
Petroleum Hydrocarbon Contents in Tropical Soils near Crude Oil Exploration and Processing Site in Niger Delta, Nigeria
*Corresponding Author(s):
Edem DennisDepartment Of Soil Science And Land Resources Management, University Of Uyo, Uyo, Nigeria
Tel:+234 8027031426,
Email:dennis.edem@gmail.com
Abstract
Petroleum Hydrocarbon contents in tropical soilshas been increasingly recognized as an important soil degradation factor in crude oil producing areas. Hydrocarbon can impact and contaminate the soil apart from playing nutrient inhibition role in the soil. The application of an effective and sensitive analytical method to determine soil contaminants is a crucial step in monitoring and remediation processes. In the present work, we optimized the analysis of Volatile Organic Compounds (VOC) commonly present in fuel: Quantitation of fluorobenzene and monoaromatic hydrocarbons such as Benzene, Toluene, Ethylbenzene and Xylene (BTEX). At impacted locations of Well-Head (WH), Group Gathering Facility (GGF), Group Processing Facility (GPF) and non-impacted sites served as the control. Headspace (HS) and Headspace-Solid Phase Microextraction (HS-SPME) were optimized in water samples, and validated for contaminated soils, using spiked soils. Contaminants were identified and quantified by Gas Chromatography coupled to Mass Spectrometry (GC/MS). Average BTEX (2.50mgkg-1) and TPH (4.0mgkg-1) were significantly higher in GGF and the concentration was below detection limit in GPF. Examination of regression lines of BTEX and TPH to internal standard on concentration using a least-squares fit demonstrated a linear relationship with correlation coefficients was consistently greater than 0.999 at the impacted sites.
Keywords
INTRODUCTION
Volatile Organic Compounds (VOCs), such as Benzene, Toluene, Ethylbenzene, Xylene and (BTEX), are important environmental contaminants because of their toxicity and widespread occurrence. They are present in aviation fuel and petrol (gasoline) and are widely used industrial solvents and raw materials [1]. Benzene, toluene and ethylbenzene are amongst the compounds designated ‘priority pollutants’ by the US EPA and action levels for BTEX [2], Soils become contaminated with BTEXC through spillage of industrial solvents, leakage of petrol from storage facilities (particularly underground holding tanks) and deposition from contaminated air [3]. Moreover, BTEX are used as solvents for many purposes and because of their relative solubility in water they can contaminate the soil and groundwater, and thus provoking environmental pollution [1].
In the recent past, their toxicity and carcinogenetic effects, in the case of benzene, have become a serious concern for human health. The admitted limits for benzene in drinking water are set at 5 and 1μgL–1 in United States and European Union, respectively [4]. After the accidental contamination of soil and water, the BTEX concentrations are generally several orders of magnitude higher. Several methods are used for BTEX determination and the choice among the various extractions, separation and detection techniques also depends on the matrix to be analyzed [5]. In water and soil the most commonly employed methods are based on solid-phase micro extraction (SPME) coupled to Gas Chromatography-Mass Spectrometry (GC-MS), or to a Gas Chromatography-Flame Ionization Detector (GC-FID). BTEX determination using gas chromatography is quite time consuming, and needs special instrumentation, and thus the development of alternative methods that could be used even on-site is highly required [2].
The aim of the present work was to quantify hydrocarbons such as Benzene, Toluene, Ethyl benzene and Xylene (BTEX) on soils at the focal point of different production sites of Qua Iboe Terminal crude oil exploration fields at Ibeno.
MATERIALS AND METHODS
Experimental site and soil
The temperature of the area like most tropical environment is generally high. The average annual maximum temperature is 34.8°C for the period of 10 years, while the average annual minimum temperature is 24.3°C for the same period [6]. February and March have the highest evaporation rate, and it is as high as 6.9mm. The least evapotranspiration rate (1.6mm) is recorded in June/July. This pattern is directly related to the pattern of rainfall/cloud cover and atmospheric temperature. Sunshine hour is also directly related to cloud cover. Daily average sunshine hours range from 7.5 hours in January to 14 hours in August. The relative humidity is relatively high throughout the year. It ranges from 60% in February to 90% in June. Thus, the highest values are recorded at the height of rainy season, while the lowest values occur during the dry months [6].
