3 |
8-35 |
|
D.O (mg/l) |
5.3±0.22 |
5.5±0.11 |
5.5±0.01 |
5.3±0.06 |
>5 |
|
pH |
6,4±0.21 |
7,2±0.25 |
7,4±0.05 |
7,2±0.15 |
6.5-8.5 |
|
Transparency (cm) |
28.1±1.2 |
28.2±1.6 |
29.6±1.4 |
29.4±0.7 |
15-40 |
|
NO2- (mg.l-1) |
0.2±0.02 |
0.1±0.01 |
0,2±0.01 |
0,2±0.02 |
<0.3 |
|
NO3- (mg.l-1) |
15±0.02 |
10±0.03 |
15±0.02 |
15±0.02 |
<25 |
Table 2: Water quality parameters (Mean±SD) during 70 days of the experimental period.
Growth parameters, survival and feed utilization
Growth performances, survival and feed nutrients utilization were assessed for each treatment by determination of Weight Gain (WG), daily body length rate (Gr), Speci?c Growth Rate (SGR), Condition factor (K), Feed Intake (FI), Feed Conversion Ratio (FCR), Protein Ef?ciency Ratio (PER), Survival Rate (SR). Calculations were carried out using the following formulae:

STATISTICAL ANALYSIS
The data obtained were tested for normality and homogeneity of variances and later compared in the feeding trials (treatments) using one-way Analysis of Variance (ANOVA), followed by Tukey’s multiple comparison test. Data are presented as mean±Standard Deviation (SD). The analyses were performed using GraphPad Prism version 6.0. Treatment effects were considered significant at p<0.05; n=3 replicates of 30 fry each.
RESULTS
Ration palatability and growth parameters
Daily observation without measure of five days old larvae during the feeding revealed that they readily accepted all feeds. However, though acceptance for the experimental feeds, characterized by high agitation of the larvae during feeding was largely better in comparison to the control, it was independently to the level of phyto-additives inclusion.
The growth parameters of C.gariepinus fry in terms of weight gain, daily body length rate and specific growth rate are presented in figures 1-3. The cumulative weight gain curve (Figure 1), shows increase in weight gain depending on the harvest period and the concentration of the phyto-additive mixture in the feed. Fry fed with the highest level of dietary supplementation of mix ginger-garlic, show a significant (p<0.05) increase in weight gain from the 21st day of the feeding trial until the end of the experimental period with significant increases of 51.17%, 50.26% and 25.52% compared to control and treatments 2 and 3 respectively.

Figure 1: Mean weight gain of Clarias gariepinus fry fed different diets for 63 days.
Means on the same rearing period carrying the same superscript were not significantly different at p?0.05.
The results of the daily body length rate of Clarias gariepinus fry, as illustrated in figure 2, show a significant increase according to concentration of ginger-garlic mixture in the diet. On the 63rd day of the experiment, the body length rate of fry in treatment 3 (15.15±2.31%/day) is significantly (p?0.05) high by 16.24% and 12.80% compared to control (12.69±1.17%/day) and treatment 2 (13.21±2.03%/day) respectively. with the highest dietary supplementation level of ginger-garlic mixture (treatment 4 or ZO-AS: 1.5%), C.gariepinus fry present the greatest daily body length rate of 16.84±2.15%/day, corresponding to significantly (p?0.05) increases of 24.64% and 21.55% compared to control (12.69±1.17%/day) and treatment 2 (13.21±2.03%/day) respectively.
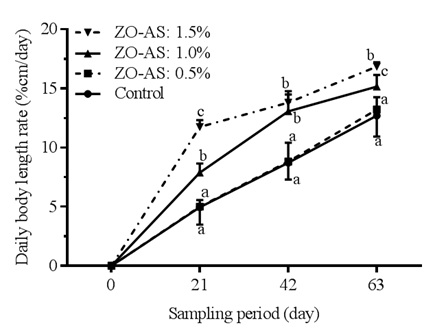
Figure 2: Daily body length rate of Clarias gariepinus fry fed different diets for 63 days.
Means on the same rearing period carrying the same superscript were not significantly different at p?0.05.
