
An Argentinian Proposal: Mesenchymal Stem Cells, Convalescent Plasma and Dexamethasone in Covid-19. Could be a Triple Strategy Effective for the Treatment of Severe Pneumonia Complications?
*Corresponding Author(s):
Matias Cristian Fernández ViñaFacultad De Medicina Y Cs De La Salud, Universidad Abierta Interamericana, Rosario, Argentina; Stem Cell Therapy Argentina, San Nicolás, Argentina; Fundación Don Roberto Fernández Viña, San Nicolás, Argentina; Clínica San Nicolás S.A, Argentina
Email:Matias.FernandezVina@UAI.edu.ar / matiasfernandezvina@hotmail.com
ABBREVIATIONS
MSC: Mesenchymal Stem Cell
AD-MSC: Adipose Derived Mesenchymal Stem Cells
UC-MSC: Umbilical Cord Mesenchymal Stem Cells
BACKGROUND
SARS-CoV-2 infections are a worldwide pandemic, posing a severe risk to health [1]. Human coronavirus, SARS-CoV-2, is highly pathogenic with severe pneumonia associated with rapid virus replication. Arising in Wuhan China December 2019, the current COVID-19 epidemic has rapidly grown with person-to-person infection expanding to become a global health emergency now on pandemic scale [2]. Since there are no currently accepted therapies for corona virus disease 2019 worldwide search for treatment options are ongoing. Past experiences with viral infections are important in this respect, like in Argentina practiced by Dr. Julio Maiztegui [3]. Convalescent plasma therapy and mesenchymal stem cell therapy are two of potential options for Covid-19, mentioned, in case series and non-randomized studies from different countries [4]. On the other hand, Glucocorticoids like Dexamethasone, may modulate inflammation-mediated lung injury and thereby reduce progression to respiratory failure and death [5].
METHODS
On this short paper communication, we propose the use of a triple strategy for Covid-19 in patients with severe pneumonia, showing safety in different separated publications reviewed Figure 1.
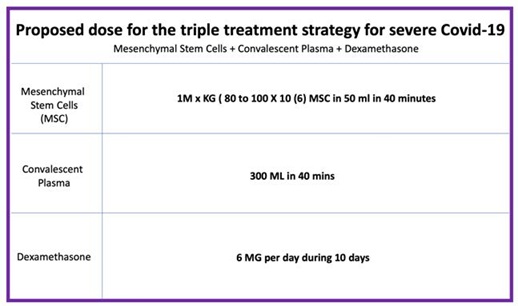 Figure 1: Triple treatment strategy with MSC + Convalescent Plasma + Dexamethasone applied on a same day for severe Covid-19.
Figure 1: Triple treatment strategy with MSC + Convalescent Plasma + Dexamethasone applied on a same day for severe Covid-19.
The triple strategy that has shown individually, safe and possible effectiveness in the treatment for Covid-19 pneumonia, could be a way of synergic treatment, following this review.
Mesenchymal stem cell therapy
Mesenchymal Stem Cell (MSC) therapy has been the subject of research in many different fields of medicine due to its immunomodulatory and reparative effects.
The alveolar injury by COVID-19 is characterized by the impairment of endothelial and type I alveolar cell (pneumocyte) barrier, which eventually results in an intense accumulation of proteinaceous fluid (alveolar edema) and mononuclear cell (lymphocytes) infiltrates in interalveolar septa. Extravasated and/or resident MSCs can trigger a series of direct and indirect repairing mechanisms. Clearance of the increased alveolar edema can be induced by the release of KGF by enhancing sodium-dependent alveolar fluid clearance through type II alveolar sodium channels. The MSC-released TSG6 decreases neutrophil functions, which directly affects improvement of the vascular endothelial and alveolar epithelial barriers [6].
The resolution of inflammation can be further enhanced by the increased release of IL-10 and decreased release of TNF-α, which are mediated by LXA4 and PGE2. Increased epithelial repair in type II alveolar cells can be restored by the release of Ang1. MSCs can also facilitate the phagocytosis of bacteria by the intra-alveolar and interalveolar macrophages by releasing the antimicrobial peptide LL-37 or by the transfer of extracellular vesicles to macrophages from MSCs [6].
