
Use of Intravitreal Ganciclovir as Monotherapy for Bilateral CMV Retinitis in a Tertiary Government Hospital
*Corresponding Author(s):
Ana Margarita A SarmientoDepartment Of Ophthalmology, Quirino Memorial Medical Center, Philippines
Email:margoasarmiento@gmail.com
Abstract
Introduction/Background: Cytomegalovirus (CMV) infection is an infection seen in both immunocompetent patients presenting as mononucleosis, and in immunocompromised patients presenting as disseminated disease. Since the rise of Human Immunodeficiency Virus (HIV) diseases, diagnosis of CMV infection also increased because coinfection of HIV and CMV is common. Cytomegalovirus retinitis is the most common ocular complication, developing in approximately half of the patients with HIV-CMV coinfection. It is also the most common cause of blindness among HIV patients as it is a progressive disease causing retinal destruction and ischemia. Even with appropriate therapy, the areas of retinal necrosis will not regenerate therefore the goal of CMV retinitis therapy is to preserve vision by preventing further retinal necrosis and reduce the risk of developing CMV-related visual complications.
Purpose/Objective: To present a case of Bilateral CMV Retinitis managed with monotherapy of intravitreal ganciclovir.
Methods: A case report of a 30-year-old Filipino male diagnosed with CMV Retinitis seen in a tertiary government hospital in the Philippines.
Results: This is a case of a 30-year-old male diagnosed with HIV 6 months before consult and was started on antiretroviral therapy (Efavirenz600mg/ Lamivudine300mg/ Tenofovir300mg). After 5 months of treatment, the patient noted gradual onset of generalized blurring of vision of both eyes associated with floaters. No other ocular symptoms were noted. The persistence of symptoms prompted the patient to seek consult at our institution. On examination, the patient’s best-corrected visual acuity is 20/80 on the right eye and 20/100 on the left. The anterior segment is unremarkable except for +1 anterior chamber cells on both eyes. Ocular motility and intraocular pressures were normal. Fundoscopic examination of both eyes showed retinal necrosis appearing as a white, fluffy lesion with an inflamed optic nerve. A diagnosis of Cytomegalovirus Retinitis was made given the clinical history and the classic retinal findings. A baseline fundus photo was done revealing neuroretinitis and retinal necrosis with overlying hemorrhages along the inferior arcade on the right eye and along the superior and inferior arcades on the left. The patient was treated with a 3-week induction phase of twice a week intravitreal ganciclovir injection (2mg) and a maintenance phase of once a week injection. Fundus photo was done before every injection. After the 3-week induction phase, the best-corrected visual acuity improved to 20/50 on the right and 20/30 on the left. Serial fundus photo revealed improvement of the disc margins and decreasing amount of hemorrhages with drying up of the lesions surrounding the macula.
Conclusion: In a tertiary government hospital where cost is an invariable concern, this report validates monotherapy of intravitreal ganciclovir as an effective option for patients due to affordability and ease of administration compared to systemic therapy. The case also exhibits the potential of intravitreal therapy to improve visual acuity in advance bilateral retinal disease.
Keywords
Case report; CMV retinitis; Ganciclovir; HIV
Introduction
Cytomegalovirus (CMV) infection is an infection seen in a wide variety of patients - both in immunocompetent patients presenting as mononucleosis and in the immunocompromised patients presenting as disseminated disease. Diagnosis of CMV has multiplied over the years due in part to the increase in Human Immunodeficiency Virus (HIV) diseases, as coinfection of HIV and CMV is fairly common [1]. As of March 2021, the HIV/AIDS & ART Registry of the Philippines reported a total of 85,651 confirmed HIV cases in the Philippines since it was first reported in 1984 [2]. The cumulative exponential growth of HIV in the country suggests a presumptive increase in CMV infections.
Cytomegalovirus retinitis is the most common ocular complication, developing in approximately half of the patients with HIV-CMV coinfection [3]. It is also the major cause of blindness among HIV-infected patients characterized by an acute and progressive neuroretinitis resulting in areas of necrosis and eventually scarring of the central and peripheral retina. In a descriptive study conducted by Berroya in the Philippines, 30 of 200 HIV seropositive patients demonstrated ocular manifestations; out of which, 40% presented with CMV retinitis [4].
