
Sepsis in Children
*Corresponding Author(s):
Donna MendezDepartment Of Pediatric Emergency Medicine, The University Of Texas-Health Science Center At Houston Medical School, Houston, Texas, United States
Tel:+44 7137044060,
Email:Donna.Mendez@uth.tmc.edu
Abstract
Background: Sepsis in children still accounts for a substantial morbidity and mortality despite the advances in treatment. Rapidly recognizing a child with SIRS or sepsis is key to having favorable outcomes. The purpose of this review is to provide the most current recommendations for evaluation and treatment including differences in the American College of Critical Care Medicine (ACCM) and Pediatric Advanced Life Support (PALS) sepsis guidelines.
Methods: The literature is from a range of sources that was reviewed and synthesized to provide an overview of the most current methods for evaluation and treatment of sepsis in children.
Results: This review summarizes the definitions, clinical manifestations, evaluation and treatment of sepsis in children. Inclusion of the most recent biomarkers of sepsis are included and reviewed.
INTRODUCTION
Systemic Inflammatory Response (SIRS) and Sepsis are important to recognize early and treat aggressively since without treatment it can lead to increased morbidity and mortality. Even though pediatric mortality has decreased with the advent of antibiotics and better management, the mortality is still 4-10 percent in pediatric patients with severe sepsis [1-3] and 13 to 34 percent in pediatric patients with septic shock [1-8].
It is important to rapidly recognize SIRS and sepsis early to have favorable outcomes in children. Early recognition and treatment with the American Critical Care Medicine (ACCM)-PALS guidelines has improved outcomes in children. In a study by Han and colleague, they showed a 92% survival versus 62% survival among patients who did not receive ACCM-PALS guidelines [8]. In a prospective study of 91 infants and children presenting to community hospitals with septic shock, each hour delay in initiation of appropriate resuscitation or persistence of hemodynamic abnormalities was associated with a clinically significant increased risk of death [8]. In a trial of goal-directed therapy in 102 children with severe sepsis or fluid-refractory septic shock treated in two pediatric intensive care units, 28-day mortality was lower in patients who received goal-directed therapy versus therapy guided by blood pressure (12 versus 39 percent) [9]. In another study, the implementation of timely goal directed interventions by a mobile intensive care team compatible with the ACCM 2002 guidelines for 331 children with meningococcemia in the United Kingdom was associated with a decrease in the case fatality rate from 23 to 2 percent over five years (annual reduction in the odds of death 0.41, 95% CI: 0.27-0.62) [10].
DATE COLLECTION
We collected all the articles that were published from January 1997 to January 2015, which described children with SIRS or sepsis. These articles were obtained by searching PUBMED using key words “sepsis”, “pediatric sepsis”, “SIRS”, “pediatric SIRS” “CRP and sepsis”, “Procalcitonin and sepsis”, “CRP and sepsis”, and “Lactate and sepsis.” Retrospective and prospective studies were included. We excluded studies which did not include children. Opinion articles were also excluded from this review. After selecting the articles, the relevant information was extracted and classified according to evaluation and treatment. After screening the articles, a total of 60 articles were considered to be relevant.
DEFINITIONS
- Core temp of >38.5?C (100.4?F) or <36?C (96.8?F)
- Tachycardia (mean HR more than 2 SD above normal for age), or for children younger than 1 year, bradycardia (mean HR <10th percentile for age).
- Mean RR more than 2 SD above normal for a age or mechanical ventilation for acute pulmonary process.
