
The Effect Gestational Diabetes has on DNA Methylation as Related to Fetal Development and the Outcomes of the Mother and Offspring
*Corresponding Author(s):
Michael P Sarras JrDepartment Of Cell Biology And Anatomy, Rosalind Franklin University Of Medicine, Illinois, United States
Tel:+1 5204054739,
Email:michael.sarras@rosalindfranklin.edu
Abstract
In the current review we will discuss the relationship of Gestational Diabetes (GDM) to DNA methylation and relate this epigenetic process to fetal development and the outcomes of offspring of mothers with GDM as well as the outcome of mothers who experience GDM. The article is organized into four sections that include: 1) DNA methylation in the placenta and placental cord, 2) Effects of gestational diabetes on DNA methylation patterns and particular genes and cell types in offspring, 3) Effects of gestational diabetes on the growth and weight of offspring, and 4) Potential epigenetic biomarkers for predicting gestational diabetes and the occurrence of T2DM following GDM. In summary, these studies indicate 1) A significant occurrence of DNA methylation in fetal placental tissues and cord blood, 2) The occurrence of specific genes known to be related to the on-set and continuance of diabetes in mothers with GDM and their offspring, 3) The occurrence in GDM of DNA methylation patterns affecting genes that control fetal growth and the weight of offspring, and 4) The potential identification of biomarkers that can be used to predict the on-set of GDM in mothers and the later on-set of T2DM in both GDM mothers and their offspring.
Keywords
ABBREVIATIONS
GDM: Gestational Diabetes
DM: Diabetes Mellitus
T2DM: Type-2 Diabetes Mellitus
INTRODUCTION
A maternal form of Diabetes Mellitus (DM) is Gestational Diabetes (GDM) that affects approximately 6% of pregnant women in the United States [1] and can later lead to DM of both the affected mothers [2-4] and their offspring [4-8]. DM [9] in general often results in long term complications [10] stemming from metabolic dysregulation [11,12]. Clinical trials have established that once initiated, these complications persist and continue to progress unimpeded even when glycemic control is achieved through pharmaceutical intervention; a phenomenon termed, “Metabolic Memory” (MM) [13-16] The mechanism(s) of MM have been examined via both animal model approaches and in vitro based studies [17-23]. These studies indicate that the initial Hyperglycemia (HG) results in permanent aberrant gene expression in DM target tissues such as the general cardiovascular system, renal system, and retina; to name just a few. The ability to sustain these complications in the absence of HG invokes a role for the epigenome to perpetuate tissue dysfunction. While extensive epigenetic research has been conducted regarding histone modifications [24-27] and micro RNA mechanisms [28-34], less is known about the role of HG-induced persistent DNA methylation changes; although data from animal models and humans indicate that aberrant DNA methylation does occur in DM & GDM. Moreover, the HG environment induces changes in specific cells types, such as Endothelial Cells (EC) involving structural, metabolic, and functional alterations as in blood vessel formation [10,35-37]. This is of particular significance because impaired blood vessel formation is a common pathology associated with all organ and tissues systems affected in DM and GDM.
In the current review we will discuss the relationship of gestational diabetes to DNA methylation and relate this epigenetic process to fetal development and the outcomes of offspring of mothers with GDM as well as the outcome of mothers who experience GDM. The articles are organized into four sections that include: 1) DNA methylation in the placenta and placental cord, 2) Effects of gestational diabetes on DNA methylation patterns and particular genes and cell types in offspring, 3) Effects of gestational diabetes on the growth and weight of offspring, and 4) Identification of potential epigenetic biomarkers for predicting gestational diabetes and the occurrence of T2DM following GDM.
DISCUSSION
DNA methylation in the placental cotyledon tissue, placental cord blood, and maternal blood of GDM mothers
Summarizing the results of groups 1-5 one finds the common features of GDM to include: 1) Differential global DNA methylation, 2) Methylation sites observed to involve both hypermethylation and hypomethylation of CpG nucleotides, and 3) A broad array of gene classes that all appear to have some tie to diabetes. The effect of these genes on the later on-set of T2DM remains to be determined, but animal model studies indicate that once DNA methylation occurs it is not fully reversed [21,46-48].
