
Journal of Stem Cells Research Development & Therapy Category: Medical
Type: Research Article
Improved Bone Formation by Differentiated Mesenchymal Stem Cells and Endothelial Progenitor Cells Seeded on High Concentrated Bioglass-Polylactic Acid Composite in Calvarial Rat Bone Defect
*Corresponding Author(s):
Dirk HenrichDepartment Of Trauma Surgery, Johann-Wolfgang-Goethe University Hospital, Theodor-Stern-Kai 7, 60590 Frankfurt, Germany
Tel:+49 6963017110,
Fax:+49 6963017108
Email:caroline.seebach@kgu.de
Caroline Seebach
Department Of Trauma Surgery, Johann-Wolfgang-Goethe University Hospital, Theodor-Stern-Kai 7, 60590 Frankfurt, Germany
Tel:+49 6963017110,
Fax:+49 6963017108
Email:caroline.seebach@kgu.de
Received Date: Feb 24, 2015
Accepted Date: May 07, 2015
Published Date: May 25, 2015
Abstract
Objective: New developed composite biomaterials with a Bioglass (BG) and Polylactic Acid (PLA) component are promising candidates for the treatment of bone defects. There is evidence that adding Mesenchymal Stem Cells (MSC) and Endothelial Progenitor Cells (EPC) significantly improve new bone formation. Thus, cell adhesion, cell viability and bone formation of these composites, when seeded with undifferentiated or differentiated progenitor cells, respectively were tested.
Materials and methods: We investigated newly developed composite material consisting of Polylactic Acid (PLA), PLA and 20% Bioglass (PLA+BG 20%) or PLA and 40% Bioglass (PLA+BG 40%). These materials were seeded with either undifferentiated MSC / EPC or differentiated MSC / EPC and tested for cell adhesion and cell viability in vitro. Moreover, these composites were evaluated for bone formation in vivo. A Critical Size Defect (CSD) was made in each calvarium of 76 rats and composites were implanted. Animals were sacrificed after 14 weeks. Formation of new bone was evaluated by histomorphometry.
Results: Cell adhesion and cell viability in vitro is not significantly influenced by our tested composites, but differentiated MSC/EPC seeded onto PLA+BG40 improve significantly bone formation in a calvarial rat bone defect in vivo and represent a novel cell-based therapy for bone regeneration.
Materials and methods: We investigated newly developed composite material consisting of Polylactic Acid (PLA), PLA and 20% Bioglass (PLA+BG 20%) or PLA and 40% Bioglass (PLA+BG 40%). These materials were seeded with either undifferentiated MSC / EPC or differentiated MSC / EPC and tested for cell adhesion and cell viability in vitro. Moreover, these composites were evaluated for bone formation in vivo. A Critical Size Defect (CSD) was made in each calvarium of 76 rats and composites were implanted. Animals were sacrificed after 14 weeks. Formation of new bone was evaluated by histomorphometry.
Results: Cell adhesion and cell viability in vitro is not significantly influenced by our tested composites, but differentiated MSC/EPC seeded onto PLA+BG40 improve significantly bone formation in a calvarial rat bone defect in vivo and represent a novel cell-based therapy for bone regeneration.
Keywords
Bioglass, Bone tissue engineering, EPC, MSC, PLA
INTRODUCTION
Over 2 million bone graft materials are used every year worldwide, thus bone is second only to blood on the list of transplanted materials. With increasing demand and known limitations with traditional bone graft materials, new approaches are developed to provide alternatives for bone regeneration [1-3].
Bone tissue engineering tries to mimic the physiologic situation [4]. The addition of osteogenic and angiogenic cells to a synthetic biomaterial increases their local density and rely on locally secreted growth and differentiation factors to induce bone formation. The biomaterials should present good biocompatibility for cell adhesion and cell viability [5,6] as well as controlled degradation kinetics to match the ratio of replacement by new tissue. Also, the biomaterials should provide an initial biomechanical support until cells generate the extracellular matrix [7,8].
Bioactive Glasses (BG) are a subset of inorganic bioactive materials, which are capable of reacting with physiological fluids to form tenacious bonds to bone through the formation of bone-like hydroxylapatite layers and the biological interaction of collagen with the material surface [9,10]. It has been found that reactions on BG surfaces lead to the release of critical concentrations of soluble Silicon (Si) and Calcium (Ca) ions, which induce favourable intracellular and extracellular responses leading to rapid bone formation [11]. Although BG has traditionally been employed for its osteoconductive and osteostimulative properties, BG also exhibit proangiogenic potential in vitro and in vivo. Soluble dissolution products of BG up-regulate the production of numerous angiogenic factors by stimulated cells providing a potentially promising strategy to enhance early vascularisation and resultant bone formation [12-15].
However, BG, compared to cortical and cancellous bones, usually present low mechanical properties, especially in porous forms [16,17]. This disadvantage significantly limits the use of these materials in a very broad range of applications. Fortunately, one solution came from mimicking nature, which provides the inspiration to design materials with optimal organized structures under dynamically changing conditions. Many of these structures are composed of an intrinsically complex matrix based on organic and inorganic components which produce a natural hybrid material, usually referred to as composites. By combining two or more materials in a predesigned manner, a biomaterial can be created with properties that are not possible to be attained when considering each of the individual components separately [18].
Synthetic polymers (e.g., Poly-Lactide Acid (PLA)) have numerous advantages, such as excellent processing characteristics, which can ensure the off-the-shelf availability as well as being biocompatible and biodegradable at rates that can be tailored for the intended application [19,20]. Additionally, synthetic polymers possess predictable and reproducible mechanical and physical properties (e.g., tensile strength, elastic modulus, and degradation rate) and can be manufactured with great precision [21].
Thus, PLA/BG composite biomaterials present an ideal clinical solution to the limitations of traditional bone graft. But the optimal composition of PLA/BG composite for cell adhesion, cell viability and bone tissue engineering is still unknown.
Therefore, we prepared and investigated three different compositions of PLA/BG composite: Polymer (PLA), PLA+BG 20% and PLA+BG 40% (the glass content is 0, 20 and 40 wt% respectively) for bone tissue engineering in vitro and in vivo.