- Leukocyte count elevated or depressed for age or >10 % immature neutrophils.
| Pediatric Systemic Inflammatory Response Syndrome Criteria | |||||
| Age Group |
Heart Rate (beats/min) |
Respiratory Rate (beats/min) |
Leukocyte Count (Leukocytes X 103mm3) |
Systollic Blood Pressure (mmHg) | |
| Tachycardia | Bradycardia | ||||
| Newborn (0 days to 1 week) | >180 | <100 | >50 | >34 | <59 |
| Neonate (1 week to 1 month) | >180 | <100 | >40 | >19.5 or <5 | <79 |
| Infant (1 month to 1 year) | >180 | <90 | >34 | >17.5 or <5 | <75 |
| Toddler and preschool (>1 to 5 years) | >140 | NA | >22 | >15.5 or <6 | <74 |
| School age (>130NA>18>13.5 or <4.5<83 | |||||
| Adolescent (>12 to>110NA>14>11 or <4.5<90 | |||||
Cardiovascular
Respiratory
Neurologic
Hematologic
Renal
Hepatic
CLINICAL DIAGNOSTICS OF SEPTIC SHOCK
Vital signs
| Temperature (F) | 0-1 year old | 2-5 year old | 6-12 year old | >12 year old |
| <100 | 180 | 140 | 130 | 110 |
| 101 | 185 | 145 | 135 | 115 |
| 102 | 190 | 159 | 140 | 120 |
| 103 | 195 | 155 | 145 | 125 |
| 104 | 200 | 160 | 150 | 130 |
| 105 | 205 | 165 | 155 | 135 |
| 106 | 210 | 165 | 155 | 140 |
Table 2: Heart rate corrected for temperature [12].
Other physical findings
Presentation of shock in children versus adults
Types of shock and clinical findings
Cold shock is characterized by an increased systemic vascular resistance and decreased cardiac output. The physical findings are: delayed capillary refill (>2 seconds), diminished pulses and mottled or cool extremities.
ETIOLOGY
Bacteria
| Age | Pathogen | Antibiotic Coverage | Adminster |
| <28 days |
Staphylococcus aureus Listeria Gram negative (especially E Coli) |
Vancomycin Ampicillin & Gentamicin |
Ampicillin & Gentamicin |
| 29 days- 3 mos |
Staphylococcus aureus |
Vancomycin |
Vancomycin & Ceftriaxone or Cefepime |
| >3 mos |
Streptococcus pneumonia Neisseria meningitidis |
Cefotaxime/Ceftriaxone & Vanc (meningitis) Cefotaxime/Ceftriaxone |
Vancomycin Ceftriaxone or Cefepime |
| Febrile neutropenia |
Gram positive (Coagulase-negative staphylococci, Staphylococcus aureus, Streptococcus pneumonia, viridans streptococci) Gram negative (Pseudomonas aureus, E coli, Klebsiella) |
Vancomycin Cefepime or Ceftazidime |
Vancomycin Cefepime or Ceftazidime |
| In hospital-acquired | Coagulase-negative staphylococci Gram negatives |
Vancomycin Cefepime or Ceftazidime |
Vancomycin Cefepime or Ceftazidime |
Viruses
- Respiratory (RSV, influenza, parainfluenza, adenovirus, metapneumovirus)
- H1N1
- EBV, CMV
- Herpes simplex, enterovirus.
Fungi
- Malignancy
- Indwelling vascular catheters
- Prolonged neutropenia (>4 days) [14]
- Other - Parasitic or Rickettsial
Culture negative sepsis
EVALUATION
Clinical evaluation
LABORATORY STUDIES
- Rapid glucose-Hypoglycemia is associated with sepsis in children. Children have less glycogen stores than adults so they become hypoglycemia more frequently those adults with sepsis. Hypoglycemia is defined as < 60 mg/dl in children, < 45 mg/dl in neonates. Treat hypoglycemia with 2.5-5mL/kg of 10% Dextrose solution (D10W) for intents to children up to 12 years, and 1-2mL/kg of 25 percent Dextrose (D25W) in adolescents.
- Arterial Blood Gas (ABG) or Venous Blood Gas (VBG)-Children with sepsis frequently have acidosis from poor tissue perfusion.
- Complete Blood Count (CBC)-Sepsis in children frequently is associated with leukocytosis or leukopenia.
- Blood lactate -Elevation of blood lactate (arterial >3.5mmol/L or venous >4.0mmol/L) is associated with sepsis.