The effect of dietary supplementation of ginger-garlic mixture on specific growth rate is presented in figure 3. it should be noted that the highest significant values observed during all sampling periods are obtained in C.gariepinus fry fed the experimental feed in treatment 4 (ZO-AS: 1.5%), followed by non-significant values in the fry of treatment 3 (ZO-AS: 1.0%) compared to fish fry of treatments 1 and 2.
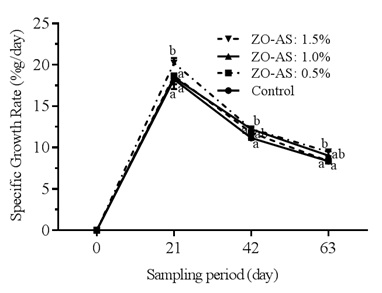
Figure 3: Specific growth rate of Clarias gariepinus fry fed different diets for 63 days.
Means on the same rearing period carrying the same superscript were not significantly different at p?0.05.
Feed efficiency
Table 3 represents feed nutrients utilization by C.gariepinus fry, fed locally made feed containing varying amounts of the ginger-garlic mixture as feed additives over a 63 days period. It appears from the table below that Feed Conversion (FCR) and protein Efficiency (FER) ratios are significantly (p<0.05) affected by the level of ginger-garlic mixture inclusion in the experimental feeds. FCR (1.29±0.17) as well as PER (1.39±0.05) is the most efficient in fish fed with diet ZO-AS: 1.5 %, followed by fish fed with diet ZO-AS: 1.0%; with the values of FER (1.71±0.09) and PER (1.18±0.01) significantly (p<0.05) different compared to control and treatment 2 (ZO-AS: 0.5%). No significant difference (p>0.05) have being noted as far as the Kondition factor (K) is concerned within different treatments.
|
Parameter |
Control |
ZO-AS: 0.5 |
ZO-AS: 1.0 |
ZO-AS: 1.5 |
|
Ni |
300 |
300 |
300 |
300 |
|
Nf |
188 |
217 |
223 |
251 |
|
Wi (g) |
0.02±00 |
0.02±00 |
0.02±00 |
0.02±00 |
|
Wf (g) |
3.70±0.12 |
3.77±0.10 |
5.67±0.17 |
7.63±0.12 |
|
Li (cm) |
0.80±0.09 |
0.80±0.09 |
0.80±0.09 |
0.80±0.09 |
|
Lf (cm) |
7.10±0.08 |
7.35±0.18 |
8.32±0.26 |
9.17±0.09 |
|
FI (g/fish) |
8.05±0.37a |
8.06±0.32a |
9.67±0.72a |
9.83±1.44a |
|
PI (g/fish) |
3.24±0.07a |
3.83±0.03b |
4.79±0.13c |
5.51±0.45c |
|
FCR |
2.20±0.04a |
2.15±0.06a |
1.71±0.09b |
1.29±0.17b |
|
PER |
1.13±0.06a |
0.98±0.03b |
1.18±0.01ac |
1.39±0.05d |
|
K |
1.03±0.02a |
0.95±0.06a |
0.99±0.06a |
0.99±0.04a |
Table 3: Feed utilization of Clarias gariepinus fry at the end of the feeding trial.
Values are mean±standard deviation of three replicates of 100 fish each. Mean within the row with different superscripts are significantly different at p
Survival rate
Cumulative survival rate of Clarias gariepinus fry, fed on different diets over a period of 63 days is represented in figure 4. It appears clearly that survival rate decrease according to the experimental period, but increase significantly depending to the concentration of ginger-garlic mixture as additive in the feed. except during the first sampling period where survival rate was significantly high in all experimental treatments compared to control, during the following two sampling periods, the highest significant increase in survival rate is obtained in treatment 4 (ZO-AS: 1.5%) compared to the rest of treatments, followed by treatments 3 and 2 respectively.
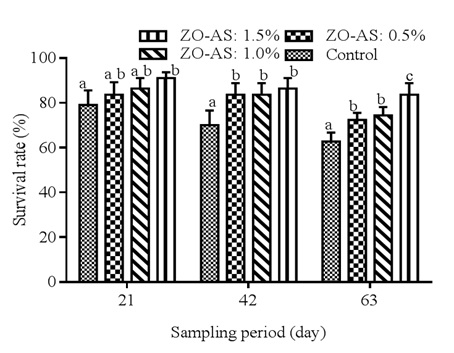
Figure 4: Cumulative survival rate of Clarias gariepinus fry fed on different diets over a period of 63 days.