Moreover, recent investigations of our laboratory have focused on implantation of undifferentiated MSC and EPC seeded onto Tricalciumphosphate (TCP) which demonstrated enhanced bone regeneration and improved vascularization of critical size bone defects [22-24]. In these previous studies EPC demonstrated real angiogenic contribution. In this context it is unknown if differentiation of MSC/EPC can enhance bone formation. Thus, we hypothesized that the localized delivery of differentiated MSC / EPC onto PLA/BG composite enhance bone formation and promote bone healing in a critical-sized calvarial bone defect in rats.
The specific aims of this study were twofold:
Bone tissue engineering tries to mimic the physiologic situation [4]. The addition of osteogenic and angiogenic cells to a synthetic biomaterial increases their local density and rely on locally secreted growth and differentiation factors to induce bone formation. The biomaterials should present good biocompatibility for cell adhesion and cell viability [5,6] as well as controlled degradation kinetics to match the ratio of replacement by new tissue. Also, the biomaterials should provide an initial biomechanical support until cells generate the extracellular matrix [7,8].
Bioactive Glasses (BG) are a subset of inorganic bioactive materials, which are capable of reacting with physiological fluids to form tenacious bonds to bone through the formation of bone-like hydroxylapatite layers and the biological interaction of collagen with the material surface [9,10]. It has been found that reactions on BG surfaces lead to the release of critical concentrations of soluble Silicon (Si) and Calcium (Ca) ions, which induce favourable intracellular and extracellular responses leading to rapid bone formation [11]. Although BG has traditionally been employed for its osteoconductive and osteostimulative properties, BG also exhibit proangiogenic potential in vitro and in vivo. Soluble dissolution products of BG up-regulate the production of numerous angiogenic factors by stimulated cells providing a potentially promising strategy to enhance early vascularisation and resultant bone formation [12-15].
However, BG, compared to cortical and cancellous bones, usually present low mechanical properties, especially in porous forms [16,17]. This disadvantage significantly limits the use of these materials in a very broad range of applications. Fortunately, one solution came from mimicking nature, which provides the inspiration to design materials with optimal organized structures under dynamically changing conditions. Many of these structures are composed of an intrinsically complex matrix based on organic and inorganic components which produce a natural hybrid material, usually referred to as composites. By combining two or more materials in a predesigned manner, a biomaterial can be created with properties that are not possible to be attained when considering each of the individual components separately [18].
Synthetic polymers (e.g., Poly-Lactide Acid (PLA)) have numerous advantages, such as excellent processing characteristics, which can ensure the off-the-shelf availability as well as being biocompatible and biodegradable at rates that can be tailored for the intended application [19,20]. Additionally, synthetic polymers possess predictable and reproducible mechanical and physical properties (e.g., tensile strength, elastic modulus, and degradation rate) and can be manufactured with great precision [21].
Thus, PLA/BG composite biomaterials present an ideal clinical solution to the limitations of traditional bone graft. But the optimal composition of PLA/BG composite for cell adhesion, cell viability and bone tissue engineering is still unknown.
Therefore, we prepared and investigated three different compositions of PLA/BG composite: Polymer (PLA), PLA+BG 20% and PLA+BG 40% (the glass content is 0, 20 and 40 wt% respectively) for bone tissue engineering in vitro and in vivo.
Moreover, recent investigations of our laboratory have focused on implantation of undifferentiated MSC and EPC seeded onto Tricalciumphosphate (TCP) which demonstrated enhanced bone regeneration and improved vascularization of critical size bone defects [22-24]. In these previous studies EPC demonstrated real angiogenic contribution. In this context it is unknown if differentiation of MSC/EPC can enhance bone formation. Thus, we hypothesized that the localized delivery of differentiated MSC / EPC onto PLA/BG composite enhance bone formation and promote bone healing in a critical-sized calvarial bone defect in rats.
The specific aims of this study were twofold:
- To compare the osteogenic potential of various concentration of bioglass in the composite: PLA; PLA+BG 20%, PLA+BG 40%
- To determine the osteogenic potential of undifferentiated MSC / EPC versus differentiated MSC / EPC.
MATERIALS AND METHODS
Ethic statements
All animal experiments were performed in accordance with the institutional animal care and oversight committee (Project No. F3/22; Regierungspräsidium Darmstadt, Germany). All efforts were made to minimize suffering.
Characteristics of composite biomaterials: PLA, PLA+BG 20 and PLA+BG 40
Depending on the rate of burning, we created a new particle size of Bioglass (BG). The composite biomaterial consists of a PLA-component supplemented with increasing amounts of BG. Tetraethyl orthosilicate (TEOS, ≥ 99%) and Nitric acid 65% were supplied by Merck Chemicals KGaA, Darmstadt, Germany. Calcium nitrate [Ca (NO3)2-4H2O, ≥ 99%], Poly (L-lactide) and Chloroform (CHCl3, ≥ 99.4%) were purchased from Sigma-Aldrich Chemie GmbH, Steinheim, Germany. All chemicals were reagent grade and were used as received without further purification. For the synthesis of BG CaO-SiO2 (SiO2 80mol-%-CaO 20mol-%), low viscosity gel was obtained by mixing 31 mL of Tetraethyl Orthosilicate (TEOS) and 8.6g of Ca(NO3)2.4H2O in a solution of 5.5mL of HNO3 2M, used as catalyst, in 31.5 mL of H2O. The initial pH of hydrolysis was 0.5. The BG was cast at room temperature in Teflon container (Thermo Scientific Nalgene, Germany) until the gel was formed. Aging was performed at 60Cº for 3 days. Drying was carried out at 120Cº. The glass was collected in laboratory porcelain crucible (Haldenwanger GmbH, Waldkraiburg, Germany) after that was burned in a muffle furnace (Nabertherm GmbH, Lilienthal, Germany) at rate 3ºC/1 minute till 700Cº, then 700ºC for 3 hours. The glass particles were grinded in small porcelain mortal (Haldenwanger GmbH, Waldkraiburg, Germany) to form glass powder. Finally, Bioglass particles sizes were sieved to be in the range of 106-125µm by Test sieves (Retsch GmbH, Haan, Germany).