In the modern era of antiretroviral therapy, the visual prognosis of AIDS and CMV retinitis has significantly improved [5-7]. The standard of care treatment focuses on intravenous, oral, and intravitreal injections of antiviral medications. In the present case, we used a readily available, inexpensive, and compounded drug, intravitreal ganciclovir. This provides a high concentration of intravitreal antiviral drugs to control intraocular infection without increasing systemic toxicity and side effects.
With limited access to other available drugs, we introduced monotherapy intravitreal ganciclovir treatment to a patient as an effective and financially acceptable therapeutic alternative. The procedure is reproducible and done on an outpatient basis under strict protocols.
Case Report
A 30-year-old HIV (+) patient experienced significant blurring of vision on both eyes. He was diagnosed with HIV six months prior using Western blot as a confirmatory test with undetectable levels of CD4 and had started on oral antiretroviral therapy (Efavirenz 600mg/ Lamivudine 300mg/ Tenofovir 300mg). After five months of treatment, he noted a gradual onset of generalized blurring of vision on both eyes, associated with seeing floaters.
On examination, visual acuity was 20/80 and 20/100 on the right and left eye, respectively. Funduscopic retinal examination showed areas of necrosis with overlying hemorrhages involving the optic nerve and macular areas.
The patient was assessed to have fulminant type of cytomegalovirus retinitis. A decision was made to initiate bilateral intravitreal ganciclovir monotherapy. Strict protocol was followed and he was treated with a 3 week induction phase of twice weekly intravitreal injections and a maintenance phase of weekly injections.
Pre injection photos (Figure 1) showed retinal necrosis appearing as yellow white lesions with hemorrhages along the inferior arcade on the right eye and along the superior and inferior arcades on the left eye. There was significant macular involvement with exudation and edema. Optic nerve heads were inflamed and obscured by the retinal hemorrhages.
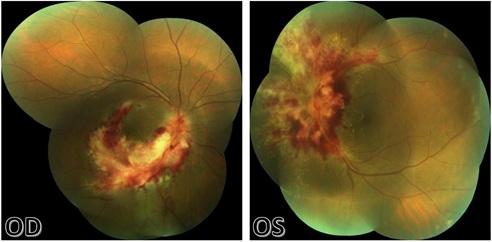 Figure 1: Fundus photo preinjection of right eye (left) and left eye (right).
Figure 1: Fundus photo preinjection of right eye (left) and left eye (right).
On the second week post intravitreal injection (Figure 2), the patient’s visual acuity improved to 20/60 right eye, and 20/30 left. There was beginning resolution of the hemorrhages and necrosis. The optic nerve of both eyes was more visible, but still with significant neuritis.
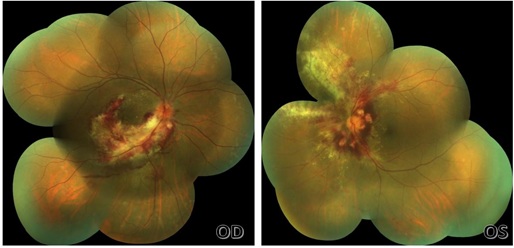 Figure 2: Fundus photo of right eye (left) and left eye (right) on second week.
Figure 2: Fundus photo of right eye (left) and left eye (right) on second week.
After the 3-week induction phase, visual acuity improved to 20/50 on the right eye and 20/30 on the left. Serial fundus photo showed a less inflamed optic nerve head with more distinct borders. The hemorrhages continue to resolve with areas of necrosis contracting. Pre retinal membranes and scars were visible (Figure 3).
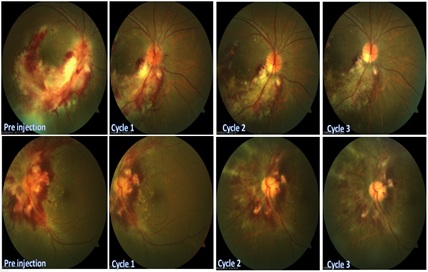 Figure 3: Serial photographs of fundus photo of the right eye (top layer) and left eye (bottom layer).