Values are mean±SD of three replicates. Means on the same sampling period carrying the same superscript were not significantly different at p<0.05.
DISCUSSION
One of the major problems in the larviculture of freshwater fish such as the African catfish is the inadequate quality of feeds used associated with a low resistance to diseases, which leads to high mortality and reduced growth. Although chemicals such as antibiotics and growth hormones are used by some fish farmers to improve fish health and growth, these practices are largely discouraged because of the potential side effects of these chemicals on the fish produced and even the end consumer. That is one of the reason why fish physiologists and aquaculture nutritionists are more and more active in the search of alternative sources such as medicinal plants, fruits, spices or plant by-products that are ecologically friendly, with little or none side effect on final product and its consumer. These alternative sources of natural products susceptible to improve feed taste or palatability as well as fish health and growth are extensively tested as additives in the manufacture of feeds for livestock. Moreover, the evaluation of their effects in the diet of fish in general and that of post-larvae in particular constitute a relatively new area of research in aquaculture with promising results [37-39].
In the present study, fry fed purely on dry formulated diet without plant additive (control) recorded the lowest growth performance and survivability from the first fish sampling until the last. While the best performance was exhibited by fry fed on diet supplemented with the highest concentration of ginger-garlic mixture powder. Throughout the feeding period, some behavioral observations were noticed in different treatments. Fry fed the control diet were visually smaller and less active in the hapas compared with those fed formulated feed enriched with the mixture of ginger and garlic like food additive. It was also observed that enriched diets with the mixture of the two phyto-additive seemed to be more attractable to the post larvae than control diet; since in addition to the fact that fry in the experimental hapas were very agitated during feeding than those of the control treatment, feed consumption in these experimental hapas were also higher compared to the control. This is an indication that the combination of these two spices as feed additive in the diets of C.gariepinus fry would have improved the feed taste leading to increase in appetite stimulation level. The above observations are in line with the findings of some research studies that plant products used as feed additives play an important role as the feed taste enhancer agent or palatability and also like appetite stimulation agent. Addition of plant products in fish fry diet also intervene in health reinforcement and in enhancement of nutrients deliverance through better digestion and absorption, which invariably influences their behavioral tendencies and survivability [40-42].
Results on the growth indicate that there was a general increase in growth performance in C.gariepinus fry fed with different rate of dietary supplementation with the mixture of ginger and garlic. However, the highest growth response was observed in fish fry fed with the highest level of plant additive combination. This indicate that the use of ginger and garlic combination as phyto-additive in the diet of C.gariepinus fry could have improved nutrient utilization, which was reflected in enhancing weight gain, body growth length rate, specific growth rate, feed conversion ratio and protein efficiency ratio. These results are in agreement with many previous studies which reported that ginger powder or garlic powder when used as additive in fish diet, in concentrations of 0.5% to 3% improve growth, feed utilization and health in fry, fingerlings or juveniles of C.gariepinus as well as other fish species [31,43-47]. Previous findings have suggested that the growth promoting effects of ginger root and garlic bulb are most attributed to their richness in bioactive materials than to their nutritional value. Indeed, according to Lebda et al. [48] and Abayomi et al. [27], ginger is enriched in phenolic compounds such as gingerols, shogaols, volatile oils, flavonoids and phenolic ketone derivatives. Ginger root through its bioactive materials stimulate the appetite, the secretion of pancreatic enzymes and the bile from the liver, and also help to balance the intestinal bacteria; all these make fast feed digestion [49-51]. Moreover, ginger root also contains high levels of proteolytic enzymes and lipolytic plants which leads to improved digestion of dietary protein and lipid [52]. Concerning garlic powder, previous studies have suggested that supplementing garlic bulb in fish feed could improve growth due to its high content of bioactive components such as allin and allicin [53-54]. Allicin is known to be the most abundant compound representing almost 70% of all present thiosulfinates [55]. According to Khalil et al. [53], allicin contained in garlic improves the performance of the intestinal flora, thus improving digestion. This consequently enhances the feed utilization, resulting in improved growth. given the involvement of ginger and garlic in the digestion mechanism, as mentioned above, it is clear that the improvement in fry growth observed in the experimental groups compared to the control could be partially attributed to the combined effects of the bioactive compounds present in each phyto-additive which would have acted in synergy to increase the digestibility of