Composite biomaterials were prepared by mixing polymer [poly(L-lactide) (PLA)] and Bioglass (BG) with 10 ml chloroform as follows: PLA, PLA/BG 20% (PLA+BG 20%) and PLA/BG 40% (PLA+BG 40%) biomaterial. The bioglass content was 0, 20 and 40 % by weight. These biomaterials will be referred to as PLA, PLA+BG 20% and PLA+BG 40%. Disc shaped specimens with a diameter of 5 mm and a thickness of 1 mm were cut and stored at room temperature under sterile conditions until use.
Composite biomaterials were prepared by mixing polymer [poly(L-lactide) (PLA)] and Bioglass (BG) with 10 ml chloroform as follows: PLA, PLA/BG 20% (PLA+BG 20%) and PLA/BG 40% (PLA+BG 40%) biomaterial. The bioglass content was 0, 20 and 40 % by weight. These biomaterials will be referred to as PLA, PLA+BG 20% and PLA+BG 40%. Disc shaped specimens with a diameter of 5 mm and a thickness of 1 mm were cut and stored at room temperature under sterile conditions until use.
Cell isolation and preparation of rat Endothelial Progenitor Cells (EPC) from rat spleen
Rat spleens (donor rats; n=7) were cut in tiny pieces and gently rubbed. Viscous solution were resuspended in Phosphate Buffered Saline (PBS) and filtered through 100mm, 70mm and 40mm mesh (BD-Bioscience, Heidelberg, Germany). Subsequently, the cell suspension was layered on a Ficoll density gradient (1.077g/mL; Biochrom, Berlin, Germany) and density gradient centrifugation (30min, 900g). Cells were washed twice with cold PBS (10min, 900g), and 4×106 cells were cultivated on a fibronectin-coated (10mg/mL; Sigma, Deisenhofen, Germany) 24-well culture dish in 1mL of endothelial basal medium (Cambrex, Verviers, Belgium) supplemented with endothelial growth medium (Cambrex) at 37.8°C. After 48h, nonadherent and weakly adherent cells were removed, the medium was changed. The cells were cultivated for an additional 72h. A parallel preparation was performed to evaluate the percentage of endothelial cell-like differentiated cells. EPC were identified using the method previously described [37]. Briefly, cells were incubated for 1h with 2.4mg=mL DiLDL (Cell-Systems, St. Katharinen, Germany) in EBM supplemented with 20% FCS. Cells were fixed with 2% paraformaldehyde for 10min, and after washing with PBSþ=þ, FITC-labeled Ulex europaeus agglutinin-1 [10 mg=mL] (lectin; Sigma) was incubated for 1h. Cells presenting double-positive fluorescence were considered to be EPC. Only preparations with a percentage of endothelial-like differentiated cells greater than 80% were used. For the experiments the cells were detached by incubation (10min) with accutase (PAA-laboratories, Linz, Austria), washed once with MesenCultþ Supplements (Cell-Systems), and subsequently adjusted to a density of 2.5x10^5 cells in 100mL.
Cell isolation of rat Mesenchymal Stem Cells (MSC) from rat femur
Mesenchymal stem cells were isolated from rat femurs (donor rats; n=3). Bone marrow aspirate was washed once using PBS. The pellet was resuspended in PBS and layered on a Ficoll density gradient (d = 1,077g/mL, Biochrom, Berlin, Germany).
After centrifugation (30 min, 1100 g) the cells in the interphase were collected and washed twice using PBS (10 min, 900g) containing 2% Fetal Bovine Serum (FBS). The cells were resuspended in 3 ml DMEM/F-12 and Supplements (gibco® by life technologies, Germany) and were counted.
After centrifugation (30 min, 1100 g) the cells in the interphase were collected and washed twice using PBS (10 min, 900g) containing 2% Fetal Bovine Serum (FBS). The cells were resuspended in 3 ml DMEM/F-12 and Supplements (gibco® by life technologies, Germany) and were counted.
Differentiation of rat Mesenchymal Stem Cells (MSC)
MSC were incubated with osteogenic differentiation medium: DMEM/F-12 (gibco® by life technologies, Germany) medium, Supplements, dexamethason [1µM], ascorbic acid [50µg/ml] and β-glycerol phosphate [10 000µM] (Stem Cell Technologies, Germany) for 3 weeks. Extracellular calcium deposition was evaluated by van Kossa staining (Figure 1A and B).
Cell seeding onto composite biomaterials
10µl medium containing 2.5x105 undifferentiated MSC and 2.5x105 EPC were dropped over one disk of biomaterial (PLA vs PLA+BG20 vs PLA+BG40), then cells seeded onto composite were incubated for one hour in CO2 incubator at 37°C. This cell seeding procedure was also performed with differentiated MSC/EPC.
Scanning Electron Microscopy (SEM) of MSC and EPC onto composite biomaterials
Qualitative analysis of the morphology of adherent MSC and EPC were assessed by Scanning Electron Microscopy (SEM). The seeded biomaterials were fixed with glutardialdehyde for 30min and subsequently dehydrated by incubation of each 15min in a 4-step ethanol gradient. Then they were incubated overnight in 1,1,1,3,3,3-hexamethyldisilazane (Merck-Schuchardt, Hohenbrunn, Germany) and drained. Afterwards the samples were sputtered with gold (3_60s, Agar Sputter Coater, Agar Scientific Ltd., UK) using a Hitachi FE-SEM S4500 (Hitachi, Düsseldorf, Germany) with a voltage of 5kV. The images (Figure 2) were digitally recorded using the Digital Image Processing System 2.6 (Point Electronic, Halle, Germany).
Cell viability of MSC and EPC after seeding onto composite biomaterials
Cells (undifferentiated MSC/EPC and differentiated MSC/EPC) were detached by 10 minutes incubation with accutase (PAA-laboratories), then washed (5minutes, 300g), resuspended in DMEM supplemented with 10% fetal calf serum (FCS; PAA Laboratories) and adjusted to a density of 500 x105 cells in 1ml medium.
In each well from 24 well plate 10µl medium which contains either 2.5x105 undifferentiated MSC and 2.5x105 EPC or 2.5x105 differentiated MSC and 2.5x105 EPC, respectively were dropped over the biomaterial and incubated in CO2 incubator at 37°C for 1hour.