Figure 3: Serial photographs of fundus photo of the right eye (top layer) and left eye (bottom layer).
Case Discussion
CMV is a human herpes virus with four stranded DNA producing either lytic, productive or a latent infection. Reactivation of CMV infection in immunocompromised individuals leads to widespread infection causing significant morbidity and mortality.
Among HIV patients, CMV retinitis is the most common cause of blindness. It is also the most common ocular complication occurring in 30% of patients with AIDS [8]. The infection primarily manifests in patients with CD4 levels of <50 and almost always when the CD4 level is less than 200 cells/uL. Genetic factors related to lower T-cell proliferative response may also contribute to the patient’s predisposition to developing CMV retinitis [9]. Patients usually cite blurring of vision, scotomas, floaters, and photopsias, depending on the site of retinal necrosis. The microvascular damage also allows the CMV to access the retinal tissue and is clinically seen as cotton wool spots [6].
CMV retinitis is a clinical diagnosis. HIV and CD4 counts are supportive tests. Retinal examination shows areas of neuroretinitis, hemorrhages and necrosis. The disease is classified as fulminant, granular and perivascular specific to cytomegalovirus infection.
The following antiviral drugs are used for treating CMV retinitis - ganciclovir, foscarnet, cidofovir and valganciclovir. These medications are administered orally, thru systemic infusion, injected intravitreally or through sustained intraocular release devices. Treatment of choice is based on proper evaluation of the patient’s immune status and the corresponding retinal lesions. The protocol follows a two-phase strategy. The induction phase requires a high dose drug administration for two to three weeks to suppress viral replication. This is followed by the maintenance phase with a lower dose to suppress viral activity [5], and is continued until there is clinical recovery, such as inactivity of retinitis, improvement of visual acuity, CD4 levels of >100 for at least three months, and a decrease in the CMV viral load.
CMV retinitis is part of a systemic infection, and as such, patients should receive systemic therapy. The protocol requires long-term intravenous therapy of Ganciclovir 5mg/kg administered twice daily. The procedure necessitates the insertion of a catheter for intravenous access. Thereafter, the patient is monitored for known side effects such as catheter-related sepsis and drug-induced neutropenia, thrombocytopenia, anemia among others. Intravenous therapy is followed by daily maintenance of oral valganciclovir (900 mg twice daily). In selected patients, to prevent infusion side effects and lower costs, oral ganciclovir/aciclovir is used at a dose of 3 mg per day (12 capsules three times a day). A disadvantage is the bioavailability of only 6-9%, which is below the needed plasma concentration.
In the Philippines, access to the above therapeutic options is limited due to acquisition and financial constraints. Specifically, only acyclovir (tablet and vial), ganciclovir (vial), valaciclovir (tablet) and valganciclovir (tablet) are commercially available for the treatment of CMV retinitis. Intravenous ganciclovir approximately costs PHP 2,593.00 per vial and oral valganciclovir 450mg cost PHP 1498 per tablet. Following the protocol for systemic medication, a 2-week induction phase with 1 week of maintenance phase would cost a patient approximately PHP 44,587 and an additional PHP 41,994 per week in the maintenance phase.
A study done by Teerawattananon et al., [10] evaluated the cost-effectiveness of intravenous, oral, and intravitreal ganciclovir on patients with CMV retinitis. The authors recommended intravitreal ganciclovir as a legitimate choice where antiretroviral therapy is unavailable and finances are limited.
Local intraocular injections are typically an alternative option for patients who cannot tolerate systemic therapy due to contraindications or side effects. For CMV retinitis treatment, intravitreal injection is performed in two phases - induction phase of ganciclovir (2mg) injections twice a week for 3 weeks, followed by maintenance therapy once a week. This strategy provides a higher concentration of drug delivery to the retinal tissues with lower systemic exposure, thereby reducing side effects [6]. However, with a short half-life, frequent injections are required. Complications such as endophthalmitis, vitreous hemorrhage, and vascular occlusions have been noted in injection-related treatments. To prevent complications, strict adherence to intravitreal protocols should be exercised. In the Philippines, the total cost of treatment using intravitreal injection is approximately PHP 15,558.00 and an additional PHP 2593 per week in the maintenance phase which is only 20% of the usual cost for systemic treatment.