macromolecules such as proteins and lipids which are part of the major constituents of the feed. The probable improvement in digestibility induced by the synergistic effects of the bioactive molecules of the plant additives could be justified by the low value of the feed conversion ratio obtained in C.gariepinus fry fed on the diet containing the highest level of the mixture of the two phyto-additives. In addition to the possible involvement of bioactive molecules of phyto-additives in the digestion mechanism of experimental fry, other studies have indicated that the beneficial effects of both ginger and garlic on the physiological and health status of fish when they are used as feed additive are largely due to their antioxydant properties [8,43]. According to Kim et al. (2007) [56] and Lebda et al. [48], ginger is considered a potent antioxidant substance which prevents free radicals generation and lipid peroxidation due to its considerable contain in phenolic compounds. The most important active molecules of ginger such as gingerdiol, zingibrene, zingerone, gingerols and shogaols are known to possess anti-oxidant activities [57]. In addition to that, it has been also reported that garlic can improve the antioxidant status of rainbow trout [58], mirror carp [59] and common carp [60], by decreasing the concentration of thiobarbutic acids in serum and tissues. The powerful antioxidant activity of garlic is expressed by scavenging Reactive Oxygen Species (ROS) and inhibiting lipid peroxidation [61]. Metwally in 2009 [62], while evaluating the effects of garlic (Allium sativum) on some antioxidant activities in tilapia nilotica indicated that S-allyl cysteine sulfoxide, isolated from garlic is an antioxidant molecule that control lipid peroxidation. Thus, in addition to the fact that the results of the present discovery on the growth of C.gariepinus fry were partially justified by the probable synergistic effects of the bioactive components of ginger and garlic in improving appetite and of the digestion mechanism. The improvement of the fry growth in experimental treatments would also be partially attributed to the synergistic antioxydant effects of the same bioactive components contained in phyto-additives through their capacity to scavenge reactive oxygen species and to inhibit lipid peroxidation. By this probable mode of action, it is obvious that the supplementation of the ginger and garlic mixture in the feed of the post larvae of C.gariepinus would have strengthened their state of health via the strengthening of their immunological status and the maintenance of a state more stable physiological.
Water quality is the most important limiting factor in larviculture as its quality directly affects fry’s behavior, feeding and survival. Any changes in the fry’s environment constitute a source of stress and the higher and faster the changes, the greater the stress. So the maintenance of the physico-chemical parameters within the acceptable limits are very essential for getting good production [36]. The average values of water quality parameters such as temperature, transparency, pH, dissolved oxygen, nitrite and nitrate recorded throughout the study were considered as suitable for good growth, the maintenance of stable physiological conditions as well as improving survival of C.gariepinus fry. These values are within the acceptable one [35-36] and could partially justify the relatively low mortality observed throughout the study, in particular in the experimental group compared to the control group. Freshwater species can be fed formulated diets as early as mouth opening [63]. It have being demonstrated the presence of acid protease, amylase and lipase specific activities at an early stage of striped catfish larvae after the appearance of the mouth even though the activity of these enzymes remain relatively low [64]. The concentration is therefore insufficient to digest efficiently compound feeds and, thus, the latter can be well ingested but larvae might die with guts full of feed. According to Sales [65], larvae fed on artificial feed have a 2.5 times higher chance to die because of various factors like incapacity to carry out complete digestion of ingested feed, starvation due to poor feed acceptance and lastly infections due to the pollution of the rearing environment [66]. All the above mention make larviculture a very sensitive activity that require too much precision in the management of the water quality as well as the choice of feed type. It is known that obtaining feeds that satisfy the nutritional needs of larvae is difficult since mechanisms of digestion and absorption, as well as nutritional requirements, change during larval development [67]. Good feeds or appropriate post larvae feed have a low feed conversion ratio which is an indication of their high level of digestibility. They allow for more fish to be grown in a pond because they produce less waste polluting the water. Thus, with better water quality the carrying capacity of the pond is increased. Once more, the relatively high mortality of about 37% obtained in the control treatment compared to 16% obtained in treatment 4 might be largely due to feed quality than to water quality. Thus, the low mortalities observed in experimental treatments, particularly in treatment 4, can be justified by the presence of phyto-additives in feeds which would have improved the feeds utilization by the synergistic actions of their bioactive molecules to stimulate the appetite and also to strengthen the mechanisms of digestion and absorption as well as the maintenance of a stable physiological state.
