
Longitudinal Assessment of tau Phosphorylation in the Brainstem of P301L tau-transgenic pR5 Mice
*Corresponding Author(s):
Stefan HuggenbergerInstitute Of Anatomy And Clinical Morphology, Witten/Herdecke University, Alfred-Herrhausen-Straße 50, 58448 Witten, Germany, Department II Of Anatomy (Neuroanatomy), University Of Cologne, 50924 Cologne, Germany
Email:stefan.huggenberger@uni-wh.de
Abstract
The pR5 mouse displays tau-hyperphosphorylation in multiple brainstem nuclei. Here we show a massive tau-phosphorylation in several nuclei of adult pR5 mice (6 to 18 months) including the motor nucleus of the trigeminal nerve (5N) which, however, did not contain neurofibrillary tangles. Body weights of pR5 mice was found to be significantly lower than in littermates. Since the 5N innervates the masticatory muscles an impaired chewing function may lead to lesser body weight of pR5 mice. Body weight loss is well known to accompany Alzheimer’s disease in humans and, so far, one study indicated tau-hyperphosphorylation in the 5N of Alzheimer’s patients.
Keywords
Alzheimer’s disease; Human P301L tau protein; Trigeminal nuclei; Masticatory muscles; Body weight; Tau-hyperphosphorylation
Introduction
To date, studies of neurofibrillary disorders focused mainly on the telencephalon in both humans and animal models [1-4]. This is due to the fact that the cardinal symptoms of many human neurodegenerative diseases are related to memory and cognitive dysfunction [5]. Comparatively few data are available for the human [6-11] and animal brainstem [12-14]. This is surprising since it is known for disorders such as Alzheimer’s disease (AD) that brainstem-related symptoms like dysphagia [15] appear rather early in the course of the disease. Furthermore, weight loss in elderly patients has been described as a marker for mild cognitive impairment (MCI)[16], preclinical AD [17] and a decreased body mass-index (BMI) as associated with cerebral AD pathology [18,19]. Among the spectrum of AD symptoms, nutrition-related problems are of similar importance as cognitive disturbances [20].
In terms of disease development progression, the supratrochlear subunit of the dorsal raphe magnus nucleus is affected by neurofibrillary tangle (NFT) formation well before the transentorhinal stage [21]. In particular the finding of intraneuronal pretangle material in the locus caeruleus of children and young adults suggests that the pathologic process leading to tau pathology may begin in selected brainstem nuclei [21] and is then propagated to other brain areas [22, 23]. Accordingly, studies in early stages of AD found considerable involvement of brainstem nuclei (reviewed in [24]). Aside from that, Wai et al. [11] described tau-hyperphosphorylation in the motor nucleus of the trigeminal nerve of patients with AD. In line with these studies, studies from our laboratory had shown that in aged pR5 mice a number of brainstem nuclei show tau-hyperphosphorylation [13]. Based on this study [13] and the human findings described above we have chosen the motor nucleus of the trigeminal nerve (5N) for a longitudinal study (8, 16 and 19 months of age). This cholinergic nucleus, which innervates the mouse masticatory muscles [25], shows pronounced tau-hyperphosphorylation [13]. Here, we have examined the 5N in detail and selected body weight as a simple functional parameter. Moreover, the longitudinal development of tau-hyperphosphorylation has been assessed for the entire brainstem. The data were presented in parts as poster presentation at the 14th International Conference on Alzheimer’s and Parkinson’s Diseases [26].
Material And Methods
Male tau tg pR5 mice [27] and gender- and age-matched non-transgenic (non-tg) littermates were used for controlling body weight and health status at the age of 6 (n=11), 14 (n=10) and 17.5 (n=11) months. Out of these mice, coronal brainstem sections (5 µm) of 13 mice at the age of 8 (n=3 pR5 mice), 16 (n=3 pR5 mice) and 19 (n=3 pR5 mice) months were made. The animals were housed under a 12h light-dark cycle with food and water ad libitum. The P301L tau transgenic pR5 mouse with neuronal expression of the longest human tau (HT) isoform was generated on a C57BL/6 background. The tau filament formation was accomplished by the missense mutation P301L in exon 10, known from frontotemporal dementia and parkinsonism linked to chromosome 17 (FTDP-17). Experiments were approved by the governmental animal care and use office (Landesamt für Natur, Umwelt und Verbraucherschutz Nordrhein-Westfalen, Recklinghausen, Germany, Protocol No. 8.87-51.05.20.10.260). All animals were kept and treated in accordance with the German Animal Welfare Act.