Generally, the cost of systemic treatment is unaffordable to the average Filipino population, and more so to patients attended in tertiary government hospitals such as ours. Left untreated, CMV retinitis will lead to permanent vision loss with deterioration to their quality of life [5]. Intravitreal ganciclovir injection presents itself as a viable and cost-effective alternative to control the progression of the CMV retinitis for patients with limited resources.
Conclusion
We describe a case of bilateral CMV retinitis on a patient with antiretroviral treatment. The efficacy and safety of intravitreal ganciclovir monotherapy injections resulted in significant retinitis resolution and improvement of visual acuity.
In a tertiary government hospital where cost is an invariable concern, this report validates monotherapy of intravitreal ganciclovir as an effective option for patients due to affordability and ease of administration compared to systemic therapy. The case also exhibits the potential of intravitreal therapy to improve visual acuity in advance bilateral retinal disease.
This report should not be taken as confirmation of the general effectiveness of using intravitreal ganciclovir monotherapy for CMV retinitis. However, the promising results in our patient are an addition to the growing evidence on the success of the intravitreal method as a therapeutic alternative in justified circumstances.
Finally, we want to emphasize that HIV patients should undergo routine posterior segment screening. If undiagnosed, CMV retinitis results in permanent blindness. Periodic fundoscopic examinations should be considered for early detection, documentation, and management. We recommend that all HIV patients be referred for ocular screening. Early therapy significantly decreases the incidence of permanent vision loss and improves the patient’s quality of life.
Acknowledgment
The authors are grateful to Dr Jan Patrick Chu for administering all the intravitreal injections to the patient.
References
- Gianella S, Letendre S (2016) Cytomegalovirus and HIV: A Dangerous Pas de Deux. J Infect Dis 214: 67-74.
- Department of Health| Epidemiology Bureau (2021) HIV/AIDS & ART Registry of the Philippine.
- Sharew G, Azage M (2015) Predictors of HIV/AIDS Related Ocular Manifestations among HIV/AIDS Patients in Felege Hiwot Referral Hospital, Northwest Ethiopia. J Ophthalmol 2015: 965627.
- Berroya R (2006) Ophthalmologic manifestations among filipinos living with hiv/aids (flwha): a descriptive study. Research Folio 2000-2006.
- Nkomazana O, Tshitswana D (2008) Ocular complications of HIV infection in sub-Sahara Africa. Curr HIV/AIDS Rep 5: 120-125.
- Manosuthi W, Chaovavanich A, Tansuphaswakiful S, Prasithsirikul W, Inthong Y, et al. (2007) Incidence and risk factors of major opportunistic infections after initiation of antiretroviral therapy among advanced HIV-infected patients in a resource-limited setting. J Infect 55: 464-469.
- Campbell RJ, Chow B, Victor G, Kravcik S, Hodge WG (2001) Treatment of CMV Retinitis with Intravitreal Ganciclovir in the HAART Era. Can J Infect Dis 12: 300-304.
- Hodge WG, Boivin J-F, Shapiro SH, Lalonde RG, Shah KC, et al. (2004) Clinical risk factors for cytomegalovirus retinitis in patients with AIDS. Ophthalmology, 111: 1326-1333.
- Stewart M (2010) Optimal management of cytomegalovirus retinitis in patients with AIDS. Clinical Ophthalmology 4: 285-299.
- Teerawattananon K, Iewsakul S, Yenjitr C, Ausayakhun S, Yenjitr W, et al. (2007) Economic Evaluation of Treatment Administration Strategies of Ganciclovir for Cytomegalovirus Retinitis in HIV/AIDS Patients in Thailand. Pharmacoeconomics 25: 413-428.
Citation: Sarmiento AMA, Fortuna EJ (2021) Use of Intravitreal Ganciclovir as Monotherapy for Bilateral CMV Retinitis in a Tertiary Government Hospital. J Ophthalmic Clin Res 8: 090.
Copyright: © 2021 Ana Margarita A Sarmiento, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