As an extension of the above mentioned studies, a number of studies have focused on the effects of DNA methylation on particular genes as analyzed by DNA sequence analysis from the placenta, cord blood, and maternal blood or tissues [3,45,49-54] (Table 1). These studies have highlighted the importance of the following genes related to the potential on-set of DM (typically T2DM in the case of GDM) [3,45,49-54]: 1) MEST gene [49], 2) The NPR1, PANK1, SCAND1, GJA4, PYGO1 and CLN8 [50] genes, 3) DNA methyltransferases to include; DNMT1, DNMT3A, and DNMT3B [3], 4) GNAS and IGF2 [52], 5) Placental Lipoprotein Lipase (LPL) [51], 6) ZFP57,TBR1, DGKZ, AGPAT1 and MAPK10 [45], 7) The studies of Chen et al. [53], identified 29 genes and 10 intergenic regions with methylated regions in the blood of offspring of mothers with GDM, and 8) PGC-1 and PDX1 [54]. These genes showed either hypermethylation or hypomethylation as a consequence of the hyperglycemic environment created in mothers with GDM. However, whether these genes directly contribute to the on-set of DM in mothers with GDM or their offspring is unclear. As stated above however, animal model studies indicate that once DNA methylation occurs it is not fully reversed [21,46-48] thereby creating a link between this epigenetic event and the future on-set of diabetes.
|
Sources of Tissue |
||
|
Placental tissue, cord blood, and maternal blood or tissues with observed DNA methylation |
Placental cord blood, placental cotyledon tissue, and mother's adipose tissue (from C-Section) as related to obesity and childhood weight |
Peripheral blood of offspring and mothers |
|
NPR1, PANK1, SCAND1, GJA4, PYGO1, CLN8, DNMT1, DNMT3A, and DNMT3B GNAS, IGF2, LPL, ZFP57,TBR1, DGKZ, AGPAT1, MAPK10, PGC-la and PDX1 |
POU2F1, PKHD1, NFE2, NHLH2, AGTR2, and LEP |
COPSE, PIL3R5, HAAO, CCDC124, C5 or f34, methylated H3K27 and H3K4 (both histones), CTNND2, HNF4A, and RREB1 |
|
Endothelial cells derived from umbilical cord or cord blood with observed DNA methylation |
||
|
1) Growth factors and proteins linked to insulin sensing (IGFBP3, IGFBP5, IRS1, SIRT1, mTOR); 2) Factors involved in ECM reorganization (TGFB2, FBN1, FBN2, COL1A1, COL3A1, DDR2, FN1, FST, FST13, CARD11, LIPG, ICAM1); 3) Non-insulin growth factors (FGF2, PDGFbR); 4) Cell cycle genes (CCND2, CDKN2B); and 5) Cell proliferation, survival, autophagy, and epithelial-to-mesenchyma I transition (PLAC8) |
||
Effects of gestational diabetes on particular genes and particular cell types in the offspring of GDM mothersAs stated in the introduction, Gestational diabetes can later lead to DM of the T2-form of both the affected mothers [2-4] and their offspring [4-8]. If T2DM does occur it often results in long term complications [10] affecting a broad spectrum of organs and tissues of the body because of the sustained metabolic dysregulation occurring in the disease [11,12]. A significant cell type targeted in the long term complications of the disease is the Endothelial Cell (EC) [10,35-37]. The hyperglycemic environment of GDM and DM induces changes in the EC involving structural, metabolic, and functional alterations as seen in the impairment of blood vessel formation [10,35-37]. Blood vessel formation is of particular importance because its impairment directly underlies many of the pathology’s seen following the initial hyperglycemic episodes. A few studies have directly studied endothelial cells under the hyperglycemic conditions of GDM (Table 1). For example, Ambra et al. [55], have performed transcriptome analysis of Human Primary Endothelial Cells (HUVEC) from umbilical cords of gestational diabetic mothers and have identified candidate sites for an epigenetic modulation of specific gene expression in these cells. They reported that four categories of gene classes that were affected by the hyperglycemic GDM environment. These four categories included: 1) Growth factors and proteins linked to insulin sensing (IGFBP3, IGFBP5, IRS1, SIRT1, mTOR); 2) Factors involved in ECM reorganization (TGFB2, FBN1, FBN2, COL1A1, COL3A1, DDR2, FN1, FST, FSTL3, CARD11, LIPG, ICAM1); 3) Non-insulin growth factors (FGF2, PDGFbR); and 4) Cell cycle genes (CCND2, CDKN2B). They interconnect these four groups into a scheme that proposes that there is a functional interplay between the groups that involves:
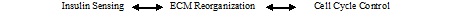
Blue et al. [56], have studied epigenetic altered function of endothelial colony-forming cells (endothelial progenitor cells) in GDM. They specifically studied the effects Placenta-Specific 8 gene (PLAC8) had on DNA methylation. PLAC8 is also known as onzin. PLAC8 was originally identified as a placental-enriched protein but later studies showed that PLAC8 was also expressed in epithelial cells, adipocytes, and hematopoietic cells [57-61]. Although the precise endogenous biochemical function of PLAC8 is unclear, more recent data indicates that it regulates adipocyte differentiation, the innate immune response, and cell proliferation/survival processes [57,60,61]. Additional studies demonstrated a role for PLAC8 in promoting tumorigenesis; possibly via mechanisms involving proliferation, survival, autophagy, and epithelial-to-mesenchymal transition [57-59,62]. For these studies, endothelial progenitor cells were obtained and cultured from isolated umbilical cord blood cells. Blue et al. [56], found that PLAC8 over expression functioned as a protective mechanism for endothelial colony-forming cells to avoid senescence. Given that hyperglycemia of GDM enhances endothelial colony-forming cells senescence and impairs vasculogenesis, their findings indicated an adaptive response of fetal endothelial colony-forming cells to circumvent the negative effects of a hyperglycemic environment. In addition, differential methylation was detected in endothelial colony-forming cells from GDM mothers in the first intron of an isoform of PLAC8. Moreover, the methylation of several individual CpG sites in these regions was seen to be negatively correlated with PLAC8 mRNA expression. This suggested a mechanistic link which the authors proposed was due to altered transcription factor binding. The authors conclude that their data indicates that neonatal endothelial progenitor cells of GDM mothers leads to altered gene expression and DNA methylation due to the hyperglycemic environment present during GDM. This would then open the possibility that altered epigenetic regulation had occurred. A cautionary note in regard to these studies concerns one aspect of the experimental protocol. Because the investigators cultured endothelial colony-forming cells for subsequent analysis, this opens the possibility that altered DNA methylation occurred due to in vitro culturing conditions; a process that in itself is suspected to induce DNA methylation.Effects of gestational diabetes on the growth and weight of offspringA number of studies have focused on the effects of GDM-induced DNA methylation on fetal growth and newborn weigh [8,63-68] (Table 1). The majority of these studies have analyzed placental cord blood [8,63-65] while two studied both cord blood and placental cotyledon tissue [66,67]. These studies shared the finding that restricted fetal growth and offspring weight correlated with changed DNA methylation patterns in genes associated with growth control such as POU2F1, PKHD1, NFE2, NHLH2, and AGTR2 [8]. Complementing these studies were the findings of Lesseur et al., who found in analysis of placental cotyledon tissue significant changes in the DNA methylation pattern of the Leptin gene [66]. Leptin is important to energy homeostasis and functions as a satiety signal. During pregnancy, leptin is produced by the placenta and has pleiotropic functions that include regulation of growth and nutrient exchange [69]. Finally, Deng et al. [68], analyzed adipose tissue of control and GDM women obtained during C-section births. They found that genes related to obesity control in adipose tissue were significantly methylated. They concluded that fetuses of GDM mothers had a higher potential for childhood obesity than those of non-GDM mothers.Potential epigenetic biomarkers for predicting gestational diabetes and the occurrence of T2DM following GDMRecent studies have attempted to identify epigenetic biomarkers that can be used to