Additionally, MSCs can exert their actions through mitochondrial transfer to injured alveolar cells, thus increasing their ATP content, which would improve bioenergetics and increase alveolar epithelial function, improving surfactant release by type II alveolar cells. MSCs can degrade or inhibit ARDS-induced fibrotic tissue formation (collagen fiber accumulation) to modulate the extracellular matrix by releasing MMPs and TIMPs. In MSC-based therapies, infusion of auto/allogeneic MSCs is applied through two primary routes (intravenous and intratracheal/intrabronchial) [7].
The most important mechanism is that MSCs release many paracrine factors, such as micro-RNA, interacting with the immune response to exert immunoregulation and anti-inflammatory effects [8].
Three publications, two from China, one case report and one case series, and one from Spain, have reported positive results of intravenous administration of Umbilical Cord-MSC (UC-MSC) or Adipose Derived-MSC (AD-MSC) in severely ill COVID-19 patients [9,10]. It has been reported that, clinical, radiological, and laboratory improvement has been observed within days of therapy in these patients [8-10]. Up to date, there are more than 50 trialsregistered on going with MSC for Covid-19 (10) (56 clinical trials were registered in clinicaltrials.gov under COVID and “mesenchymal” terms as of the writing date of this review).
Treatment with intravenous administration of AD-MSC or UC-MSC in severe COVID-19 pneumonia under mechanical ventilation in a small case series did not induce significant adverse events and was followed by clinical and biological improvement in most subjects [8-10]. It is considered that infection of mesenchymal stem cells with SARS-CoV-10 is not an expected condition since they lack ACE2 receptors [11-13]. As well, ongoing cytokine production stimulate these cells to produce anti-inflammatory and trophic mediators, which help to confine the cytokine storm and the resulting lung injury. The remarkable new data using MSC demonstrate successful harnessing of natural endogenous pathways with powerful protective properties [2]. The dose recommended in the BALMYS-19-“BAttLe against COvid using MesenchYmal Stromal cells” was 1.000.000 MSC X kg [10].
Dexamethasone
In patients hospitalized with Covid-19, the preliminary results of the controlled, open-label Randomized Evaluation of Covid-19 Therapy (RECOVERY) trial of dexamethasone showed that the use of it resulted in lower 28-day mortality among those who were receiving either invasive mechanical ventilation or oxygen alone at randomization but not among those receiving no respiratory support [5].
The benefit was also clear in patients who were being treated more than 7 days after symptom onset, when inflammatory lung damage is likely to have been more common [5].
The protocol combines the methods that were used in large, simple trials of treatments for acute myocardial infarction in the 1980s with the opportunities provided by digital health care in the 2020s [14-16].
The RECOVERY trial provides evidence that treatment with dexamethasone at a dose of 6 mg once daily for up to 10 days reduces 28-day mortality in patients with Covid-19 who are receiving respiratory support [5].
Dexamethasone is on the list of essential medicines of the World Health Organization and is readily available worldwide at low cost. Guidelines issued by the U.K. chief medical officers and by the National Institutes of Health in the United States have already been updated to recommend the use of glucocorticoids in patients hospitalized with Covid-19 [17,18].
Slower clearance of viral RNA has been observed in patients with SARS, MERS, and influenza who were treated with systemic glucocorticoids, but the clinical significance of these findings is unknown. Unlike with SARS, in which viral replication peaks in the second week of illness, viral shedding in SARS-CoV-2 appears to be higher early in the illness and de- clines thereafter [18].
Convalescent plasma
One of the potential treatment options that is being evaluated extensively is convalescent plasma therapy, which provides passive immunization. With convalescent plasma therapy, viral neutralization and consequently decreased target organ injury is aimed. The rationale behind this treatment is basic knowledge and experience from the recent studies on MERS, SARS and H1N1 infections and several other infections at the beginning of twentieth century [20]. In Argentina it was used for the treatment of the Argentine hemorrhagic fever with very good results to reduce mortality [3] a few years ago by Dr. Julio Maiztegui.
Patients who want to donate for convalescent plasma must have recovered from COVID-19 symptoms, their SARS-CoV-2 PCR tests should have become negative, and they must have adequate antibody response to SARS-CoV-2. It is considered to be most effective during the initial week, and it may be considered to be ineffective after 14th day [21-23].