In order to stain MSC and EPC nuclei, cells were fixed with 3% paraformaldehyde for 10 minutes and after washing with PBS++(with calcium and magnesium), 1µL 40,6-Diamidino-2-Phenylindole (DAPI; Sigma-Aldrich, Deisenhofen, Germany; final concentration 1µg=mL) were added to each well followed by further incubation for 15 minutes at 37C°.
In order to detect EPC, cells were incubated for 1h with 2.9 µg/mL 1,1=-dioctadecyl-3,3,3=,3=-tetramethylindo-carbocyanine-labeled acetylated low density lipoprotein (DiLDL; Cell-Systems, St. Katharinen, Germany) in EBM supplemented with 20% FCS.
After three washes with PBS++, the biomaterials were subjected to fluorescence microscopy (Axio Observer; Zeiss, Göttingen, Germany) in order to view DAPI-stained MSC as well as DiLDL/DAPI-stained EPC (Figure 3).
In each well from 24 well plate 10µl medium which contains either 2.5x105 undifferentiated MSC and 2.5x105 EPC or 2.5x105 differentiated MSC and 2.5x105 EPC, respectively were dropped over the biomaterial and incubated in CO2 incubator at 37°C for 1hour.
In order to stain MSC and EPC nuclei, cells were fixed with 3% paraformaldehyde for 10 minutes and after washing with PBS++(with calcium and magnesium), 1µL 40,6-Diamidino-2-Phenylindole (DAPI; Sigma-Aldrich, Deisenhofen, Germany; final concentration 1µg=mL) were added to each well followed by further incubation for 15 minutes at 37C°.
In order to detect EPC, cells were incubated for 1h with 2.9 µg/mL 1,1=-dioctadecyl-3,3,3=,3=-tetramethylindo-carbocyanine-labeled acetylated low density lipoprotein (DiLDL; Cell-Systems, St. Katharinen, Germany) in EBM supplemented with 20% FCS.
After three washes with PBS++, the biomaterials were subjected to fluorescence microscopy (Axio Observer; Zeiss, Göttingen, Germany) in order to view DAPI-stained MSC as well as DiLDL/DAPI-stained EPC (Figure 3).
Animals and cell transplantation
76 sixteen-week-old male albino (Sprague Dawely Strains) rats (Charles River, Germany) weighting approximately 350-450g were housed, four animals per cage under standardized conditions: 15-21°C, air flow, 12h light cycle, rat food and water ad libitum. The rats were randomly allocated to the experimental groups (Table 1).
| Group | Biomaterial | Cells | Animals ( n) |
| 1 | bone | -- | 6 |
| 2 | Empty | -- | 6 |
| 3 | PLA | -- | 6 |
| 4 | BG 20 | -- | 5 |
| 5 | BG 40 | -- | 6 |
| 6 | PLA | EPC + MSC | 8 |
| 7 | PLA+ BG20% | EPC + MSC | 8 |
| 8 | PLA+ BG40% | EPC + MSC | 7 |
| 9 | PLA | EPC + d.MSC | 8 |
| 10 | PLA+ BG20% | EPC + d.MSC | 8 |
| 11 | PLA+ BG40% | EPC + d.MSC | 8 |
PLA: Polymer (poly (L-lactide)); PLA+Bioglass 20% (PLA+BG20): composite of 80% PLA and 20% Bioglass; PLA+ Bioglass 40% (PLA+BG40): composite of 60% PLA and 40% Bioglass; EPC: Endothelial Progenitor Cells; MSC: Mesenchymal Stem Cells; dMSC: osteogenic differentiated MSC
According to the experimental groups (Table 1) the composite implants were immediately placed in the defects. Some defects were left unfilled to confirm that the defect was critical sized. The inczision was closed with a continuous suture (4-0 nylon, Ethicon, Somerville, NJ). Animals had free access to food and water and were monitored daily in the postoperative period for any complications or abnormal behaviour.
The animals were sacrificed with an overdose of pentobarbital (150mg/kg intraperitoneal) and weighed after 14 weeks. The skull bone was dissected free and removed. Bones were wrapped in gauze moistened with physiologic PBS-solution and stored at -80°C until preparation for histomorphometrical examination.
Histomorphometry of bone formation
Skull bones were decalcified over 7 days in a 10% Tris buffered EDTA-solution under continuous stirring and embedded in paraffin in an established procedure of our lab. Sections (5µm) of the decalcified specimens parallel to the long axis of the head were stained with Hematoxylin and Eosin (H&E). All slides were analyzed using light microscopy (Axioobserver Z1, Zeiss, Göttingen, Germany) in combination with a computer-supported imaging picture analysis system (Axiovision 4.7; Zeiss, Göttingen, Germany). Histomorphometric assessments were performed by Image J to evaluate the new bone formation stained areas. Bone formation was counted in 6 non overlapping images/slide/animal surrounding the defect area and the mean value was calculated. These mean values were subsequently used for statistical analysis, which were examined in random order and blinded to the group setup.
STATISTICS
Results are presented as mean ± SD. Statistically significant differences were determined using F test (one way ANOVA and Post hoc Tests by SPSS program), statistical significance was achieved with p<0.05.
RESULTS
Characterization of MSC and EPC by FACS analysis showed a typical pattern of MSC (CD34-, CD45-, CD71+, CD73+, CD90+, CD105+) and EPC (CD31, vWF, VEGFR-2) surface markers. Osteogenic differentiation in MSC was observed using von Kossa staining, while the induction of endothelial differentiation in EPC was evidenced by DilDL uptake and binding of UEA-1-lectin.
Differentiation of rat Mesenchymal Stem Cells (MSC)
Using von Kossa staining [area%] differentiated MSC (33.05 ± 2.33) demonstrated a significant higher calcium deposition in comparison to undifferentiated MSC (2.33 ± 0.12; Figure 1A and B).
 Figure 1A: Images of von Kossa staining of undifferentiated MSC and differentiated MSC.
Figure 1A: Images of von Kossa staining of undifferentiated MSC and differentiated MSC.
Increased calcium deposition of differentiated MSC (right image) versus undifferentiated MSC (left image) obtained from the rat femur. Original magnification 50×, space bar indicates 200µm.