predict the on-set of T2DM following the occurrence of GDM [70-72] (Table 1). The studies of Wu et al. [70], stand out because they identified maternal DNA methylation changes prior to the diagnosis of GDM; thereby pointing to specific genes (COPS8, PIL3R5, HAAO, CCDC124, and C5 or f34) that may be used as biomarkers for the on-set of GDM prior to its clinical diagnosis. Studying the epigenetic process of histone methylation, as opposed to DNA methylation, Michalczyk et al. [71], studied the blood of GDM versus non-GDM mothers and determined that methylated H3K27 was significantly lower at 8-10 and 20 weeks postpartum in women with GDM who developed T2DM, compared with nondiabetic women. Likewise methylated H3K4 was significantly lower at 8-10 weeks postpartum in women with GDM who later developed T2DM compared with women who had GDM who did later develop the disease. They concluded that because the percentage of methylated H3K27 and H3K4 varied with diabetic state; it was a potential predictive biomarker for the identification of women who will later development T2DM after experiencing GDM. Finally, the studies of Kim et al. [72], focused on DNA methylation analysis in sibling pairs who were either exposed to in utero GDM or were not exposed to in utero GDM. Analysis of their data indicated that several suggestive epigenetic markers could be identified. For example, one CpG site, cg04988367, which was located 39 kb downstream of Catenin Delta 2 (CTNND2) showed a significant difference. An additional 11 CpG sites also had significant methylation differences. This later group included cg08407434 which was positioned at the intron of Hepatocyte Nuclear Factor 4A (HNF4A). This gene is known to be related to maturity on-set diabetes of the young. Another CpG marker, cg18255813, was located at intron of Ras Responsive Element Binding protein 1 (RREB1), which was reported to be associated with visceral fat mass and glycemic traits.
All of these studies suggest that GDM related biomarkers exist, but extensive studies must be completed to firmly establish any one of them as true biomarkers for GDM.
SUMMARY AND CONCLUSION
The literature clearly established that DNA methylation is induced by gestational diabetes as analyzed in placental cotyledon tissue and cord blood. The DNA methylation patterns affect both exons and introns and are observed as both hypermethylation and hypomethylation of CpG dinucleotides. The genes affected by this epigenetic process include a broad spectrum of functional groups involved in such pathways as metabolic processes related to insulin control and subsequent diabetic dysregulation, growth control, and childhood obesity. Finally, recent studies have begun to identify potential biomarkers for the prediction of those women who may develop GDM and the later on-set of T2D in GDM mothers and their offspring.In regard to the effect of DNA methylation on gene/protein function we propose that these epigenetic changes affect regulatory regions upstream of genes that cause these genes to either up or down regulate depending on whether hypermethylation or hypomethylation has occurred. This would result in dysfunction of tissues/organs in the long term metabolic memory state because of these alterations in gene expression patterns.
ACKNOWLEDGMENT
The authors wishes to express their appreciation to the National Institutes of Health, USA (DK092721) for funds that supported preparation and writing of this review and to Dr. Robert Intine who provided valued discussions along the course of the writing of this review.
CONFLICT OF INTEREST
The authors have no conflict of interests or commercial interests as related to the information provided in this review.
REFERENCES
- Mack LR, Tomich PG (2017) Gestational diabetes: Diagnosis, classification, and clinical care. Obstet Gynecol Clin North Am 44: 207-217.
- Baz B, Riveline JP, Gautier JF (2016) Endocrinology of pregnancy: Gestational diabetes mellitus: Definition, aetiological and clinical aspects. Eur J Endocrinol 174: 43-51.
- Yan J, Yang H (2014) Gestational diabetes mellitus, programing and epigenetics. J Matern Fetal Neonatal Med 27: 1266-1269.