Soil Sampling
 Figure 1: Samplings near the exploration sites at QIT.
Figure 1: Samplings near the exploration sites at QIT.Analysis of soil samples
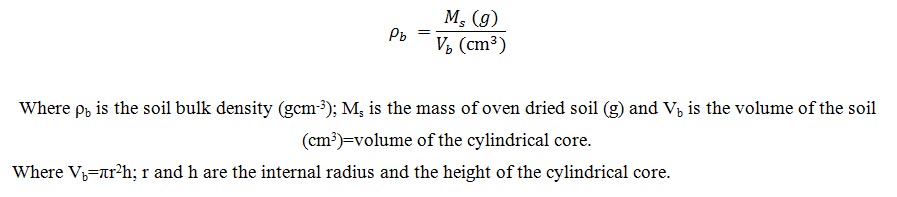
Total porosity: (TP) was determined from the relationship between the bulk density and the particle density as:


Moisture content: (MC) expressed on volumetric basis, was estimated as the different between Field Capacity (FC) obtained at 10 kPa (-100cm water) and Permanent Wilting Point (PWP) at 1500 kPa (-15,000cm water) using:
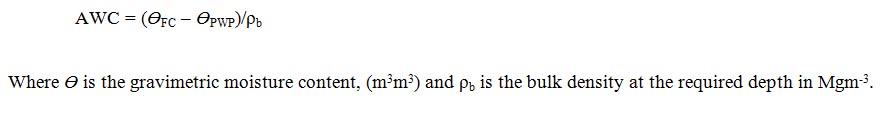
To determine the micro porosity of the aggregates, recall:
Total porosity and that the volume of a sphere:

Fifty gram (50g) of air-dry soil aggregates was weighed, after passing through 8 mm sieve. The initial masswas recorded as W1. The soil sample was thereafter placed on the uppermost (4.75mm) sieve with other nest of sieves: 2.0mm, 1.0mm, 0.25mm and 0.045mm placed below it in that order.The nest of sieves was immersed in water such that the soil at the top of 4.75mm sieve was wet by capillarity. The height of the nest of sieves was adjusted such that the soil sample on the sieves remains immersed in water on the upstroke of the dipping machine. The set of sieves was cycled through a column of water for 10 minutes (30 cycles per min, 4.0cm stroke length).
The soil retained on each sieve was washed into moisture can with distilled water. Each fraction of the retained soil was oven dried at 105oC to a constant mass W2. Water and 10 ml of calgon (sodium hexametaphosphate) (0.5% w/v) were added to the oven-dried soil for chemical dispersion and thereafter dispersed for 10 minutes using mechanical stirrer. The two dispersion processes were carried out to separate the sand particles from the soil aggregates. The sand particles were washed into the corresponding moisture can and then oven dried at 105oC to a constant mass W3.

Analysis of petroleum hydrocarbons from contaminated soils
Volatile Organic Compounds (VOCs) penetration was also measured from each borehole using a Photo Ionization Detector. Stand-type piezometers were installed at performed boreholes for groundwater level and quality monitoring.
Statistical analysis of experimental data
RESULTS AND DISCUSSION
Soil physical properties
Particle size distribution
|
Locations |
Sand |
Silt |
Clay |
Texture |
|
Surface Soil (0-15cm) |
||||
|
WH |
49.21c |
28.59a |
22.2b |
Loam |
|
GGF |
48.64c |
28.03a |
23.33b |
Loam |
|
GPF |
55.33b |
19.1b |
25.57a |
Sandy loam |
|
Control |
76.33a |
13.2c |
10.47c |
Sandy loam |
|
|
|
|
|
|
|
Sub Surface Soil (15-30cm) |
||||
|
WH |
69.5a |
13.83c |
16.67d |
Sandy loam |
|
GGF |
38.67c |
28.0a |
33.33b |
Clay loam |
|
GPF |
45.83b |
18.5b |
35.67a |
Loam |
|
Control |
68.17a |
12.83c |
19.02c |
Sandy loam |
Table 1: Particle size analysis for surface and subsurface soil at near exploration sites.