CONCLUSION
The results of this study revealed that the use of Z.officinale root powder combined with A.sativum bulb powder as an additive in the feed of C.gariepinus fry is beneficial to improve growth performance, feed utilization and the survival of C.gariepinus fry. This could be attributed to the synergistic actions of the bioactive molecules of each of the phyto-additives as growth promoting agents by enhancing both the appetite and the digestion mechanism as well as the state of health in the fry. The highest inclusion level of 1.5% Z.officinale-A.sativum powder in combination in the feed of C.gariepinus fry is best considered because of its good feed conversion ratio, its high protein efficiency ratio, specific growth rate, weight gain, growth body length and survival rate. In order to better understand the results of this study, it would be necessary to evaluate the effect of the combination of these two food additives on the growth of post-larvae in hatcheries.
ACKNOWLEDGEMENT
The authors wish to thank Mr. Michel DIOGNI, head of the fish farm “GIC AIO in Batié” for the reception and the provision of technical equipment necessary for the realization of this research work.
REFERENCES
- Kouam J, Hishamunda N, Halwart M, Moehl J, Pouomogne V, et al. (2003) Strategic framework for sustainable aquaculture development in cameroon. FAO Technical Report, Cameroon.
- Olurin KB, Iwuchukwu PO, Oladapo O (2012) Larval rearing of african catfish, Clarias gariepinus fed decapsulated artemia, wild copepods or commercial starter diet. African Journal of Food Science and Technology 3: 182-185.
- Beingana A, Kwikiriza G, Bwanika G, Abaho I, Izaara AA (2016) Performance of african catfish Clarias gariepinus (Clarridae) fry fed on live rotifers (Brachionus calyciflorus), formulated diet and a mixture of rotifers and formulated diet. International Journal of Fisheries and Aquatic Studies 4: 11-15.
- FAO (2006) Regional review on aquaculture development 4. Sub-Saharan Africa - 2005. FAO, Rome, Italy. Pg no: 93.
- Mwanja WW, Akol A, Abubaker L, Mwanja M, Msuku BS, et al. (2006) Status and impact of rural aquaculture practice on Lake Victoria basin wetlands. African Journal of Ecology 45: 165-174.
- Nabulime M, Rutaisire J, Ssekaayi J, Smith D, Kabasa JD (2015) Development of fry diets for African catfish (gariepinus) larvae in Uganda. Journal of Fisheries and Aquatic Science 10: 45-53.
- Baruah K, Norouzitallab P, Debnath D, Pal AK, Sahu NP (2008) Organic acids as non-antibiotic nutraceuticals in fish and prawn feed. Aquacult. Health Internat 12: 4-6.
- Mahmoud R, Aziza A, Marghani B, Eltaysh R (2019) Influence of ginger and garlic supplementation on growth performance, whole body composition and oxidative stress in the muscles of Nile tilapia (o. niloticus). Adv Anim Vet Sci 7: 397-404.
- Levic J, Sinisa M, Djuragic O, Slavica S (2008) Herbs and organic acids as an alternative for antibiotic- growth-promoters. Archiva Zootechnica 11: 5-11.
- Bairwa MK, Jakhar JK, Satyanarayana Y, Reddy AD (2012) Animal and plant originated immunostimulants used in aquaculture. Journal of Natural Products Plant Resources 2: 397400.
- Yanishlieva NV, Marinova E, Pokorny J (2006) Natural antioxidants from herbs and spices. European J Lipid Sci Technol 108: 776-793.
- Weidner MS, Sigwart K (2000) The safety of a ginger extract in the rat. J Ethnopharmacol 73: 513-520.
- Chang Y, Lin C, Wu C, Chiang C, Lian J, et al. (2012) Dietary administration of zingerone to enhance growth, non-speci?c immune response, and resistance to vibrio alginolyticus in paci?c white shrimp (Litopenaeus vannamei) juveniles. Fish Shellfish Immunol 32:284-290
- Ghasemzadeh A, Jaafar HZE, Rahmat A, Wahab PEM, Halim MRA (2010) Effect of different light intensities on total phenolics and ?avonoids synthesis and anti-oxidant activities in young ginger varieties (Zingiber of?cinale Roscoe). Int J Mol Sci 11: 3885-3897.