For the perfusion-fixation mice were deeply anesthetized with tribromethanol (0.55mg/g b.w.; i.p.) and transcardially perfused with 0.1 M phosphate buffered saline (PBS; pH 7.4) for 3 min followed by 4% paraformaldehyde in PBS for 15 min. Brains were removed, postfixed in 4% paraformaldehyde in PBS overnight at 4°C, dehydrated and embedded in paraffin using a Shandon Citadel tissue processor (Thermo Shandon, Frankfurt am Main, Germany).
Subsequently, sections were stained with cresylviolet and immunostained using the antibodies described below. The following anti-tau antibodies were used: conformation and phosphorylation independent mouse monoclonal antibody HT7 (Thermo Scientific Pierce Protein Research Products, Rockford, USA, catalog # MN1000; diluted 1:400), recognizing residues 159–163 to detect human tau [28]; mouse monoclonal antibodies AT180 and AT8 (both from Thermo Scientific Pierce Protein Research Products, catalog # MN1040 and MN1020; diluted 1:2400 and 1:1000) against hyperphosphorylated epitopes Thr231/Ser235 [29] and Ser202/Thr205 [30].
For immunostaining of cholinergic neurons a goat polyclonal antibody (Millipore, Billerica, USA, catalog # AB144P; diluted 1:200) raised against ChAT (UniProt Number: P28329) was used. Biotinylated donkey anti-mouse IgG (diluted 1:400), goat anti-rabbit IgG (diluted 1:400 and 1:450) and donkey anti-goat IgG (diluted 1:450) secondary antibodies were purchased from Dianova (Hamburg, Germany).
Paraffin sections were counterstained with nuclear fast red (5 min) and than treated as described in detail by Morcinek et al. [13] and coverslipped with DPX mounting medium (Sigma-Aldrich). Negative controls included sections incubated without the primary antibody. In the 5N, the number of HT7-, AT8-, and AT180-immunoreactive (ir) neurons were quantified and normalized to the number of choline acetyltransferase (ChAT) - ir cells. To check for the presence of classical neurofibrillary tangles the Gallyas silver staining was performed [31]. Further details of staining procedures can be found in [32]. Immunostained structures were identified using the mouse brain atlas books of Hof et al. [33] and of Paxinos and Franklin [34] (Table 1).