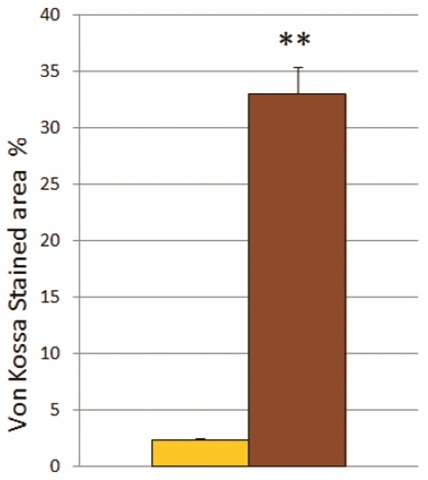 Figure 1B: Calcium deposition of undifferentiated MSC or differentiated MSC.
Figure 1B: Calcium deposition of undifferentiated MSC or differentiated MSC.
Analysis of von Kossa stained area [%] of undifferentiated MSC (yellow column) versus differentiated MSC (brown column).
** The mean difference is significant at the 0.01 level.
Adhesion of cells onto composite biomaterials
At 2hours after cell seeding onto various biomaterials no significant differences in cell adhesion of undifferentiated MSC+EPC (Figure 2, upper panel) or differentiated MSC+EPC, respectively (Figure 2, lower panel) were observed. Neither various biomaterials (PLA, BG20%; BG40%) influenced cell adhesion in vitro (Figure 2).
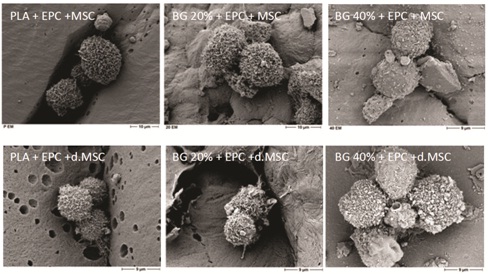 Figure 2: Cell adhesion and phenotype of MSC/EPC on the composite biomaterial.
Figure 2: Cell adhesion and phenotype of MSC/EPC on the composite biomaterial.
Surface characteristics and direct proof of undifferentiated MSC+EPC (upper panel) and differentiated MSC+EPC (lower panel) on various biomaterials: PLA (first column); PLA+BG 20% (second column) or PLA+BG 40% (third column), respectively by SEM. The scale bar indicates 9µm and 10µm, respectively
MSC and EPC viability
Viability of undifferentiated MSC+EPC and differentiated MSC+EPC, respectively was investigated on all tested biomaterials (PLA; PLA+BG20%; PLA+BG40%). We could not detect any difference in cell viability on day 1 according neither differentiation of cells nor biomaterials (Figure 3).
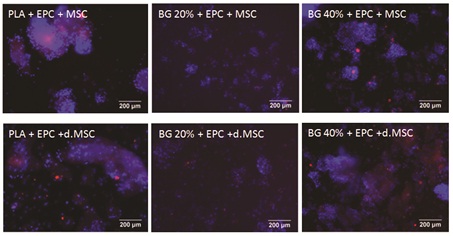 Figure 3: Fluorescence microscopy of co-cultured MSC and EPC onto composite biomaterials.
Figure 3: Fluorescence microscopy of co-cultured MSC and EPC onto composite biomaterials.
Fluorescence microscopy of co-cultured MSC and EPC adhering to three different biomaterials: PLA (first column), PLA +BG20% (second column), PLA+ -BG40% (third column). At day 1, staining of EPC (DiLDL, red) and cell nuclei of MSC and EPC (DAPI, blue), respectively, were performed. Endothelial-like differentiated cells were stained with both an orange-red and blue fluorescence, whereas cells without endothelial-like differentiation appear only blue, DAPI stained nucleus. The EPC uptake of DiLDL indicating endothelial cell differentiation.
The scale bar indicates 200µm.
Histology of bone formation
All specimens were included in this study at 14 weeks after surgery. No infection or delayed wound healing were observed.
When CSD remained empty, newly formed bone surrounded by an osteoid matrix rich in osteoblasts were only close to the borders of the surgical defect observed (Figure 4). The connective tissue in the central part of the defect was thinner than the original calvarium. It was well vascularized and rich in fibroplasts with oriented collagen fibers. Thus, in the histomorphometrical analysis (Figure 4) bone formed area [%] were evaluated in the empty defect (10.5 ± 4.2) of the skull bone. Cell based therapy with seeding of undifferentiated MSC+EPC to our three tested composites, only PLA+BG40% demonstrated a significant increase of bone formation (39.5 ± 12.1) compared to empty defect. PLA+MSC+EPC (24.6 ± 10.3) and PLA+BG20%+MSC+EPC (30.0 ± 8.1) showed bone formation with tendency to rise.
When CSD remained empty, newly formed bone surrounded by an osteoid matrix rich in osteoblasts were only close to the borders of the surgical defect observed (Figure 4). The connective tissue in the central part of the defect was thinner than the original calvarium. It was well vascularized and rich in fibroplasts with oriented collagen fibers. Thus, in the histomorphometrical analysis (Figure 4) bone formed area [%] were evaluated in the empty defect (10.5 ± 4.2) of the skull bone. Cell based therapy with seeding of undifferentiated MSC+EPC to our three tested composites, only PLA+BG40% demonstrated a significant increase of bone formation (39.5 ± 12.1) compared to empty defect. PLA+MSC+EPC (24.6 ± 10.3) and PLA+BG20%+MSC+EPC (30.0 ± 8.1) showed bone formation with tendency to rise.
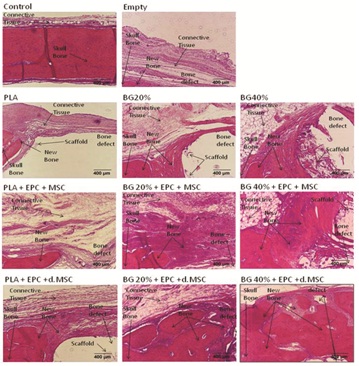 Figure 4: PLA+BG40%+dMSC/EPC increase bone formation in CSD after implantation.