Prior to administration presence of IgA deficiency or known allergy to contents (i.e. citrate) of the product should be ruled out. Presence of IgA deficiency may result in severe anaphylactic reaction [20].
Evidence shows that convalescent plasma from patients who have recovered from viral infections can be used as a treatment without the occurrence of severe adverse events. Therefore, it might be worthwhile to test the safety and efficacy of convalescent plasma transfusion in SARS-CoV-2-infected patients [22].
The doses of Convalescent Plasma used as described by the different studies are varied. A Chinese pilot study showed a minimal use of a single dose of 200 mL convalescent plasma with neutralizing antibody titers >1:640. Another study by Bin Zhang et al., reported a maximum of 2400 mL of convalescent plasma administered to a 73 years old male patient [23].
The dose we recommend of Convalescent Plasma is 300 mL/unique dose.
THE ARGENTINIAN PROTOCOL. TRIPLE TREATMENT STRATEGY AS IMMUNOMODULATORY AND ANTIVIRAL EFFECT FOR COVID-19
Although it has been observed from one case that intravenous infusion of plasma and mesenchymal stem cells have a synergistic therapeutic effect on patients with severe COVID-19 pneumonia [8], in theory, the mechanisms of action of convalescent plasma and mesenchymal stem cells have complementary characteristics. We believe that a triple treatment strategy, including dexamethasone, might benefit patients with COVID-19 pneumonia without better options to stop the cytokine storm.
ELIGIBILITY FOR MSC + CONVALESCENT PLASMA + DEXAMETHASONE AS A TRIPLE TREATMENT STRATEGY ON A SAME DAY
Inclusion criteria
- Patient 18 years old or older.
- Hospitalized with highly severe COVID-19 respiratory symptoms and confirmation of infectionpositive using the RT-PCR test COVID-19 SARS-CoV-2 (real-time PCR).
- Patient duly informed, to the extent of their conditions, who is a voluntary signer of a comprehensive Informed Consent Form. In case of not being able to do so, the authorizing signature of the legal representative, the support, the spouse, the cohabitant, the relative or the close friend will be obtained.
- Severely compromised patient, presenting any of these symptoms and / or parameters:
- • Dyspnea
- • Respiratory rate ≥ 30 / min
- • Blood oxygen saturation ≤ 93%
- • Ratio between partial pressure of arterial oxygen and fraction of inspired oxygen <300
- • Lung infiltrates> 50% within 24 to 48 hours
Or that it is in a terminal state and for whom the doctor considers that it has a high-riskmortality
- • Respiratory failure
- • Septic shock
- • Multiple organ failure or dysfunction
Exclusion criteria
- Contraindication to transfusion: history of anaphylaxis to blood products.
- In relation to other comorbidities: according to the criteria of the treating physician.
CONCLUSION
Based on evidence reported in systematic reviews, meta-analysis and clinical trials including systemic applied MSC, Convalescent Plasma and Dexamethasone, this triple theoretical therapy strategy appears to be safe in different conditions and situations and could prove a useful way for COVID 19 patients given their immunomodulatory and anti-inflammatory properties. This theoretical treatment stimulates our group to design a Phase 1 protocol for the treatment of severe pneumonia for Covid-19 in Argentina.
ACKNOWLEDGEMENTS
We thank Dr Marcelo A. Fernandez Vina for his helpful comments and invaluable insights that contributed to the development of this treatment protocol.
DECLARATIONS
MFV, LC, MIS, RFV, have nothing to declare related to this manuscript.
FUNDING INFORMATION
This work was funded by Fundación Don Roberto Fernández Viña.
REFERENCES
- Casadevall A, Pirofski LA (2020) The convalescent sera option for containing COVID-19. J Clin Invest 30: 1545-1548.
- Metcalfe SM (2020) Mesenchymal stem cells and management of COVID-19 pneumonia. Med Drug Discov 5: 100019.
- Maiztegui JI (1975) Clinical and epidemiological patterns of Argentine hemorrhagic fever. Bull World Health Organ 52: 567-575.