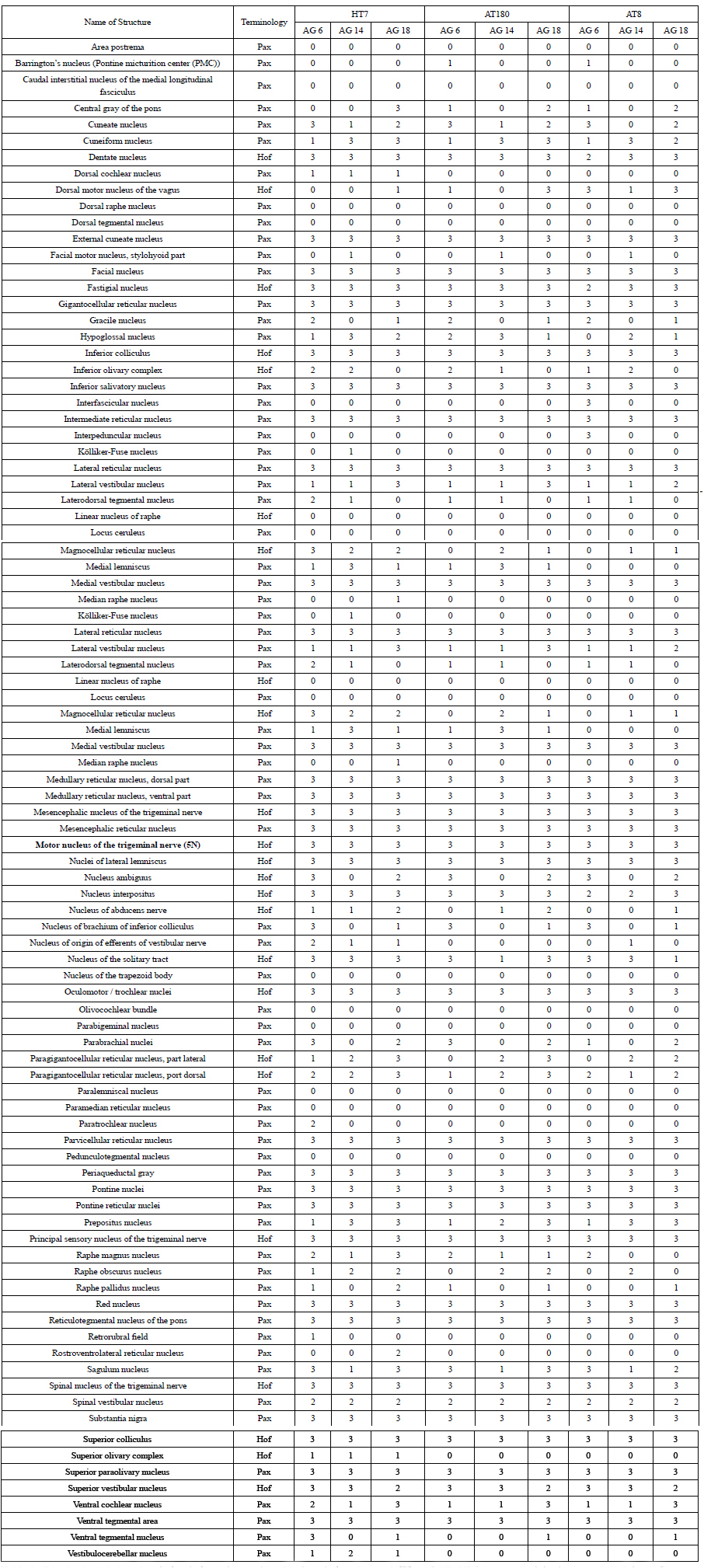 Table 1: Distribution of phosphorylation-independent anti-tau HT7- and phospho-tau (AT8- and AT180)-immunoreactivity in structures of the pR5 mouse brainstem. Values indicate the absolute number of brains in the different age groups (AG) of 8, 16, and 19 months, respectively, showing HT7-AT8- or AT180-immunoreactive neurons in the corresponding structure. Terminology after Paxinos and Franklin [34] (Pax) and Hof et al. [33] (Hof).
Table 1: Distribution of phosphorylation-independent anti-tau HT7- and phospho-tau (AT8- and AT180)-immunoreactivity in structures of the pR5 mouse brainstem. Values indicate the absolute number of brains in the different age groups (AG) of 8, 16, and 19 months, respectively, showing HT7-AT8- or AT180-immunoreactive neurons in the corresponding structure. Terminology after Paxinos and Franklin [34] (Pax) and Hof et al. [33] (Hof).
Digital images were obtained with an Olympus BX43 motorized microscope equipped with a DP25 digital microscope camera and cellSens Dimension - Imaging Software 2.3 (Olympus, Hamburg, Germany). Pictures were adjusted for equal brightness, color and contrast in Adobe Photoshop CS5 Extended 12.0 (Adobe Systems, San Jose, USA).
All statistical analyses utilized IBM SPSS Statistics (Version 21) or Microsoft Excel (2010). Statistical significance of independent means was assessed by means of Student’s t-test. Data are indicated as mean ± standard error of the mean (SEM) based on n, the number of independent experiments.