- Alexander J, Teague AM, Chen J, Aston CE, Leung YK, et al. (2018) Offspring sex impacts DNA methylation and gene expression in placentae from women with diabetes during pregnancy. PLoS One 13.
- Haertle L, Hajj NE, Dittrich M, Muller T, Nanda I, et al. (2017) Epigenetic signatures of gestational diabetes mellitus on cord blood methylation. Clin Epigenetics 9: 28.
- Rong C, Cui X, Chen J, Qian Y, Jia R et al. (2015) DNA methylation profiles in placenta and its association with gestational diabetes mellitus. Exp Clin Endocrinol Diabetes 123: 282-288.
- Januar V, Desoye G, Novakovic B, Cvitic S, Saffery R (2015) Epigenetic regulation of human placental function and pregnancy outcome: Considerations for causal inference. Am J Obstet Gynecol 213: 182-196.
- Quilter CR, Cooper WN, Cliffe KM, Skinner BM, Prentice PM, et al. (2014) Impact on offspring methylation patterns of maternal gestational diabetes mellitus and intrauterine growth restraint suggest common genes and pathways linked to subsequent type 2 diabetes risk. FASEB J 28: 4868-4879.
- Shaw JE, Sicree RA, Zimmet PZ (2010) Global estimates of the prevalence of diabetes for 2010 and 2030. Diabetes Res Clin Pract 87: 4-14.
- Inzucchi S, Majumdar S (2015) Glycemic targets: What is the evidence? Med Clin North Am 99: 47-67.
- Brownlee M (2005) The pathobiology of diabetic complications: A unifying mechanism. Diabetes 54: 1615-1625.
- Baynes JW (1991) Role of oxidative stress in development of complications in diabetes. Diabetes 40: 405-412.
- Riddle MC (2010) Effects of intensive glucose lowering in the management of patients with type 2 diabetes mellitus in the Action to Control Cardiovascular Risk in Diabetes (ACCORD) trial. Circulation 122: 844-846.
- Skyler JS, Bergenstal R, Bonow RO, Buse J, Deedwania P, et al. (2009) Intensive glycemic control and the prevention of cardiovascular events: Implications of the ACCORD, ADVANCE, and VA diabetes trials. Diabetes Care 32: 187-192.
- Ismail-Beigi F, Craven T, Banerji MA, Basile J, Calles J, et al. (2010) Effect of intensive treatment of hyperglycaemia on microvascular outcomes in type 2 diabetes: An analysis of the ACCORD randomised trial. Lancet 376: 419-430.
- Intine RV, Sarras MP Jr (2012) Metabolic memory and chronic diabetes complications: Potential role for epigenetic mechanisms. Curr Diab Rep 12: 551-559.
- Roy S, Sala R, Cagliero E, Lorenzi M (1990) Overexpression of fibronectin induced by diabetes or high glucose: Phenomenon with a memory. Proc Natl Acad Sci U S A 87: 404-408.
- Kowluru RA, Chakrabarti S, Chen S (2004) Re-institution of good metabolic control in diabetic rats and activation of caspase-3 and nuclear transcriptional factor (NF-kappaB) in the retina. Acta Diabetol 41: 194-199.
- Hammes HP, Klinzing I, Wiegand S, Bretzel RG, Cohen AM, et al. (1993) Islet transplantation inhibits diabetic retinopathy in the sucrose-fed diabetic Cohen rat. Invest Ophthalmol Vis Sci 34: 2092-2096.
- Engerman RL, Kern TS (1987) Progression of incipient diabetic retinopathy during good glycemic control. Diabetes 36: 808-812.
- Olsen AS, Sarras MP Jr, Leontovich A, Intine RV (2012) Heritable transmission of diabetic metabolic memory in zebrafish correlates with DNA hypomethylation and aberrant gene expression. Diabetes 61: 485-491.
- Kowluru RA (2003) Effect of reinstitution of good glycemic control on retinal oxidative stress and nitrative stress in diabetic rats. Diabetes 52: 818-823.