- Serum Electrolytes-Electrolyte abnormalities (eg: Hyponatremia, hyperkalemia, hypokalemia, and hypophosphatemia) may be associated with sepsis through syndrome of inappropriate secretion of antidiuretic hormone and capillary leak.
- Serum calcium-Ionized calcium <1.1mml/L, or nonionized (4.8mg/dL) may affect myocardial function & vascular tone. Treat with calcium gluconate 10% at dose of 50-100mg/kg up to 2g slowly by IV or IO (don’t give with bicarbonate). Treat hypocalcemia with calcium chloride 10% in dose of 10-20mg/kg up to 1g in central line or IO. Infants under 12 months may rely on extracellular calcium to maintain cardiac contractility. Animal models suggest improvement, but no human studies do, so it is recommended only to treat if hypocalcemia.
- Serum creatinine-Renal insufficiency suggested by a serum creatinine >2 times upper limit of normal for age or twofold increase in baseline creatinine is support the diagnosis of septic shock.
- Serum total bilirubin and Alanine aminotransferase- A total bilirubin >4mg/dL, (not in newborn) or Alanine aminotransferase (ALT) >2 time upper limit of normal for age indicates liver dysfunction in sepsis.
- Prothrombin Time (PT) Partial Thromboplastin Time (aPTT), and International Normalized Ratio (INR)-An elevation of PT and aPTT or INR suggests Disseminated Intravascular Coagulopathy (DIC).
- Fibrinogen & D-Dimer-Decreased fibrinogen and increased D-Dimer support DIC
- Cultures- blood, urine, CSF, wound
BIOMARKERS
C-reactive protein CRP
CRP and Procalcitonin (PCT)
Procalcitonin (PCT)
Soluble adhesion molecules
Interleukin 6 (IL-6)
IL-6 has been shown to be a sensitive biomarker for predicting bacteremia/clinical sepsis in children with febrile neutropenia [45,46]. In severe sepsis, the best diagnostic accuracies were found for I-6 and procalcitonin and these were significantly higher than those for Lipopolysaccharide-Binding Protein (LBP) on admission [46].
Lactate
In another study by Duke and colleagues of children with sepsis syndrome or septic shock, blood lactate level was the earliest predictor of outcome in children with sepsis. The following data were recorded at admission, 12, 24 and 48 h: heart rate, mean arterial pressure, arterial pH, base deficit, arterial lactate, gastric intramucosal pH (pHi) and DCO (intramucosal carbon dioxide tension minus arterial partial pressure of carbon dioxide). The principal outcome measure was survival. The secondary outcome measure was the number of organ systems failing at 48 h after admission. There were 10 deaths and 21 survivors. No variable discriminated survival from death at presentation. Blood lactate level was the earliest discriminator of survival. Using univariate logistic regression, lactate discriminated survivors from those who died at 12 and 24 h after admission, but not at 48 h (p = 0.049, 0.044 and 0.062, respectively [48,49].
MANAGEMENT
Clinical management
Endotracheal intubation with Rapid Sequence Intubation (RSI) is often necessary in children with septic shock to protect the airway, assist with ventilation and/or promote oxygenation. Endotracheal intubation and sedation reduces the work of breathing which will retain cardiac output to other organs than the muscles of respiration. As per the updated CCM guidelines in 2007, high-flow heated and humidified oxygen by nasal cannula until intubation is done is recommended [18].
The decision to intubate and ventilate should be based on clinical assessment of increased work of breathing, hypoventilation or impaired mental status. Waiting for confirmatory laboratory tests is discouraged since 40% of cardiac output is used for work of breathing and delay of intubation could be deleterious [18].
Overview of RSI:
- Preoxygenate: Preoxygenate: Preoxygenate with 100% for 2-5 minutes. Also preoxygenate with high flow oxygen 15L/min with Nasal Cannula (NC). Preoxygenation is essential due to a child's intolerance for apnea. A high-flow NC (15L in adolescents, up to 8L in toddlers) can be used for preoxygenation in addition to the face mask and while intubating. It has been shown to decrease apnea and desaturations during RSI [50,51].