Results
Motor nucleus of the trigeminal nerve (5N)
The 5N neurons, innervating the masticatory muscles, presently showed the known characteristics of this motor nucleus, i.e. large motoneuron-type ChAT-ir perikarya with a clearly visible nucleolus (Figure 1). Numbers of ChAT-ir as well as of Nissl-stained neurons did not differ between age groups nor genotype.
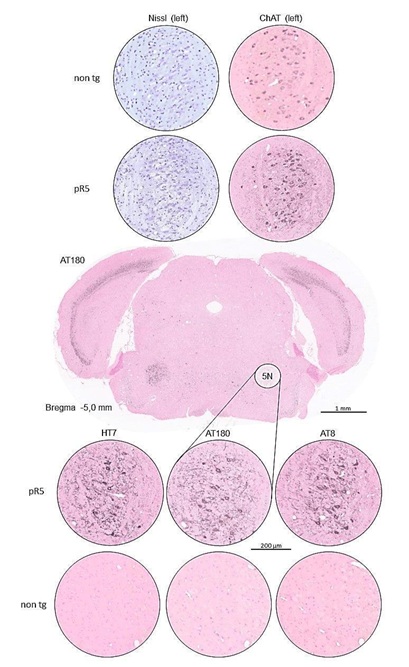 Figure 1: Coronal brainstem sections of a P301L tau transgenic pR5 mouse (19 months) at the level of the motor nucleus of the trigeminal nerve (5N). The survey section is immunostained for the AT180 phospho-tau antibody and counterstained with nuclear fast red. The inserts from the overview section and from consecutive sections of the same animal and of a non-tg mouse (non-tg littermate) show the 5N at a higher magnification and are stained with cresylviolet (Nissl) or immunostained with choline acetyltransferase antibody (ChAT), HT7, AT180, 14 AT8 and counterstained with nuclear fast red. Nissl- and ChAT-staining visualize the numerous large neuronal perikarya typical for spinal cord and brainstem motor neurons. There were no qualitative differences between wild type and pR5 neurons. While sections immunostained with HT7, AT180, and AT8 in the non-tg mouse are immunonegative, the pR5 mouse shows a massive infestation of the 5N with AT180-ir as well as with AT8-ir neurons.
Figure 1: Coronal brainstem sections of a P301L tau transgenic pR5 mouse (19 months) at the level of the motor nucleus of the trigeminal nerve (5N). The survey section is immunostained for the AT180 phospho-tau antibody and counterstained with nuclear fast red. The inserts from the overview section and from consecutive sections of the same animal and of a non-tg mouse (non-tg littermate) show the 5N at a higher magnification and are stained with cresylviolet (Nissl) or immunostained with choline acetyltransferase antibody (ChAT), HT7, AT180, 14 AT8 and counterstained with nuclear fast red. Nissl- and ChAT-staining visualize the numerous large neuronal perikarya typical for spinal cord and brainstem motor neurons. There were no qualitative differences between wild type and pR5 neurons. While sections immunostained with HT7, AT180, and AT8 in the non-tg mouse are immunonegative, the pR5 mouse shows a massive infestation of the 5N with AT180-ir as well as with AT8-ir neurons.
The 5N belongs to the group of brainstem nuclei which displayed HT7-, AT8-, and AT180-immunoreactvity (IR) in all animals of all three age groups (Table 1 and Figure 1). However, the Gallyas impregnation used to reveal NFTs failed in pR5 mice in the whole brainstem. Quantitative assessment of the number of AT8- and AT180-ir neurons in the 5N of older pR5 mice as compared to younger (6 months of age) pR5 mice showed a statistically significant increase with age (8 months vs. 16 months of age: p<0.01; 9 months vs. 19 months of age: p<0.05). No significant differences in the number of AT8- and AT180-ir neurons in the 5N were observed between 16- and 19-months-old pR5 mice. The data refer to the number of ir-neurons normalized by the number of ChAT-ir neurons (Figures 2A-2C). Interestingly, the body weight of pR5 mice of the age groups of 6, 14 and 18 months was significantly lower (p<0.001) than that of age-matched non-tg littermate controls (Figure 2D).