Note: WH is Well Head, GGF is Group Gathering Facility, and GPF is Group Processing Facility
Means followed by different letter is significantly different (?=0.05)
|
Stable Aggregates |
|
||||||
|
BD |
Ksat |
Porosity |
Moisture content |
Micro aggregates |
Macro aggregates |
MWD |
|
|
Locations |
Mgcm-3 |
×10 cmhr-1 |
m3m-3 |
m3m-3 |
% |
% |
mm |
|
Surface |
|||||||
|
WH |
0.41 |
1.0 |
0.845 |
0.63 |
14.8 |
1.53 |
1.24 |
|
GGF |
0.38 |
0.8 |
0.856 |
0.75 |
11.48 |
1.42 |
1.29 |
|
GPF |
0.78 |
1.4 |
0.705 |
0.57 |
11.53 |
1.86 |
1.3 |
|
Control |
1.43 |
3.7 |
0.460 |
0.42 |
13.56 |
1.12 |
1.87 |
|
Subsurface |
|||||||
|
WH |
0.46 |
1.4 |
0.826 |
0.66 |
12.15 |
3.69 |
0.95 |
|
GGF |
0.52 |
0.2 |
0.804 |
0.78 |
48.75 |
1.01 |
0.96 |
|
GPF |
0.84 |
1.2 |
0.683 |
0.59 |
14.35 |
2.98 |
1.18 |
|
Control |
1.67 |
2.6 |
0.370 |
0.48 |
14.7 |
2.59 |
2.09 |
Total porosity followed a reciprocal trend to soil bulk density, and there were significant differences among the sites (Table 2). At the surface soil, total porosity for the impacted sites was higher than the control. Surface porosity for the impacted sites was; 0.85, 0.86 and 0.71m3m-3 for WH, GGF, and GPF, respectively and control site was 0.46m3m-3. In the subsurface layer, porosity values were 0.83, 0.80, and 0.63m3m-3 respectively at the impacted site and 0.37m3m-3 in the control site. In comparison to total porosity, the soil total porosity for the control site was lower than oil impacted site.
The extraction of individual hydrocarbon
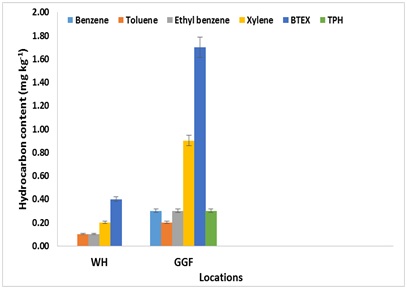 Figure 2: Individual BTEX and TPH concentrations on surface soil of polluted site at Ibeno.
Figure 2: Individual BTEX and TPH concentrations on surface soil of polluted site at Ibeno.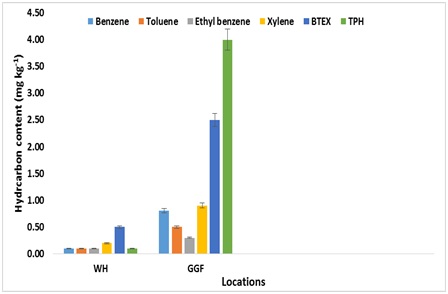 Figure 3: Individual BTEX and TPH concentrations on subsurface soil of polluted site at Ibeno.
Figure 3: Individual BTEX and TPH concentrations on subsurface soil of polluted site at Ibeno.Toluene, ethyl benzene, and xylene remain in solvent and quantitatively retaining the BTEX compound at WH and GGF and were below detection limit at GPF and the control sites. The lowest detectable individual concentration of total BTEX were benzene and toluene estimated as 0.3mgkg-1. These soil samples were further examined and found to contain mixed matrix including stable aggregates. The total BTEX was most detected in soils of GGF (depression) followed by WH. These results suggest that the probability of detecting spill petroleum compounds in an undulating topography site is greater at depressed location compared the crest.
Assessments of TPH and BTEX Linearity
SUMMARY AND CONCLUSION
Soil samples collected from the three impacted sites represented the pollution sites around Mobil Production Company at QIT. The contaminated soils collected from sites consisted of a range of soil textures, moisture contents and bulk densities. The soils included clay loam soils, sandy loam with traces of organic matter and fine sand. The TPH concentrations for the six contaminated soils ranged between 0.1-4.0mgkg-1. The moisture content for the surface soil samples ranged from 42% to 75% (w/w) with average moisture content of 59.3% (w/w) and 48% to 78% with average of 62.7% in the subsurface soil. When the total BTEX and TPH concentrations were examined among the soil samples, total BTEX and TPH were not detected at GGF area of the impacted site due to elevated topographic position that enhanceddownward flow of the petroleum compounds.