- Preparation of equipment and medications
- Pretreatment: Atropine- All children
- Sedation
- Etomidate- Not recommended per the 2007 Clinical Guidelines for Hemodynamic Support of Neonates and Children with Septic Shock from American College of Critical Care Medicine.
- Ketamine: Safe with hemodynamic instability if patient is not catecholamine depleted. Dose 1-2mg/kg IV (If no IV access, can be given IM at dose of 3-7mg/kg)
- Midazolam- May cause hemodynamic instability at doses required for sedation. Dose: 0.2-0.3mg/kg IV (maximum dose 2mg, onset of effect requires 2-3 minutes).
- Thioopental- Not recommended with hemodynamic instability. Dose: 3-5mg/kg iV.
- Paralytic
- Succinycholine- Not recommended with chronic myopathy or denervating neuromuscular disease: 48-72 hours after burn, crush or denervating injury: malignant hyperthermia; or pre-existing hyperkalemia. Dose: infants and young children: 2mg/kg IV, older children: 1-1.5mg/kg IV. (If IV access unobtainable, can be given IM at dose of 3-5mg/kg)
- Rocuronium- Has none of the adverse effects of succinylcholine, making it an ideal alternative. Dose: 1mg/kg.
The first hour goals include restoring and maintaining normal heart rate, capillary refill
The following therapeutic endpoints should be targeted goals:
- Strong, distal pulses equal to central pulses
- Capillary refill
- Normal mental status
- Urine output (>1mL/kg/hour, up to 40mL per hour
- Blood pressure in children 1 month to 10 years (systolic pressure at least fifth percentile for age: 60 mmHg
- Lactate (<4mmol/L or >10 percent decrease per hour until normal)
- Central venous oxygen saturation (ScvO2), (>70 percent)
Treatment Guidelines
The 2007 American College of Critical Care Medicine (ACCM) guidelines
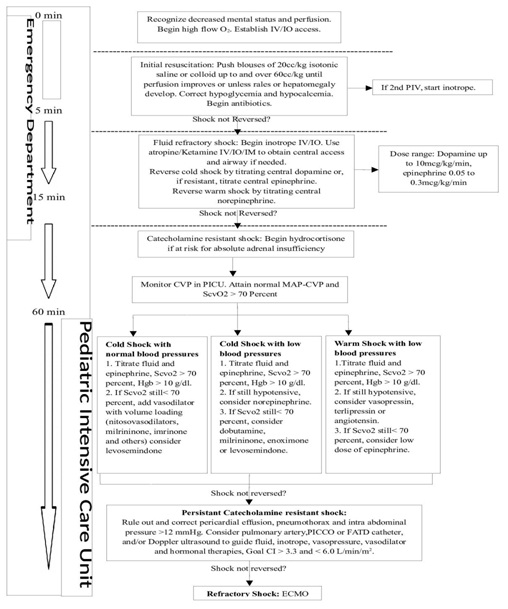 Figure 1: ACCM Guidelines.
Figure 1: ACCM Guidelines.Using these ACCM guidelines has shown a reduction in mortality of severe sepsis in children [9,52,53]. In a trial of goal-directed therapy in 102 children with severe sepsis or fluid-refractory septic shock treated in two pediatric intensive care units, 28-day mortality was lower in patients who were treated by the ACCM guideline versus therapy guided by blood pressure (12 versus 39 percent, respectively) [9]. In a single centered center in Pittsburgh, patients treated according to ACCM guidelines found that every hour's delay in resuscitation in the emergency room was associated with a 40% increase in mortality in children with septic shock [8]. In another study, institution of the ACCM guidelines by a mobile intensive care team, in the care for 331 children with meningococcemia in the United Kingdom was associated with a decrease in the case fatality rate from 23 to 2 percent over five years (annual reduction in the odds of death 0.41, 95% CI: 0.27-0.62) [10].