Figure 4: PLA+BG40%+dMSC/EPC increase bone formation in CSD after implantation.
Representative images of a histological sections (H & E staining) of the Critical Size Defect (CSD) in calvarial rat bone were demonstrated. Due to the decalcification process, the BG was almost completely dissolved and left a gap corresponding to the BG area. At the border skull bone was detectable.
The scale bar indicates 400µm.
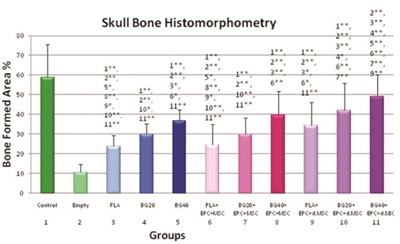 Figure 5: Quantitative analysis of Skull Bone Histomorphometry.
Figure 5: Quantitative analysis of Skull Bone Histomorphometry.
Bone formation area [%] in CSD increased significantly in animals when PLA+BG40 was used as biomaterials compared to PLA groups. Moreover significant bone formation area was evaluated in animals treated with PLA+ BG40%+differentiated MSC/EPC, compared to PLA+BG40% alone.
* indicates significant level (0.05)
** indicates high significant level (0.01) compared to group number x
DISCUSSION
In this study, we established a new developed bioglass-PLA-composite. Depending on the rate of burning, we created a new particle size of bioglass (106-125µm) for cell based therapy of a calvarial critical size defect in rats. Here we could demonstrate that PLA+BG40% seeded with EPC in coculture with differentiated MSC significantly improve bone formation in CSD.
Optimal biomaterials for bone tissue engineering should be biocompatible, biodegradable, possess an ideal porosity for cell attachment and cell integration, respectively as well as useful biomechanical stability. Single component materials do not meet all these requirements, thus composite biomaterials are needed.
PLA is highly biocompatible with a better thermal procedure, compared to other biopolymers. The main limitations of PLA are poor toughness, slow degradation and hydrophobic properties, which results in low cell affinity [25]. Pure bioglass is hard and brittle but offers a surface suitable for cell attachment. It is highly biodegradable and influences the local environment by releasing bioactive ions such as ionic calcium [26], which may lead to improved cellular responses at the implantation site [27].
Disadvantages of BG like lack of porosity occurred because it crystallizes during sintering. Recently, this has been overcome by understanding how the glass composition can be tailored to prevent crystallization [28]. Procedure developments have now allowed the production of bioactive glass polymer hybrids (composite of PLA and BG, e.g., PLA+BG40%) for bone regeneration which share load with bone and are not brittle under cyclic loads [26,29-32]. In several studies, bioactive glasses are reported to be able to induce the up-regulation of genes in bone cells and their effect in enhancing bone formation [33]. Due to their dissolution products bioactive glasses stimulating osteoprogenitor cells at the genetic level and bond with bone more rapidly than other bioceramics [11,34,35]. Moreover, early vascularization is a prerequisite for successful bone healing and Endothelial Progenitor Cells (EPC), seeded on appropriate biomaterials, can improve vascularization. In our former study, PLA+BG40% released the most calcium, and improved endothelial differentiation and vitality. This indicated that Ca2+ release improved EPC differentiation and enhanced early vascularization in critical size bone defects [36].
Interestingly in our present study, various BG content in PLA/BG composite (PLA; PLA+BG20%; PLA+BG40%) did not effect cell adhesion and cell viability in vitro when MSC and EPC were seeded on these biomaterials.
But, high concentrated PLA+BG40% demonstrate its osteogenic potential for bone formation. Moreover, pre-seeding this composite biomaterial (PLA+BG40%) with tissue-specific cells (MSC/EPC) prior to implant, especially when MSC are pre-differentiated, enhance bone formation significantly in vivo at 14 weeks compared to bioglass/PLA alone (cell free). This can be due both to the osteogenic and the vascular differentiation potential of MSC and EPC. In fact, cell-based therapy of MSC and EPC has been previously reported in literature in different studies [22,23,37-40] indicating a potential to provide vascularization for constructs used in bone regeneration. Our findings, that pre-differentiated MSC/EPC showed higher bone formation by trend, confirm these cell-based strategies.
These findings are in a line to Yu et al., [41]. It is one method which is being examined to improve bone tissue regeneration. According to safety of a bioglass-polylactic acid composite scaffold seeded with progenitor cells in a rat skull critical-size bone defect we observed in a previous study no side effects or complications [42].
One limitation of this study is that we observed at a very early time point for cell adhesion and viability, but according to our previous study it is possible to detect differences [5]. Also 2 hours after cell seeding is in this experimental setting more practical.
Cell transplantation onto an optimal biomaterial is a promising alternative to the ‘‘gold standard’’ of autologous bone grafting to stimulate bone repair even in this presented severely compromised model of bone healing. As known a skull defect model without bone marrow inside is a severely compromised model because recruitment of progenitor cells is more difficult.
Therefore, our data support the hypothesis that this new created bioglass/PLA composite is a useful biomaterial, which improve bone formation at a critical-sized bone defect.
This work provides important insights into the interaction between cell-based therapy (EPC/MSC) and the currently available PLA/Bioglass composites. This information can be valuable for choosing which substitute to use clinically and, more importantly, for further development of these and new materials.
Optimal biomaterials for bone tissue engineering should be biocompatible, biodegradable, possess an ideal porosity for cell attachment and cell integration, respectively as well as useful biomechanical stability. Single component materials do not meet all these requirements, thus composite biomaterials are needed.
PLA is highly biocompatible with a better thermal procedure, compared to other biopolymers. The main limitations of PLA are poor toughness, slow degradation and hydrophobic properties, which results in low cell affinity [25]. Pure bioglass is hard and brittle but offers a surface suitable for cell attachment. It is highly biodegradable and influences the local environment by releasing bioactive ions such as ionic calcium [26], which may lead to improved cellular responses at the implantation site [27].