- Li SL, Reddy MA, Cai Q, Meng L, Yuan H, et al. (2006) Enhanced proatherogenic responses in macrophages and vascular smooth muscle cells derived from diabetic db/db mice. Diabetes 55: 2611-2619.
- Zhong Q, Kowluru RA (2011) Epigenetic changes in mitochondrial superoxide dismutase in the retina and the development of diabetic retinopathy. Diabetes 60: 1304-1313.
- Zhong Q, Kowluru RA (2010) Role of histone acetylation in the development of diabetic retinopathy and the metabolic memory phenomenon. J Cell Biochem 110: 1306-1313.
- Brasacchio D, Okabe J, Tikellis C, Balcerczyk A, George P, et al. (2009) Hyperglycemia induces a dynamic cooperativity of histone methylase and demethylase enzymes associated with gene-activating epigenetic marks that coexist on the lysine tail. Diabetes 58: 1229-1236.
- Yu J, Auwerx J (2010) Protein deacetylation by SIRT1: An emerging key post-translational modification in metabolic regulation. Pharmacol Res 62: 35-41.
- Prattichizzo F, Giuliani A, Ceka A, Rippo MR, Bonfigli AR, et al. (2015) Epigenetic mechanisms of endothelial dysfunction in type 2 diabetes. Clin. Epigenetics 7: 56.
- Putta S, Lanting L, Sun G, Lawson G, Kato M, et al. (2012) Inhibiting microRNA-192 ameliorates renal fibrosis in diabetic nephropathy. J Am Soc Nephrol 23: 458-469.
- Wang B, Koh P, Winbanks C, Coughlan MT, McClelland A, et al. (2011) miR-200a prevents renal fibrogenesis through repression of TGF-β2 expression. Diabetes 60: 280-287.
- Kato M, Zhang J, Wang M, Lanting L, Yuan H (2007) MicroRNA-192 in diabetic kidney glomeruli and its function in TGF-beta-induced collagen expression via inhibition of E-box repressors. Proc Natl Acad Sci U S A 104: 3432-3437.
- McArthur K, Feng B, Wu Y, Chen S, Chakrabarti S (2011) MicroRNA-200b regulates vascular endothelial growth factor-mediated alterations in diabetic retinopathy. Diabetes 60: 1314-1323.
- Feng B, Chen S, McArthur K, Wu Y, Sen S, et al. (2011) miR-146a-Mediated extracellular matrix protein production in chronic diabetes complications. Diabetes 60: 2975-2984.
- Ruan Q, Wang T, Kameswaran V, Wei Q, Johnson DS, et al. (2011) The microRNA-21-PDCD4 axis prevents type 1 diabetes by blocking pancreatic beta cell death. Proc Natl Acad Sci U S A 108: 12030-12035.
- Costa PZ, Soares R (2013) Neovascularization in diabetes and its complications. Unraveling the angiogenic paradox. Life Sci 92: 1037-1045.
- Carmeliet P (2005) Angiogenesis in life, disease and medicine. Nature 438: 932-936.
- Menegazzo L, Albiero M, Avogaro A, Fadini GP (2012) Endothelial progenitor cells in diabetes mellitus. Biofactors 38: 194-202.
- Reichetzeder C, Dwi Putra SE, Pfab T, Slowinski T, Neuber C, et al. (2016) Increased global placental DNA methylation levels are associated with gestational diabetes. Clin Epigenetics 8: 82.
- Weng X, Liu F, Zhang H, Kan M, Wang T, et al. (2018) Genome-wide DNA methylation profiling in infants born to gestational diabetes mellitus. Diabetes Res Clin Pract 142: 10-18.
- Haertle L, Hajj NE, Dittrich M, Muller T, Nanda I, et al. (2017) Epigenetic signatures of gestational diabetes mellitus on cord blood methylation. Clin Epigenetics 9: 28.