Pediatric Advanced Life Support (PALS) guidelines for septic shock
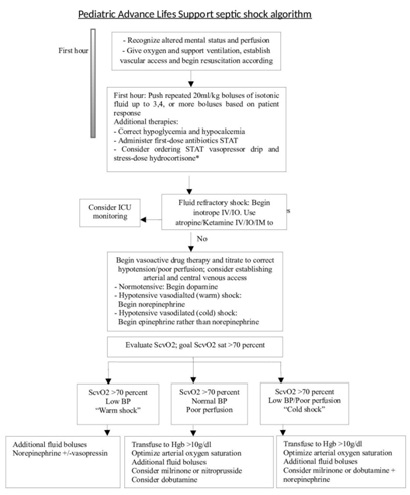 Figure 2: PALS Guidelines.
Figure 2: PALS Guidelines.Implementation of the PALS septic shock guidelines has also shown a reduction in mortality and morbidity. In a retrospective study over a 4-year period from 5 regional pediatric centers’ specialty care transport teams showed a significant decrease in morbidity and mortality. Four thousand eight hundred fifty-six patients ranging in age from newborn to 18 years of age were included. Use of the PALS guidelines resulted in a 2 fold reduction in mortality (5.06% vs 16.37%) and functional morbidity (1.56% vs 4.11%) [54].
COMPARING THE ACCM AND PALS GUIDELINES
The 2007 American College of Critical Care Medicine (ACCM) guidelines and Pediatric Advanced Life Support are similar but the ACCM guidelines provide a tighter time frame of delivery of initial intravenous fluids boluses. The ACCM guidelines recommend fluid boluses within the first 15 minutes and PALS recommend fluid boluses within first 60 minutes. Literature has shown the quicker shock treatment is administered the better the morbidity and mortality in children with septic shock. Acting quickly and it’s relation to morbidity was shown in a study by Han. Every hour’s delay in resuscitation was associated with a 40% increase in mortality [8]. Other observational evidence from pediatric studies suggest that vigorous fluid resuscitation may play a major role in preventing end-organ damage and improve survival in sepsis [9,10,37]. In a recent quality improvement study, a reduction in severe sepsis mortality (4.0-2.4%) was shown with the delivery of fluid boluses and antibiotics in the first hour in a pediatric emergency department [55]. The improved morbidity and mortality associated with fluid boluses is due to it disrupting the normal progression of sepsis. The normal progression of patients in shock is for them to be initially tachycardic then as shock progresses; they become vasoconstricted (prolonged capillary refill of more than 3 seconds) and then eventually hypotensive. Mortality risks increase as the patient progresses through shock. Carcillo and colleagues examined over 5,000 children and found the mortality with tachycardia alone was 4.5% and increased to 33.7% when tachycardia was associated with hypotension and prolonged capillary refill. If shock treatment guidelines are initiated rapidly one can reverse this mortality. By treating with normal saline boluses early when the patient is tachycardic prevents their progression to tachycardia associated with hypotension and prolonged capillary refill. It makes sense that the ACCM guidelines would prove to be superior to the PALS guidelines since the ACCM guidelines recommend fluid boluses be administered in the first 15 minutes and the PALS guidelines recommend this within the first 60 minutes.
PHARMACOLOGICAL TREATMENT
Antibiotics
Corticosteroids
REFERENCES
- Hartman ME, Linde-Zwirble WT, Angus DC, Watson RS (2013) Trends in the epidemiology of pediatric severe sepsis*. Pediatr Crit Care Med 14: 686-693.
- Ruth A, McCracken CE, Fortenberry JD, Hall M, Simon HK, et al. (2014) Pediatric severe sepsis: current trends and outcomes from the Pediatric Health Information Systems database. Pediatr Crit Care Med 15: 828-838.
- Balamuth F, Weiss SL, Neuman MI, Scott H, Brady PW, et al. (2014) Pediatric severe sepsis in US children’s hospitals. Pediatr Crit Care Med 15: 798-805.
- Watson RS, Carcillo JA, Linde-Zwirble WT, Clermont G, Lidicker J, et al. (2003) The epidemiology of severe sepsis in children in the United States. Am J Respir Crit Care Med 167: 695-701.