- Valk SJ, Piechotta V, Chai KL, Doree C, Monsef I, et al. (2020) Convalescent plasma or hyperimmune immunoglobulin for people with COVID-19: A Rapid Review. Cochrane Database Syst Rev 5: CD013600.
- Lane HC, Fauci AS (2020) Research in the Context of a Pandemic. N Engl J Med: 32678528.
- Laffey JG, Matthay MA (2017) Fifty years of research in ARDS. Cell-based therapy for acute respiratory distress syndrome. Biology and poten- tial therapeutic value. Am J Respir Crit Care Med 196: 266-273.
- Can A, Coskun H (2020) The rationale of using mesenchymal stem cells in patients with COVID-19- related acute respiratory distress syndrome: What to expect. STEM CELLS Transl Med 1-16.
- Peng H, Gong T, Huang X, Sun X, Luo H, et al. (2020) A synergistic role of convalescent plasma and mesenchymal stem cells in the treatment of severely ill COVID-19 patients: A clinical case report. Stem Cell Res Ther 11: 291.
- Leng Z, Zhu R, Hou W, Feng Y, Yang Y, et al. (2020) Transplantation of ACE2- mesenchymal stem cells improves the outcome of patients with COVID-19 pneumonia. Aging Dis 11: 216-228.
- Sanchez-Guijo F, García-Arranz M, López-Parra M, Monedero P, Mata-Martínez C, et al, (2020) Adipose-derived mesenchymal stromal cells for the treatment of patients with severe SARS- CoV-2 pneumonia requiring mechanical ventilation. A proof of concept study. E Clinical Medicine: 100454.
- Mesenchymal Stem Cells Covid-19 (2019) National Institutes of Health, USA.
- Metcalf SM (2020) Mesenchymal stem cells and management of COVID-19 pneumonia. Med Drug Discov 5: 100019.
- Atluri S, Manchikanti L, Hirsch JA (2020) Expanded umbilical cord mesenchymal stem cells (UC-MSCs) as a therapeutic strategy in managing critically ill COVID-19 patients: The case for compassionate use. Pain Physician 23: 71-83.
- Yusuf S, Collins R, Peto R (1984) Why do we need some large, simple randomized trials? Stat Med 3: 409-422.
- ISIS-2 (Second International Study of Infarct Survival) Collaborative Group (1988) Randomised trial of intravenous streptokinase, oral aspirin, both, or neither among 17,187 cases of suspected acute myocardial infarction: ISIS-2. Lancet 2: 349-360.
- Collins R, Bowman L, Landray M, Peto R (2020) The magic of randomization versus the myth of real-world evidence. N Engl J Med 382: 674-678.
- Whitty C (2020) Dexamethasone in the treatment of COVID-19: Implementation and management of supply for treatment in hospitals. Medicines and Healthcare Products Regulatory Agency, London, UK.
- COVID-19 treatment guidelines (2020) National Institutes of Health, Bethesda, Maryland, United States.
- Horby P, Lim WS, Emberson JR, Mafham M, Bell JL, et al. Dexamethasone in Hospitalized Patients with Covid-19 - Preliminary Report. N Engl J Med: 32678530.
- Altintas ND (2020) Convalescent Plasma and Mesenchymal Stem Cell Therapy. J Crit Intensive Care 11: 41-42.
- Huang C, Wang Y, Li X, Ren L, Zhao J, et al. (2020) Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 395: 497-506.
- Chen L, Xiong J, Bao L, Shi Y (2020) Convalescent plasma as a potential therapy for COVID-19. Lancet Infect Dis 20: 398-400.
- Rajendran K, Krishnasamy N, Rangarajan J, Rathinam J, Natarajan M, et al. (2020) Convalescent plasma transfusion for the treatment of COVID?19: Systematic review. J Med Virol:1-9.
Citation: Viña MCF, Viña RF, Camozzi L, Spitaleri MI, Viña LF (2020) An Argentinian Proposal: Mesenchymal Stem Cells, Convalescent Plasma and Dexamethasone in Covid-19. Could be a Triple Strategy Effective for the Treatment of Severe Pneumonia Complications?. J Stem Cell Res Dev Ther 6: 044.
Copyright: © 2020 Matias Cristian Fernández Viña, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