Disadvantages of BG like lack of porosity occurred because it crystallizes during sintering. Recently, this has been overcome by understanding how the glass composition can be tailored to prevent crystallization [28]. Procedure developments have now allowed the production of bioactive glass polymer hybrids (composite of PLA and BG, e.g., PLA+BG40%) for bone regeneration which share load with bone and are not brittle under cyclic loads [26,29-32]. In several studies, bioactive glasses are reported to be able to induce the up-regulation of genes in bone cells and their effect in enhancing bone formation [33]. Due to their dissolution products bioactive glasses stimulating osteoprogenitor cells at the genetic level and bond with bone more rapidly than other bioceramics [11,34,35]. Moreover, early vascularization is a prerequisite for successful bone healing and Endothelial Progenitor Cells (EPC), seeded on appropriate biomaterials, can improve vascularization. In our former study, PLA+BG40% released the most calcium, and improved endothelial differentiation and vitality. This indicated that Ca2+ release improved EPC differentiation and enhanced early vascularization in critical size bone defects [36].
Interestingly in our present study, various BG content in PLA/BG composite (PLA; PLA+BG20%; PLA+BG40%) did not effect cell adhesion and cell viability in vitro when MSC and EPC were seeded on these biomaterials.
But, high concentrated PLA+BG40% demonstrate its osteogenic potential for bone formation. Moreover, pre-seeding this composite biomaterial (PLA+BG40%) with tissue-specific cells (MSC/EPC) prior to implant, especially when MSC are pre-differentiated, enhance bone formation significantly in vivo at 14 weeks compared to bioglass/PLA alone (cell free). This can be due both to the osteogenic and the vascular differentiation potential of MSC and EPC. In fact, cell-based therapy of MSC and EPC has been previously reported in literature in different studies [22,23,37-40] indicating a potential to provide vascularization for constructs used in bone regeneration. Our findings, that pre-differentiated MSC/EPC showed higher bone formation by trend, confirm these cell-based strategies.
These findings are in a line to Yu et al., [41]. It is one method which is being examined to improve bone tissue regeneration. According to safety of a bioglass-polylactic acid composite scaffold seeded with progenitor cells in a rat skull critical-size bone defect we observed in a previous study no side effects or complications [42].
One limitation of this study is that we observed at a very early time point for cell adhesion and viability, but according to our previous study it is possible to detect differences [5]. Also 2 hours after cell seeding is in this experimental setting more practical.
Cell transplantation onto an optimal biomaterial is a promising alternative to the ‘‘gold standard’’ of autologous bone grafting to stimulate bone repair even in this presented severely compromised model of bone healing. As known a skull defect model without bone marrow inside is a severely compromised model because recruitment of progenitor cells is more difficult.
Therefore, our data support the hypothesis that this new created bioglass/PLA composite is a useful biomaterial, which improve bone formation at a critical-sized bone defect.
This work provides important insights into the interaction between cell-based therapy (EPC/MSC) and the currently available PLA/Bioglass composites. This information can be valuable for choosing which substitute to use clinically and, more importantly, for further development of these and new materials.
REFERENCES
- De Long WG Jr, Einhorn TA, Koval K, McKee M, Smith W, et al. (2007) Bone grafts and bone graft substitutes in orthopaedic trauma surgery. A critical analysis. J Bone Joint Surg Am 89: 649-658.
- Laurencin CT, Khan Y, El-Amin SF (2006) Bone graft substitutes. Expert Rev Med Devices 1: 49-57.
- Mroz TE, Joyce MJ, Lieberman IH, Steinmetz MP, Benzel EC, et al. (2009) The use of allograft bone in spine surgery: is it safe? Spine J 9: 303-308.
- Vacanti CA, Bonassar LJ (1999) An overview of tissue engineered bone. Clin Orthop Relat Res 367: 375-381.
- Seebach C, Schultheiss J, Wilhelm K, Frank J, Henrich D (2010) Comparison of six bone-graft substitutes regarding to cell seeding efficiency, metabolism and growth behaviour of human Mesenchymal Stem Cells (MSC) in vitro. Injury 41: 731-738.
- Schultheiss J, Seebach C, Henrich D, Wilhelm K, Barker JH, et al. (2011) Mesenchymal Stem Cell (MSC) and Endothelial Progenitor Cell (EPC) growth and adhesion in six different bone graft substitutes. Eur J Trauma Emerg Surg 37: 635-644.
- Blaker JJ, Nazhat SN, Maquet V, Boccaccini AR (2011) Long-term in vitro degradation of PDLLA/Bioglass bone scaffolds in a cellular simulated body fluid. Acta Biomater 7: 829-840.
- Fu Q, Rahaman MN, Fu H, Liu X (2010) Silicate, borosilicate, and borate bioactive glass scaffolds with controllable degradation rate for bone tissue engineering applications. I. Preparation and in vitro degradation. J Biomed Mater Res A 95:164-171.
- Hench LL (1998) Bioceramics. J Am Ceram Soc 81:1705-1728.
- Salinas AJ, Vallet-Regi M, Izquierdo-Barba I (2001) Biomimetic Apatite Deposition on Calcium Silicate Gel Glasses. Journal of Sol-Gel Science and Technology 21: 13-25.
- Xynos ID, Edgar AJ, Buttery LDK, Hench LL, Polak M (2001) Gene expression profiling of human osteoblasts following treatment with the ionic products of Bioglass 45S5 dissolution. J Biomed Mater Res 55: 151-157.
- Leach JK, Kaigler D, Wang Z, Krebsbach PH, Mooney DJ (2006) Coating of VEGF-releasing scaffolds with bioactive glass for angiogenesis and bone regeneration. Biomaterials 27: 3249-3255.
- Day RM (2005) Bioactive glass stimulates the secretion of angiogenic growth factors and angiogenesis in vitro. Tissue Eng 11: 768-777.
- Leu A, Leach JK (2008) Proangiogenic potential of a collagen-bioactive glass substrate. Pharm Res 25:1222-1229.
- Day RM, Boccaccini AR, Shurey S, Roether JA, Forbes A, et al. (2004) Assessment of polyglycolic acid mesh and bioactive glass for soft-tissue engineering scaffolds. Biomaterials 25: 5857-5866.
- Valliant EM, Jones JR (2011) Softening bioactive glass for bone regeneration: sol-gel hybrid materials. Soft Matter 7: 5083-5095.