- Weiss SL, Parker B, Bullock ME, Swartz S, Price C, et al. (2012) Defining pediatric sepsis by different criteria: discrepancies in populations and implications for clinical practice. Pediatr Crit Care Med 13: 219-226.
- Kutko MC, Calarco MP, Flaherty MB, Helmrich RF, Ushay HM, et al. (2003) Mortality rates in pediatric septic shock with and without multiple organ system failure. Pediatr Crit Care Med 4: 333-337.
- Jaramillo-Bustamante JC, Marín-Agudelo A, Fernández-Laverde M, Bareño-Silva J (2012) Epidemiology of sepsis in pediatric intensive care units: first Colombian multicenter study. Pediatr Crit Care Med 13: 501-508.
- Han YY, Carcillo JA, Dragotta MA, Bills DM, Watson RS, et al. (2003) Early reversal of pediatric-neonatal septic shock by community physicians is associated with improved outcome. Pediatrics 112: 793-799.
- de Oliveira CF, de Oliveira DS, Gottschald AF, Moura JD, Costa GA, et al. (2008) ACCM/PALS haemodynamic support guidelines for paediatric septic shock: an outcomes comparison with and without monitoring central venous oxygen saturation. Intensive Care Med 34: 1065-1075.
- Booy R, Habibi P, Nadel S, de Munter C, Britto J, et al. (2001) Reduction in case fatality rate from meningococcal disease associated with improved healthcare delivery. Arch Dis Child 85: 386-390.
- Goldstein B, Giroir B, Randolph A, International Consensus Conference on Pediatric Sepsis (2005) International Consensus Conference on Pediatric Sepsis. International pediatric sepsis consensus conference: definitions for sepsis and organ dysfunction in pediatrics. Pediatr Crit Care Med 6: 2-8.
- Hanna CM, Greenes DS (2004) How much tachycardia in infants can be attributed to fever? Ann Emerg Med 43: 699-705.
- Davies P, Maconochie I (2009) The relationship between body temperature, heart rate and respiratory rate in children. Emerg Med J 26: 641-643.
- Zaoutis TE, Prasad PA, Localio AR, Coffin SE, Bell LM, et al. (2010) Risk factors and predictors for candidemia in pediatric intensive care unit patients: implications for prevention. Clin Infect Dis 51: 38-45.
- Gaines NN, Patel B, Williams EA, Cruz AT (2012) Etiologies of septic shock in a pediatric emergency department population. Pediatr Infect Dis J 31: 1203-1205.
- Nadel S, Goldstein B, Williams MD, Dalton H, Peters M, et al. (2007) Drotrecogin alfa (activated) in children with severe sepsis: a multicentre phase III randomised controlled trial. Lancet 369: 836-843.
- Lucignano B, Ranno S, Liesenfeld O, Pizzorno B, Putignani L, et al. (2011) Multiplex PCR allows rapid and accurate diagnosis of bloodstream infections in newborns and children with suspected sepsis. J Clin Microbiol 49: 2252-2258.
- Brierley J, Carcillo JA, Choong K, Cornell T, Decaen A, et al. (2009) Clinical practice parameters for hemodynamic support of pediatric and neonatal septic shock: 2007 update from the American College of Critical Care Medicine. Crit Care Med 37: 666-688.
- Standage SW, Wong HR (2011) Biomarkers for pediatric sepsis and septic shock. Expert Rev Anti Infect Ther 9: 71-79.
- McCabe RE, Remington JS (1984) C-reactive protein in patients with bacteremia. J Clin Microbiol 20: 317-319.
- Jaye DL, Waites KB (1997) Clinical applications of C-reactive protein in pediatrics. Pediatr Infect Dis J 16: 735-746.
- Hofer N, Zacharias E, Müller W, Resch B (2012) An update on the use of C-reactive protein in early-onset neonatal sepsis: current insights and new tasks. Neonatology 102: 25-36.
- Marshall JC, Reinhart K, International Sepsis Forum (2009) Biomarkers of sepsis. Crit Care Med 37: 2290-2298.