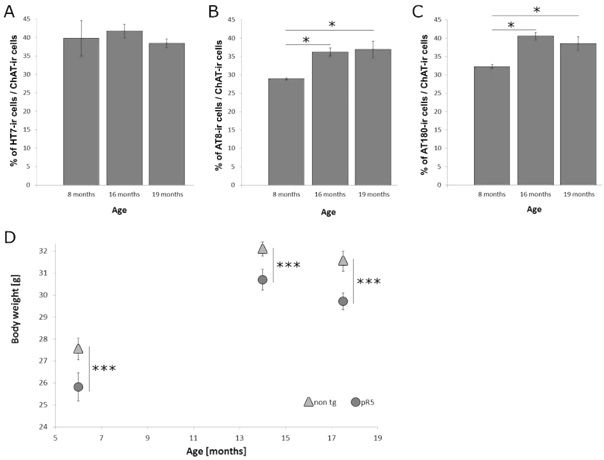 Figure 2: Time course of numbers of HT7-ir (A), hyper phosphorylated tau (B: AT8, C: AT180) positive neurons in the motor nucleus of the trigeminal nerve (5N) of male pR5 mice (age: 8, 16 and 19 months) normalized to the number of ChAT-ir cells (* p<0.05). The body weight of non-tg and pR5 mice (D) shows an age dependent increase of the body weight (6 to 14 months) with a slight decline to the senescent stage (18 months). Overall, however, there is a statistically highly significant (*** p<0.001) difference between the wildtype and the pR5 mice with each of the age classes.
Figure 2: Time course of numbers of HT7-ir (A), hyper phosphorylated tau (B: AT8, C: AT180) positive neurons in the motor nucleus of the trigeminal nerve (5N) of male pR5 mice (age: 8, 16 and 19 months) normalized to the number of ChAT-ir cells (* p<0.05). The body weight of non-tg and pR5 mice (D) shows an age dependent increase of the body weight (6 to 14 months) with a slight decline to the senescent stage (18 months). Overall, however, there is a statistically highly significant (*** p<0.001) difference between the wildtype and the pR5 mice with each of the age classes.
Other brainstem nuclei
An overview about the presence of neurofibrillary changes in the individual brainstem nuclei over time is given in Table 1. The triple-ir nuclei (HT7-, AT8- and AT180-IR in all animals of all three age groups) show that numerous brainstem nuclei were infested with tau-hyperphosphorylation already at the age of 6 months. However, a quantitative assessment of all these nuclei was beyond the scope of the present study.
Discussion
Our present study has shown that neurons of different brainstem nuclei display massive tau-hyperphosphorylation in pR5 mice from 8 months up to 19 months of age (Figure 1 and Table 1) while non-tg mice were devoid of immunoreactivity for AT8 and AT180. Concurrently, the pR5 mice showed a significantly lower body weight compared to non-tg littermates (Figure 2D). In general, these facts point to a relationship between the hyperphosphorylated tau (HFT) pathology and body weight due to the following reasons: weight loss in elderly humans has been suggested to be a marker for preclinical AD / mild cognitive impairment [16,17] and full-blown AD [18]. Wai et al. [11] have reported about HFT in the ‘trigeminal sensory/motor complex’ in human AD without providing well verifiable histological data. These pathological changes in AD [11] as well as those in pR5 mice (this study) strengthen the hypothesis that the loss of body weight is at least partly attributable to a dysfunction of 5N since this nucleus controls the main chewing musculature.
In contrast, Blautzik et al. [19] have developed the hypothesis that the observed weight loss in patients with mild cognitive impairment may be related to a leptin-mediated hypothalamic dysfunction. This dysfunction, however, is unlikely in pR5 mice since there is no apolipoprotein E4 promoted accumulation of cortical amyloid as suggested by Blautzik et al. [19].