Since all of the BTEX compounds are toxic, the consumption of BTEX infected plant and water, especially, at WH and GGF, respectively may produce immediate health effects. These symptoms, ranging from mild to severe nature, may even lead to serious health conditions including brain damage and leukemia. Considering the high water table in the vicinity of Ibeno, groundwater contaminated with BTEX compounds is inevitable and it is difficult to remediate.
REFERENCES
- Saarela K, Tirkkonen T, Laine-Ylijoki J, Jurvelin J, Nieuwenhuijsen MJ, et al. (2003) VTT Technical Research Centre of Finland. Atmosphere Environment 37: 5563.
- Agency for Toxic Substances and Disease Registry (2007) Toxicological profile for benzene. Agency for Toxic Substances and Disease Registry, Atlanta, Georgia. Pg no: 1-438.
- Zhang Z, Pawliszyn J (1993) Analysis of organic compounds in environmental samples by headspace solid phase microextraction. J High Res Chromatography. 16: 689-692.
- http://eurlex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:1998:330:0032:0054:EN:PDF.
- http://www.epa.gov/safewater/contaminants/index.html#1.
- Mansuy L, Philp RP, Allen JA (1997) Nigerian Meteorological Agency (NIMET). Nigeria Climate Review Bulletin, Nigeria.
- Ogban PI, Edem ID (2005) Physical fertility of degraded acid sands in South-eastern Nigeria. Soil Science Society of Nigeria 25: 102-108.
- Grossman RB, Reinsch TG (2002) Bulk density and linear extensibility: Core method. Dane JH, Topp GC (Eds.), Methods of soil analysis. Part 4; Physical methods Soil Science Society of America, Inc. Madison. Wisconsin. Pp 208-228.
- Reynolds WD, Elrick DE, Young EG, Amoozegar A, Booltink HWG (2002) Saturated and field-saturated water flow parameters. Dane JH, Topp GC (Eds.), Methods of Soil Analysis. Part 4. Physical Methods. Soil Science Society of America, Inc., Madison, Wisconsin. Pp 797-878.
- Reynolds WD, Elrick DE (1990) Ponded infiltration from a single ring: I. analysis of steady flow. Science Society of America Journal 64: 478-484.
- Levy GJ, Levin J, Shainberg I (1994) Seal formation and interrill soil erosion. Soil Science Society of America 58: 203-209.
- Pavón JL, Martín SH, Pinto CG, Cordero BM (2009) Programmed temperature vaporizer based method for the sensitive determination of trihalomethanes and benzene, toluene, ethylbenzene and xylenes in soils. J Chromatogr A 1216: 6063-6070.
- Kelley CA, Hammer BT, Coffin RB (1997) Environ. Sci. Technol 31: 2469.
- Rosell M, Lacorte S, Barceló D (2006) Simultaneous determination of methyl tert-butyl ether, its degradation products and other gasoline additives in soil samples by closed-system purge-and-trap gas chromatography -mass spectrometry. J Chromatogr A 1132: 28-38.
- United States Environmental Protection Agency (USEPA) (2003) Volatile organic compounds in various sample matrices using equilibrium headspace analysis. Washington: US Environmental Protection Agency.
- Edwards DA, Andriot MD, Amoruso MA, Tummey AC, Bevan CJ, et al. (1997) Development of fraction specific Reference Doses (RfDs) and Reference Concentrations (RfCs) for Total Petroleum Hydrocarbons (TPH), Vol. 4, Amherst Scientific Publishers, Amherst, USA.
- Weisman WD (1998) Analysis of petroleum hydrocarbons in environmental media. Amherst Scientific Publishers, Amherst, Massachusetts, USA.
Citation: Udoinyang U, Edem D, Akabio JHU (2019) Petroleum Hydrocarbon Contents in Tropical Soils near Crude Oil Exploration and Processing Site in Niger Delta, Nigeria. J Environ Sci Curr Res 2: 007.
Copyright: © 2019 Uduak Udoinyang, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