- Ghasemzadeh A, Jaafar HZE, Rahmat A (2010) Synthesis of phenolics and flavonoids in ginger (Zingiber officinale Roscoe) and their effects on photosynthesis rate. Int J Mol Sci 11: 4539-4555.
- USDA (2014) Essential oils, oleoresins (solvent-free), and natural extractives (including distillates). Substance generally Recognised As Safe 182: 1820.
- Iheanacho S, Ogunji JO, Ogueji EO, Nwuba LA, Nnatuanya IO, et al. (2017) Comparative assessment of ampicillin antibiotic and ginger (Zingiber officinale) effects on growth, haematology and biochemical enzymes of clarias gariepinus Juvenile. J Pharmacog Phytochem 6: 761-767.
- Shirin PR, Prakash J (2010) Chemical composition and antioxidant properties of ginger root (Zingiber officinale). Journal of Medicinal Plant Research 4: 2674-2679.
- Amar MJSA, Amar EC, Faisan JrJP, Pakingking JrRV, Satoh S (2012) Dietary onion and ginger enhance growth, hemato-immunological responses, and disease resistance in brown-marbled grouper, Epinephelus fuscoguttatus. AACL Bioflux 5: 231-239.
- Talpur AD, Ikhwanuddin M, Abol Munafi AB (2013) Nutritional effects of ginger (Zingiber officinale Roscoe) on immune response of Asian sea bass, Lates calcarifer (Bloch) and disease resistance against Vibrio harveyi. Aquaculture 400-401: 46-52.
- Ogueji EO, Iheanacho SC, Dada AO, Yaji AJ, Ifejimalu A, et al. (2017) Effect of Roselle (Hibiscus sabdariffa) and ginger (Zingiber officinale) as feed additives, on growth and haematology of Clarias gariepinus African Journal of Biotechnology 16: 2242-2247.
- Ghadikolaei HA, Kamali A, Soltani M, Sharifian M (2017) Effects of zingiber officinale powder on growth parameters, survival rate and biochemical composition of body in juvenile common carp (Cyprinus carpio). Iranian Journal of Fisheries Sciences 16: 67-85.
- Salah MA, Ahmed SD, George J, Yasser AH, Mohamed FM (2008) Effect of garlic, black seed and Biogen as immunostimulants on the growth and survival of Nile tilapia, Oreochromis niloticus (Teleostei: Cichlidae), and their response to artificial infection with Pseudomonas fluorescens. African Journal of Aquatic Science 33: 63-68.
- Jegede T (2012) Effect of garlic (Allium sativum) on growth, nutrient utilization, resistance and survival of Tilapia zillii (Gervais 1852) Fingerlings. J Agric Sci 4: 269.
- Yeh YY, Liu L (2001) Cholesterol-lowering effect of garlic extracts and organosulfur compounds: Human and animal studies. J Nutr 131: 9895-9935.
- Harris JC, Cottrell SL, Plummer S, Lloyd D (2001) Antimicrobial properties of Allium sativum (garlic). Appl Microbiol Biotechnol 57: 282-286.
- Abayomi Y, Fagbuaro SS, Fajemilehin SOK (2018) Chemical composition, phytochemical and mineral profile of garlic (Allium sativum). Journal of Bioscience and Biotechnology Discovery 3: 105-109.
- Corzo Martinez M, Corzo N, Marvillamiel L (2007) Biological properties of onions and garlic. Trends in food science and technology 18: 609-625.
- Tatara MR, Sliwa E, Dudek K, Gawron A, Piersiak T, et al. (2008) Aged garlic extract and allicin improve performance and gastrointestinal tract development of piglets reared in artificial sow Ann Agric Environ Med 15: 63-69.
- Shalaby AM, Khattab YM, Rahman AMA (2006) Effects of garlic (Allium sativum) and chloramphenicol on growth performance, physiological parameters and survival of Nile Tilapia (Oreochromis niloticus). J Venom Anim Toxins incl Trop Dis 12: 172-201.
- Lee DH, Lim SR, Han JJ, Lee SW, Ra CS, et al. (2014) Effects of dietary garlic powder on growth, feed utilization and whole body composition changes in fingerling sterlet sturgeon, acipenser ruthenus. Asian Australas J Anim Sci 27: 1303-1310.