Krox-20 is expressed in the 5N and in a number of other HFT-infested nuclei [35]. Krox-20 is a C2H2-type zinc-finger transcription factor playing an important role in rhombomere development [36]. As to the motor nuclei among the HFT nuclei it is of interest that Krox-20 null mutants point to a critical role of this transcription factor in the development of oral-motor and respiratory brainstem networks [35].
In AD, dysphagia is known to be an important problem [15]. As to the other chewing/swallowing-related nuclei [37] like the ambiguus nucleus, the dorsal motor nucleus of the vagus nerve and the hypoglossal nucleus, presently there were no congruent data for AT180- and AT8-IR. At least tau-hyperphosphorylation is present in these nuclei at certain time points in adult life of pR5 mice (Table 1).
The 5N is a well-known target in amyotrophic lateral sclerosis [38]. There are two naturally occurring murine models (progressive motor neuronopathy [pmn] and wobbler) and a transgenic mouse model with a human G93A mutation in the superoxide dismutase-1 (SOD1) gene [39,40]. Comparisons of neuron numbers in the respective end stage of all three models showed significant losses of neurons in 5N [40]. Magnetic resonance imaging revealed degeneration of the 5N in vivo in the superoxide dismutase 1 transgenic mouse [41]. Recently, the dynein heavy chain mutation Loa has been shown to induce a significant reduction of 5N neurons associated with changes in dendritic and ultrastructural morphology [42]. In terms of function, Lever et al. [43] have shown that the post disease SOD1-G93A transgenic mice showed significantly lower body weights and mastication rates and a degeneration of the 5N compared to non-tg littermates.
In summary, we could presently show that there is a correlation between the massive hyperphosphorylation of the tau protein in neurons of the pR5 mouse 5N and the body weight as compared to non-tg littermates. Since there were no statistically significant differences in the numbers of 5N neurons between transgenic and non-tg mice the weight differences cannot be attributed to a simple cell loss in the 5N. These findings are the basis for (1) the prospective evaluation of potentially involved transcription factors and (2) the electrophysiological characterization of 5N neurons and those of other triple-ir brainstem nuclei.
Acknowledgements
The authors are grateful to Sigrun Kuhlage, Kirsten Pilz, and Maja Dinekov for excellent technical assistance and for animal care, and Mascha Recks for help with digitalizing illustrations. This work was supported by grants from the Käthe-Hack and the Hochhaus foundation of the Faculty of Medicine (University of Cologne, Germany). The authors do not have conflicts of interest.
References
- Duyckaerts C, Delatour B, Potier MC (2009) Classification and basic pathology of Alzheimer disease. Acta Neuropathol 118: 5-36.
- LaFerla FM, Green KN (2012) Animal models of Alzheimer disease. Cold Spring Harb Perspect Med 2: 006320.
- Kent BA, Heath CJ, Kim CH, Ahrens R, Fraser PE, et al. (2018) Longitudinal evaluation of Tau-P301L transgenic mice reveals no cognitive impairments at 17 months of age. Brain Behav 8: 00896.
- Kumar K, Kumar A, Keegan RM, Deshmukh R (2018) Recent advances in the neurobiology and neuropharmacology of Alzheimer's disease. Biomed Pharmacother 98: 297-307.
- Draganski B, Lutti A, Kherif F (2013) Impact of brain aging and neurodegeneration on cognition: evidence from MRI. Curr Opin Neuro l26: 640-645.
- Parvizi J, Van Hoesen GW, Damasio A (2001) The selective vulnerability of brainstem nuclei to Alzheimer's disease. Neurol 49: 53-66.
- German DC, White CL, Sparkman DR (1987) Alzheimer's disease: neurofibrillary tangles in nuclei that project to the cerebral cortex. Neuroscience 21: 305-312.
- Lee JH, Ryan J, Andreescu C, Aizenstein H, Lim HK (2015) Brainstem morphological changes in Alzheimer's disease. Neuroreport 26: 411-415.
- Rub U, Del Tredici K, Schultz C, Thal DR, Braak E, et al. (2000) The evolution of Alzheimer's disease-related cytoskeletal pathology in the human raphe nuclei. Neuropathol Appl Neurobiol 26: 553-567.