- Kaplan JM, Wong HR (2011) Biomarker discovery and development in pediatric critical care medicine. Pediatr Crit Care Med 12: 165-173.
- Luaces-Cubells C, Mintegi S, García-García JJ, Astobiza E, Garrido-Romero R, et al. (2012) Procalcitonin to detect invasive bacterial infection in non-toxic-appearing infants with fever without apparent source in the emergency department. Pediatr Infect Dis J 31: 645-647.
- Maniaci V, Dauber A, Weiss S, Nylen E, Becker KL, et al. (2008) Procalcitonin in young febrile infants for the detection of serious bacterial infections. Pediatrics 122: 701-710.
- Hatzistilianou M, Rekliti A, Athanassiadou F, Catriu D (2010) Procalcitonin as an early marker of bacterial infection in neutropenic febrile children with acute lymphoblastic leukemia. Inflamm Res 59: 339-347.
- Juutilainen A, Hämäläinen S, Pulkki K, Kuittinen T, Nousiainen T, et al. (2011) Biomarkers for bacteremia and severe sepsis in hematological patients with neutropenic fever: multivariate logistic regression analysis and factor analysis. Leuk Lymphoma 52: 2349-2355.
- Samransamruajkit R, Uppala R, Pongsanon K, Deelodejanawong J, Sritippayawan S, et al. (2014) Clinical outcomes after utilizing surviving sepsis campaign in children with septic shock and prognostic value of initial plasma NT-proBNP. Indian J Crit Care Med 18: 70-76.
- Aird WC (2003) The role of the endothelium in severe sepsis and multiple organ dysfunction syndrome. Blood 101: 3765-3777.
- Carman CV, Springer TA (2008) Trans-cellular migration: cell-cell contacts get intimate. Curr Opin Cell Biol 20: 533-540.
- Springer TA (1990) Adhesion receptors of the immune system. Nature 346: 425-434.
- Springer TA (1990) Traffic signals for lymphocyte recirculation and leukocyte emigration: the multi-step paradigm. Nature 346:425-433.
- Garton KJ, Gough PJ, Raines EW (2006) Emerging roles for ectodomain shedding in the regulation of inflammatory responses. J Leukoc Biol 79: 1105-1116.
- Briassoulis G, Papassotiriou I, Mavrikiou M, Lazaropoulou C, Margeli A (2007) Longitudinal course and clinical significance of TGF-beta1, sL- and sE-Selectins and sICAM-1 levels during severe acute stress in children. Clin Biochem 40: 299-304.
- Paize F, Sarginson R, Makwana N, Baines PB, Thomson AP, et al. (2012) Changes in the sublingual microcirculation and endothelial adhesion molecules during the course of severe meningococcal disease treated in the paediatric intensive care unit. Intensive Care Med 38: 863-871.
- Ng PC, Cheng SH, Chui KM, Fok TF, Wong MY, et al. (1997) Diagnosis of late onset neonatal sepsis with cytokines, adhesion molecule, and C-reactive protein in preterm very low birthweight infants. Arch Dis Child Fetal Neonatal Ed 77: 221-227.
- Gonzalez BE, Mercado CK, Johnson L, Brodsky NL, Bhandari V (2003) Early markers of late-onset sepsis in premature neonates: clinical, hematological and cytokine profile. J Perinat Med 31: 60-68.
- Ng PC (2004) Diagnostic markers of infection in neonates. Arch Dis Child Fetal Neonatal Ed 89: 229-235.
- Bhartiya D, Kapadia C, Sanghvi K, Singh H, Kelkar R, et al. (2000) Preliminary studies on IL-6 levels in healthy and septic Indian neonates. Indian Pediatr 37: 1361-1367.
- Buck C, Bundschu J, Gallati H, Bartmann P, Pohlandt F (1994) Interleukin-6: a sensitive parameter for the early diagnosis of neonatal bacterial infection. Pediatrics 93: 54-58.