- Manoppo H, Magdalena EF, Kolopita, Malatunduh R (2016) Growth promoter effect of garlic (Allium sativum) on carp (Cyprinus carpio L). International Journal of PharmTech Research 9: 283-288.
- Nyadjeu P, Alim H, Djopnang DJ, Ayamba AE, Tabi Tomedi EM (2019) Substituting fish meal with pre-treated lima bean meal on growth, feed utilization and nutrient retention in Clarias gariepinus fingerlings reared in hapa-in-pond system. Global Journal of Science Frontier Research: Agriculture and Veterinary 19.
- Association of Official Analytical Chemist (1990) Official methods of analysis association of official analytical chemists, 15th Washington DC, USA. Pg no: 1298.
- Santhosh B, Singh NP (2007) Guidelines for water quality management for fish culture in Tripura. ICAR. Pg no: 1-10.
- Bhatnagar AN, Devi PO (2013) Water quality guidelines for the management of pond fish culture. International Journal of Environmental Science Pg no: 1-30.
- Citarasu T (2010) Herbal biomedicines: A new opportunity for aquaculture industry. Aquaculture International 18: 403-414.
- Coutteau P, van Halteren A, Ceulemans S (2011) Botanical extracts improve productivity of shrimp, Pangasius. Global Aquaculture Advocate 90-92.
- Caipang CMA, Mabuhay Omar J, Gonzales Plasus MM (2019) Plant and fruit waste products as phytogenic feed additives in aquaculture. AACL Bioflux 12: 261-268.
- Reverter M, Bontemps N, Lecchini D, Banaigs B, Sasal P (2014) Use of plant extracts in fish aquaculture as an alternative to chemotherapy: Current status and future perspectives. Aquaculture 433: 50-61.
- Mathe A (2015) Introduction: Utilization/signi?cance of medicinal and aromatic plants. In: Mathe A (eds.). Medicinal and aromatic plants of the world: Scientific production, commercial and utilization aspects, (1st edn). Springer, Dordrecht, Germany. Pg no: 1-14.
- Kuebutornye FKA, Abarike ED (2020) The contribution of medicinal plants to tilapia aquaculture: A review. Aquaculture International.
- Akbary P, Jafarbeigi YN, Sondakzehi A (2016) Effects of garlic (Allium sativum L) extract on growth, feed utilization and carcass composition in Mugil cephalus (Linnaeus, 1758) larvae. Iranian Journal of Fisheries Sciences 15: 552- 557.
- Jahanjoo V, Yahyavi M, Akrami R, Bahri AH (2017) Influence of adding garlic (Allium sativum), ginger (Zingiber officinale), thyme (thymus vulgaris) and their combination on the growth performance, haematoImmunological parameters and disease resistance to photobacterium damselae in sobaity sea bream (sparidentex hasta) fry. Turkish Journal of Fisheries and Aquatic Sciences 18: 633-645.
- Tiamiyu AM, Adedeji OB, Olatoye IO (2017) Growth Performance of the African catfish, Clarias gariepinus, fed varying inclusion levels of Allium sativum as feed additives. American Journal of Biotechnology and Bioinformatics 1: 1.
- Iheanacho SC, Ogbu M, Ude E, Ayotunde E, Ogueji E (2018) Growth, hematology and immuno-modulatory potential of ginger (Zingiber officinale) supplemented diets in Clarias gariepinus juvenile (Burchell, 1822). Aquaculture Studies 18: 41-49.
- Adegbesan SI, Obasa SO, Akintokun AK, Abdulraheem I (2019) Effects of dietary supplementation of Zingiber officinale root-powder on growth, nutrient utilization and intestinal microbes of african mud catfish (Clarias gariepinus) Fingerlings. J Aquac Fisheries 3: 016.
- Lebda MA, Taha NM, Korshom MA, Mandour AEA, ElMorshedy AM (2012) Biochemical effect of ginger on some blood and liver parameters in male New Zealand J Anim Feed Res 2: 197-202.
- Platel K, Srinivasan K (2004) Digestive stimulant action of spices: A myth or reality. Indian Journal of Medical Reaserch 119: 167-179.