- Nomura Y, Lambertini L, Rialdi A, Lee M, Mystal EY, et al. (2014) Global methylation in the placenta and umbilical cord blood from pregnancies with maternal gestational diabetes, preeclampsia, and obesity. Reprod Sci 21: 131-137.
- Finer S, Mathews C, Lowe R, Smart M, Hillman S, et al. (2015) Maternal gestational diabetes is associated with genome-wide DNA methylation variation in placenta and cord blood of exposed offspring. Hum Mol Genet 24: 3021-3029.
- Kang J, Lee CN, Li HY, Hsu KH, Lin SY (2017) Genome-wide DNA methylation variation in maternal and cord blood of gestational diabetes population. Diabetes Res Clin Pract 132: 127-136.
- Enquobahrie DA, Moore A, Muhie S, Tadesse MG, Lin S, et al. (2015) Early pregnancy maternal blood DNA methylation in repeat pregnancies and change in gestational diabetes mellitus status-A pilot study. Reprod Sci 22: 904-910.
- Petropoulos S, Guillemin C, Ergaz Z, Dimov S, Suderman M, et al. (2015) Gestational diabetes alters offspring DNA methylation profiles in human and rat: Identification of key pathways involved in endocrine system disorders, insulin signaling, diabetes signaling, and ILK signaling. Endocrinology 156: 2222-2238.
- Olsen AS, Sarras MP Jr, Intine RV (2010) Limb regeneration is impaired in an adult zebrafish model of diabetes mellitus. Wound Repair Regen 18: 532-542.
- Sarras MP Jr, Leontovich AA, Intine RV (2015) Use of zebrafish as a model to investigate the role of epigenetics in propagating the secondary complications observed in diabetes mellitus. Comp Biochem Physiol C Toxicol Pharmacol 178: 3-7.
- Leontovich J, Sarras MP (2019) Distribution of methylated regions within GDMNA in acute and chronic phases of diabetes mellitus. The handbook of nutrition diet and epigenetics (In Press).
- El Hajj N, Pliushch G, Schneider E, Dittrich M, Müller T, et al. (2013) Metabolic programming of MEST DNA methylation by intrauterine exposure to gestational diabetes mellitus. Diabetes 62: 1320-1328.
- West NA, Kechris K, Dabelea D (2013) Exposure to maternal diabetes in utero and DNA methylation patterns in the offspring. Immunometabolism 1: 1-9.
- Houde AA, St-Pierre J, Hivert MF, Baillargeon JP, Perron P, et al. (2014) Placental lipoprotein lipase DNA methylation levels are associated with gestational diabetes mellitus and maternal and cord blood lipid profiles. J Dev Orig Health Dis 5: 132-141.
- Chen D, Zhang A, Fang M, Fang R, Ge J, et al. (2014) Increased methylation at differentially methylated region of GNAS in infants born to gestational diabetes. BMC Med Genet 15: 108.
- Chen P, Piaggi P, Traurig M, Bogardus C, Knowler WC, et al. (2017) Differential methylation of genes in individuals exposed to maternal diabetes in utero. Diabetologia 60: 645-655.
- Wang L, Fan H, Zhou L, Wu Y, Lu H, et al. (2018) Altered expression of PGC-1α and PDX1 and their methylation status are associated with fetal glucose metabolism in gestational diabetes mellitus. Biochem Biophys Res Commun 501: 300-306.
- Ambra R, Manca S, Palumbo MC, Leoni G, Natarelli L, et al. (2014) Transcriptome analysis of Human Primary Endothelial Cells (HUVEC) from umbilical cords of gestational diabetic mothers reveals candidate sites for an epigenetic modulation of specific gene expression. Genomics 103: 337-348.
- Blue EK, Sheehan BM, Nuss ZV, Boyle FA, Hocutt CM, et al. (2015) Epigenetic regulation of placenta-specific 8 contributes to altered function of endothelial colony-forming cells exposed to intrauterine gestational diabetes mellitus. Diabetes 64: 2664-2675.