- Maamouri G, Boskabadi H, Tavakkolafshari J, Shakeri M (2006) Evaluation Quantities Interleukin 6 in diagnosis of Neonatal sepsis. Med J Mashad Univ Med Sci 93: 253-260.
- Krueger M, Nauck MS, Sang S, Hentschel R, Wieland H, et al. (2001) Cord blood levels of interleukin-6 and interleukin-8 for the immediate diagnosis of early-onset infection in premature infants. Biol Neonate 80: 118-123.
- Romagnoli C, Frezza S, Cingolani A, De Luca A, Puopolo M, et al. (2001) Plasma levels of interleukin-6 and interleukin-10 in preterm neonates evaluated for sepsis. Eur J Pediatr 160: 345-350.
- Kitanovaski L, Jazbec J, Hojker S, Dergane M (2014) Diagnostic accuracy of lipopolysaccharide-binding protein for predicting bacteremia/clinical sepsis in children with febrile neutropenia: comparison with interleukin-6, procalcitonin, and C-reactive protein. Support Care Cancer 22:269-277.
- Kitanovski L, Jazbec J, Hojker S, Gubina M, Derganc M (2006) Diagnostic accuracy of procalcitonin and interleukin-6 values for predicting bacteremia and clinical sepsis in febrile neutropenic children with cancer. Eur J Clin Microbiol Infect Dis 25: 413-415.
- Scott HF, Donoghue AJ, Gaieski DF, Marchese RF, Mistry RD (2012) The utility of early lactate testing in undifferentiated pediatric systemic inflammatory response syndrome. Acad Emerg Med 19: 1276-1280.
- Duke TD, Butt W, South M (1997) Predictors of mortality and multiple organ failure in children with sepsis. Intensive Care Med 23: 684-692.
- Asfar P, Calzia E, Huber-Lang M, Ignatius A, Radermacher P (2012) Hyperoxia during septic shock--Dr. Jekyll or Mr. Hyde? Shock 37: 122-123.
- Lee JH, Rehder KJ, Williford L, Cheifetz IM, Turner DA (2013) Use of high flow nasal cannula in critically ill infants, children, and adults: a critical review of the literature. Intensive Care Med 39: 247-257.
- Weingart SD, Levitan RM (2012) Preoxygenation and prevention of desaturation during emergency airway management. Ann Emerg Med 59: 165-175.
- Booy R, Habibi P, Nadel S, de Munter C, Britto J, et al. (2001) Reduction in case fatality rate from meningococcal disease associated with improved healthcare delivery. Arch Dis Child 85: 386-390.
- Maat M, Buysse CM, Emonts M, Spanjaard L, Joosten KF, et al. (2007) Improved survival of children with sepsis and purpura: effects of age, gender, and era. Crit Care 11: 112.
- Carcillo JA, Kuch BA, Han YY, Day S, Greenwald BM, et al. (2009) Mortality and functional morbidity after use of PALS/APLS by community physicians. Pediatrics 124: 500-508.
- Cruz AT, Perry AM, Williams EA, Graf JM, Wuestner ER, et al. (2011) Implementation of goal-directed therapy for children with suspected sepsis in the emergency department. Pediatrics 127: 758-766.
- Dellinger RP, Levy MM, Rhodes A, Annane D, Gerlach H, et al. (2013) Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock 2012. Crit Care Med 41: 580- 637.
- Deminger RF, Levy MM, Rhodes A, Annane D, Gerlach H, et al. (2013) Surviving sepsis campaign international guidelines for management of severe sepsis and septic shock 2012. Intensive Care Med 3: 165-228.
- Russell JA (2006) Management of sepsis. N Engl J Med 355: 1699-1713.
- Brierley J, Carcillo JA, Choong K, Cornell T, Decaen A, et al. (2009) Clinical practice parameters for hemodynamic support of pediatric and neonatal septic shock: 2007 update from the American College of Critical Care Medicine. Crit Care Med 37: 666-688.
Citation: Mendez D, Lapus R (2015) Sepsis in Children. J Emerg Med Trauma Surg Care 2: 010.
Copyright: © 2015 Donna Mendez, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