- Rub U, Del Tredici K, Schultz C, Thal DR, Braak E, et al. (2001) The autonomic higher order processing nuclei of the lower brain stem are among the early targets of the Alzheimer's disease-related cytoskeletal pathology. Acta Neuropathol 101: 555-564.
- Wai MS, Liang Y, Shi C, Cho EY, Kung HF, et al. (2009) Co-localization of hyperphosphorylated tau and caspases in the brainstem of Alzheimer's disease patients. Biogerontology 10: 457-469.
- Dutschmann M, Menuet C, Stettner GM, Gestreau C, Borghgraef P, et al. (2010) Upper airway dysfunction of Tau-P301L mice correlates with tauopathy in midbrain and ponto-medullary brainstem nuclei. J Neurosci 30: 1810-1821.
- Morcinek K, Köhler C, Götz J, Schröder H (2013) Pattern of tau hyperphosphorylation and neurotransmitter markers in the brainstem of senescent tau filament forming transgenic mice. Brain Res 1497: 73-84.
- Overk CR, Kelley CM, Mufson EJ (2009) Brainstem Alzheimer's-like pathology in the triple transgenic mouse model of Alzheimer's disease. Neurobiol Dis 35: 415-425.
- Kalia M (2003) Dysphagia and aspiration pneumonia in patients with Alzheimer's disease. Metabolism 52: 36-38.
- Alhurani RE, Vassilaki M, Aakre JA, Mielke MM, Kremers WK, et al. (2016) Decline in Weight and Incident Mild Cognitive Impairment: Mayo Clinic Study of Aging. JAMA Neurol 73: 439-446.
- Jimenez A, Pegueroles J, Carmona-Iragui M, Vilaplana E, Montal V, et al. (2017) Weight loss in the healthy elderly might be a non-cognitive sign of preclinical Alzheimer's disease. Oncotarget 8: 104706-104716.
- Mathys J, Gholamrezaee M, Henry H, von Gunten A, Popp J (2017) Decreasing body mass index is associated with cerebrospinal fluid markers of Alzheimer's pathology in MCI and mild dementia. Exp Gerontol 100: 45-53.
- Blautzik J, Kotz S, Brendel M, Sauerbeck J, Vettermann F, et al. (2018) Relationship Between Body Mass Index, ApoE4 Status, and PET-Based Amyloid and Neurodegeneration Markers in Amyloid-Positive Subjects with Normal Cognition or Mild Cognitive Impairment. J Alzheimers Dis 65: 781-791.
- Pivi GAK, Bertolucci PHF, Schultz RR (2012) Nutrition in severe dementia. Curr Gerontol Geriatr Res 2012: 983056.
- Grinberg LT, Rub U, Ferretti RE, Nitrini R, Farfel JM, et al. (2009) The dorsal raphe nucleus shows phospho-tau neurofibrillary changes before the transentorhinal region in Alzheimer's disease. A precocious onset? Neuropathol Appl Neurobiol 35: 406-416.
- Clavaguera F, Hench J, Goedert M, Tolnay M (2015) Invited review: Prion-like transmission and spreading of tau pathology. Neuropathol Appl Neurobiol 41: 47-58.
- Ghosh A, Torraville SE, Mukherjee B, Walling SG, Martin GM, et al. (2019) An experimental model of Braak'spretangle proposal for the origin of Alzheimer's disease: the role of locus coeruleus in early symptom development. Alzheimers Res Ther 11: 59.
- Grinberg LT, Rueb U, Heinsen H (2011) Brainstem: Neglected locus in neurodegenerative diseases. Front Neurol 2: 42.
- Baverstock H, Jeffery NS, Cobb SN (2013) The morphology of the mouse masticatory musculature. J Anat 223: 46-60.
- Schröder H, Götz J, Huggenberger S, Müller-Thomsen L, Borgmann D, et al. (2019) in Alzheimer's & Parkinson's Diseases.