- Reinbach HC, Martinussen T, Møller P (2010) Effects of hot spices on energy intake, appetite and sensory specific desires in humans. Food Quality and Preference 21: 655-661.
- Ibidunni AS, Olubodun OS, Ikililu A (2017) Metabolic activities and health indices of African catfish (Clarias gariepinus) fed varying levels of Zingiber officinale root. Journal of Applied Biology & Biotechnology 5: 021-028.
- Venkataramalingam K, Godwin CJ, Citarasu T (2007) Zingiber officinalis an herbal appetizer in the tiger shrimp Penaeus monodon (Fabricius) larviculture. Aquaculture Nutrition 13: 439-443.
- Khalil RH, Nadia BM, Soliman MK (2001) Effects of biogen and levamisol Hcl on the immune response of cultured oreochromis niloticus to aeromonas hydrophila vaccine. Beni-Suef Assiut Veterinary Medical Journal, Egypt XI, 2: 381-392.
- Lawal AR, Olayinka BU, Murtadha RA, Ayinla A, Etejere EO (2018) Comparative Analysis of Phytochemical and Proximate Composition of Allium sativum L. and Zingiber officinale Rosc. Nigerian Journal of Basic and Applied Science 26: 82-87.
- Han JL, Lawson Han G, Han P (1995) Spectrophotometric method for quantitative determination of allicin and total garlic thiosulfinates. Analytical Biochemistry 225: 157-160.
- Kim JK, Kim Y, Na KM, Surh YJ, Kim TY (2007) Gingerol prevents UVB-induced ROS production and COX-2 expression in vitro and in vivo. Free Radic Res 41: 603-614.
- Jambrenghi AC, Colonna MA, Giannico F, Favia R, Minuti F, et al. (2005) Dietary supplementation of garlic and rosemary: Effects on colour stability and lipid oxidation in lamb meat. Italian J Anim Sci 4: 366-368.
- Mohebbi A, Nematollahi A, Dorcheh EE, Asad FG (2012) Influence of dietary garlic (Allium sativum) on the antioxidative status of rainbow trout (Oncorhynchus mykiss). Aquacult Res 43: 1184-1193.
- Xu Qy, Tang L, Wan CA, Xu , Sun DJ (2010) Effects of garlic stem powder and oregano leaf powder on antioxidant capacity, non-specific immune performance and meat quality of the carp (Cyprinus specularis). Acta Agriculturae Boreali-Sinica 2: 133-139.
- Naeiji N, Shahsavani D, Baghshani H (2013) Effect of dietary garlic supplementation on lipid peroxidation and protein oxidation biomarkers of tissues as well as some serum biochemical parameters in common carp Cyprinus carpio. Fisheries Sci 79: 699-705.
- Carmia B (2001) Antioxidant health effects of aged garlic extract. J Nutr 131: 1010-1015.
- Metwally MAA (2009) Effects of garlic (Allium sativum) on some antioxidant activities in tilapia nilotica (Oreochromis niloticus). World J Fish Mar Sci 1: 56-64.
- Cahu CL, Infante JLZ (2001) Substitution of live food by formulated diets in marine fish larvae. Aquaculture 200: 161-180.
- Rangsin W, Areechon N, Yoonpundh R (2012) Digestive enzyme activities during larval development of striped cat?sh, Pangasianodon hypophthalmus (Sauvage, 1878). Kasetsart J Nat Sci 46: 217-228.
- Sales J (2011) First feeding of freshwater fish larvae with live feed versus compound diets: A meta-analysis. Aquacult Int 19: 1217-1228.
- Abdulraheem I, Otubusin SO, Agbebi OT, Olowofeso O, Alegbeleye WO, et al. (2012) The growth response of Clarias gariepinus hatchlings to different dry feeds. J. Agric. Sci 4: 75-80.
- Dabrowski K (1984) The feeding of fish larvae: Present state of the art and perspectives. Reprod Nutr Dev 24: 807-833.
Citation: Nyadjeu P, Ekemeni RGM, Tomedi MET (2020) Growth Performance, Feed Utilization and Survival of Clarias gariepinus Post-larvae Fed with a Dietary Supplementation of Zingiber officinale-Allium sativum Mixture. J Aquac Fisheries 4: 028.
Copyright: © 2020 Paulin Nyadjeu, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