- Jones JR, Ehrenfried LM, Hench LL (2006) Optimising bioactive glass scaffolds for bone tissue engineering. Biomaterials 27: 964-973.
- Peitl O, Zanotto ED, Serbena FC, Hench LL (2012) Compositional and microstructural design of highly bioactive P2O5-Na2O-CaO-SiO2 glass-ceramics. Acta Biomater 8: 321-332.
- Vacanti JP (2000) Structure tissue engineering. California: Academic Press 671-682.
- Middleton JC, Tipton AJ (2000) Synthetic biodegradable polymers as orthopedic devices. Biomaterials 21: 2335-2346.
- Boccaccini AR, Blaker JJ (2005) Bioactive composite materials for tissue engineering scaffolds. Expert Rev Med Devices 2: 307-317.
- Seebach C, Henrich D, Kahling C, Wilhelm K, Tami AE, et al. (2010) Endothelial progenitor cells and mesenchymal stem cells seeded onto beta-TCP granules enhance early vascularization and bone healing in a critical-sized bone defect in rats. Tissue Eng Part A 16: 1961-1970.
- Seebach C, Henrich D, Wilhelm K, Barker JH, Marzi I (2012) Endothelial progenitor cells improve directly and indirectly early vascularization of mesenchymal stem cell-driven bone regeneration in a critical bone defect in rats. Cell Transplant 21: 1667-1677.
- Henrich D, Seebach C, Kaehling C, Scherzed A, Wilhelm K, et al. (2009) Simultaneous cultivation of human endothelial-like differentiated precursor cells and human marrow stromal cells on beta-tricalcium phosphate. Tissue Eng Part C Methods 15: 551-560.
- Rasal RM, Janokar AV, Hirt DE (2010) Poly (lactic acide) modifications. Prog Polymer Sci 35: 338-356.
- Jones JR, Lin S, Yue S, Lee PD, Hanna JV, et al. (2010) Bioactive glass scaffolds for bone regeneration and their hierarchical characterisation. Proc Inst Mech Eng H 224: 1373-1387.
- Gorustovich AA, Roether JA, Boccaccini AR (2010) Effect of bioactive glasses on angiogenesis: a review of in vitro and in vivo evidences. Tissue Eng Part B Rev 16: 199-207.
- el-Ghannam A, Ducheyne P, Shapiro IM (1995) Bioactive material template for in vitro synthesis of bone. J Biomed Mater Res 29: 359-370.
- Jones JR (2013) Review of bioactive glass: from Hench to hybrids. Acta Biomater 9: 4457-4586.
- Liu A, Hong Z, Zhuang X, Chen X, Cui Y, et al. (2008) Surface modification of bioactive glass nanoparticles and the mechanical and biological properties of poly(L-lactide) composites. Acta Biomater 4: 1005-1015.
- Maquet V, Boccaccini AR, Pravata L, Notingher I, Jérôme R (2004) Porous poly(alpha-hydroxyacid)/Bioglass composite scaffolds for bone tissue engineering. I: Preparation and in vitro characterisation. Biomaterials 25: 4185-4194.
- Blaker JJ, Gough JE, Maquet V, Notingher I, Boccaccini AR (2003) In vitro evaluation of novel bioactive composites based on Bioglass-filled polylactide foams for bone tissue engineering scaffolds. J Biomed Mater Res A 67: 1401-1411.
- Bi L, Jung S, Day D, Neidig K, Dusevich V, et al. (2012) Evaluation of bone regeneration, angiogenesis, and hydroxyapatite conversion in critical-sized rat calvarial defects implanted with bioactive glass scaffolds. J Biomed Mater Res Part A 100: 3267-3275.
- Chen QZ, Thompson ID, Boccaccini AR (2006) 45S5 Bioglass-derived glass-ceramic scaffolds for bone tissue engineering. Biomaterials 27: 2414-2425.
- Hoppe A, Güldal NS, Boccaccini AR (2011) A review of the biological response to ionic dissolution products from bioactive glasses and glass-ceramics. Biomaterials 32: 2757-2774.
- Eldesoqi K, Seebach C, Nguyen Ngoc C, Meier S, Nau C, et al. (2013) High calcium bioglass enhances differentiation and survival of endothelial progenitor cells, inducing early vascularization in critical size bone defects. PLoS One 8: 79058.
- Henrich D, Wilhelm K, Warzecha J, Frank J, Barker J, et al. (2013) Human endothelial-like differentiated precursor cells maintain their endothelial characteristics when cocultured with mesenchymal stem cell and seeded onto human cancellous bone. Mediators Inflamm 364:591.
- Duttenhoefer F, Lara de Freitas R, Meury T, Loibl M, Benneker LM, et al. (2013) 3D scaffolds co-seeded with human endothelial progenitor and mesenchymal stem cells: evidence of prevascularisation within 7 days. Eur Cell Mater 26: 49-64.
- Rozen N, Bick T, Bajayo A, Shamian B, Schrift-Tzadok M, et al. (2009) Transplanted blood-derived Endothelial Progenitor Cells (EPC) enhance bridging of sheep tibia critical size defects. Bone 45: 918-924.
- Kaigler D, Krebsbach PH, Wang Z, West ER, Horger K, et al. (2006) Transplanted endothelial cells enhance orthotopic bone regeneration. J Dent Res 85: 633-637.
- Yu H, Vandevord PJ, Gong W, Wu B, Song Z, et al. (2008) Promotion of osteogenesis in tissue-engineered bone by pre-seeding endothelial progenitor cells-derived endothelial cells. J Orthop Res 26: 1147-1152.
- Eldesoqi K, Henrich D, El-Kady AM, Arbid MS, Abd El-Hady BM, et al. (2014) Safety evaluation of a bioglass-polylactic acid composite scaffold seeded with progenitor cells in a rat skull critical-size bone defect. PLoS One 9: 87642.
Citation: Thorpe AA, Sammon C, Le Maitre CL (2015) ‘Cell or Not to Cell’ that is the Question: For Intervertebral Disc Regeneration? J Stem Cell Res Dev Ther 2: 004.
Copyright: © 2015 Karam Eldesoqi, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

Journal Highlights
© 2026, Copyrights Herald Scholarly Open Access. All Rights Reserved!