- Götz J, Chen F, van Dorpe J, Nitsch RM (2001) Formation of neurofibrillary tangles in P301l tau transgenic mice induced by Abeta 42 fibrils. Science 293: 1491-1495.
- Mercken M, Vandermeeren M, Lubke U, Six J, Boons J, et al. (1992) Monoclonal antibodies with selective specificity for Alzheimer Tau are directed against phosphatase-sensitive epitopes. Acta Neuropathol 84: 265-272.
- Goedert M, Jakes R, Crowther RA, Cohen P, Vanmechelen E, et al. (1994) Epitope mapping of monoclonal antibodies to the paired helical filaments of Alzheimer's disease: identification of phosphorylation sites in tau protein. Biochem J 301: 871-877.
- Goedert M, Jakes R, Vanmechelen E (1995) Monoclonal-Antibody At8 Recognizes Tau-Protein Phosphorylated at Both Serine-202 and Threonine-205. Neurosci Lett 189: 167-170.
- Gallyas F (1971) Silver Staining of Alzheimers Neurofibrillary Changes by Means of Physical Development. Acta Morphol Acad Sci Hung 19: 1-8.
- Paterno M (2016) in Faculty of Medicine University of Cologne.
- Hof PR, Young WG, Bloom FE, Belichenko PV, Celio MR (2000) Comparative cytoarchitectonic atlas of the C57BL/6 and 129/Sv mouse brains, Elsevier, Amsterdam.
- Paxinos G, Franklin K (2019) Paxinos and Franklin's the Mouse Brain in Sterotaxic Coordinates Elsevier (5th Edn), Amsterdam.
- De S, Shuler CF, Turman JE Jr (2003) The ontogeny of Krox-20 expression in brainstem and cerebellar neurons. J Chem Neuroanat 25: 213-226.
- De S, Nguyen AQ, Shuler CF, Turman JE Jr (2005) Mesencephalic trigeminal nucleus development is dependent on Krox-20 expression. Dev Neurosci 27: 49-58.
- Sang Q, Goyal RK (2000) Lower esophageal sphincter relaxation and activation of medullary neurons by subdiaphragmatic vagal stimulation in the mouse. Gastroenterology 119: 1600-1609.
- DePaul R, Abbs JH, Caligiuri M, Gracco VL, Brooks BR (1988) Hypoglossal, trigeminal, and facial motoneuron involvement in amyotrophic lateral sclerosis. Neurology 38: 281-283.
- Ferrucci M, Spalloni A, Bartalucci A, Cantafora E, Fulceri F, et al. (2010) A systematic study of brainstem motor nuclei in a mouse model of ALS, the effects of lithium. Neurobiol Dis 37: 370-383.
- Haenggeli C, Kato AC (2002) Differential vulnerability of cranial motoneurons in mouse models with motor neuron degeneration. Neurosci Lett 335: 39-43.
- Zang DW, Yang Q, Wang HX, Egan G, Lopes EC, et al. (2004) Magnetic resonance imaging reveals neuronal degeneration in the brainstem of the superoxide dismutase 1 transgenic mouse model of amyotrophic lateral sclerosis. Eur J Neurosci 20: 1745-1751.
- Wiggins LM, Kuta A, Stevens JC, Fisher EM, von Bartheld CS (2012) A novel phenotype for the dynein heavy chain mutation Loa: altered dendritic morphology, organelle density, and reduced numbers of trigeminal motoneurons. J Comp Neurol 520: 2757-2773.
- Lever TE, Gorsek A, Cox KT, O'Brien KF, Capra NF, et al. (2009) An animal model of oral dysphagia in amyotrophic lateral sclerosis. Dysphagia 24: 180-195.
Citation: Huggenberger S, Paterno M, Morcinek K, Müller-Thomsen1 L, Dawidowski B, et al. (2021) Longitudinal Assessment of tau Phosphorylation in the Brainstem of P301L tau-transgenic pR5 Mice. J Alzheimers Neurodegener Dis 7: 051.
Copyright: © 2021 Stefan Huggenberger, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