- Rogulski K, Li Y, Rothermund K, Pu L, Watkins S, et al. (2005) Onzin, a c-Myc-repressed target, promotes survival and transformation by modulating the Akt-Mdm2-p53 pathway. Oncogene 24: 7524-7541.
- Kinsey C, Balakrishnan V, O’Dell MR, Huang JL, Newman L, et al. (2014) Plac8 links oncogenic mutations to regulation of autophagy and is critical to pancreatic cancer progression. Cell Rep 7: 1143-1155.
- Li C, Ma H, Wang Y, Cao Z, Graves-Deal R, et al. (2014) Excess PLAC8 promotes an unconventional ERK2-dependent EMT in colon cancer. J Clin Invest 124: 2172-2187.
- Jimenez-Preitner M, Berney X, Uldry M, Vitali A, Cinti S, et al. (2011) Plac8 is an inducer of C/EBPβ required for brown fat differentiation, thermoregulation, and control of body weight. Cell Metab 14: 658-670.
- Ledford JG, Kovarova M, Koller BH (2007) Impaired host defense in mice lacking ONZIN. J Immunol 178: 5132-5143.
- McMurray HR, Sampson ER, Compitello G, Kinsey C, Newman L, et al. (2008) Synergistic response to oncogenic mutations defines gene class critical to cancer phenotype. Nature 453: 1112-1116.
- Yang IV, Zhang W, Davidson EJ, Fingerlin TE, Kechris K, et al. (2018) Epigenetic marks of in uteroexposure to gestational diabetes and childhood adiposity outcomes: The EPOCH study. Diabet Med 35: 612-620.
- Agha G, Hajj H, Rifas-Shiman SL, Just AC, Hivert MF, et al. (2016) Birth weight-for-gestational age is associated with DNA methylation at birth and in childhood. Clin Epigenetics 8: 118.
- Hillman SL, Finer S, Smart MC, Mathews C, Lowe R, et al. (2015) Novel DNA methylation profiles associated with key gene regulation and transcription pathways in blood and placenta of growth-restricted neonates. Epigenetics 10: 50-61.
- Lesseur C, Armstrong DA, Paquette AG, Li Z, Padbury JF, et al. (2014) Maternal obesity and gestational diabetes are associated with placental leptin DNA methylation. Am J Obstet Gynecol 211: 654.
- Díaz M, García C, Sebastiani G, de Zegher F, López-Bermejo A, et al. (2017) Placental and cord blood methylation of genes involved in energy homeostasis: Association with fetal growth and neonatal body composition. Diabetes 66: 779-784.
- Deng X, Yang Y, Sun H, Qi W, Duan Y, et al. (2018) Analysis of whole genome-wide methylation and gene expression profiles in visceral omental adipose tissue of pregnancies with gestational diabetes mellitus. J Chin Med Assoc 81: 623-630.
- Tessier DR, Ferraro ZM, Gruslin A (2013) Role of leptin in pregnancy: Consequences of maternal obesity. Placenta 34: 205-211.
- Wu P, Farrell WE, Haworth KE, Emes RD, Kitchen MO, et al. (2018) Maternal genome-wide DNA methylation profiling in gestational diabetes shows distinctive disease-associated changes relative to matched healthy pregnancies. Epigenetics 13: 122-128.
- Michalczyk AA, Dunbar JA, Janus ED, Best JD, Ebeling PR, et al. (2016) Epigenetic markers to predict conversion from gestational diabetes to type 2 diabetes. J Clin Endocrinol Metab 101: 2396-2404.
- Kim E, Kwak SH, Chung HR, Ohn JH, Bae JH, et al. (2017) DNA methylation profiles in sibling pairs discordant for intrauterine exposure to maternal gestational diabetes. Epigenetics 12: 825-832.
Citation: Sarras MP Jr, Leontovich A (2018) The Effect Gestational Diabetes has on DNA Methylation as Related to Fetal Development and the Outcomes of the Mother and Offspring. J Diabetes Metab Disord 5: 025.
Copyright: © 2018 Michael P Sarras Jr, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

