
Updated Diagnosis and Management of Osteoarticular Tuberculosis
*Corresponding Author(s):
Kow-Tong ChenDepartment Of Occupational Medicine, Tainan Municipal Hospital, College Of Medicine, Tainan, Taiwan, Province Of China
Tel:88662609926,
Fax:88662606351
Email:ktchen@mail.ncku.edu.tw
Abstract
Methods
Results
Conclusion
Keywords
INTRODUCTION
Tuberculosis (TB) remains a major health problem worldwide. In 2011, the World Health Organization (WHO) reported an estimated 8.7 million incident cases of TB, 12 million prevalent cases of TB, and 1.4 million deaths due to TB [1]. Most cases occurred in Asia (59%) and Africa (26%) [1,2]. As for active clinical TB, the most common form of tubercular disease is Pulmonary TB (PTB); nevertheless, in a widely variable proportion of cases, TB can also affect extra-pulmonary sites within the body, exclusive of or in combination with PTB [3].
Musculoskeletal tuberculosis is a relatively rare extra-pulmonary complication of Mycobacterium tuberculosis. Specific skeletal involvement is observed in 1% to 3% of patients with TB [4]. Among them, approximately one-half of cases show spinal involvement, and the remaining involve the extraspinal osteoarticular joint [5-7]. TB tenosynovitis and arthritis is usually monoarticular, and the organism can be isolated from the joint [8].
TB tenosynovitis and arthritis occurs primarily through hematogenous spread from a primary focus such as the lung, kidney, or lymph node or, infrequently, through contiguous spread from adjacent tissues by direct inoculation [8-12]. TB of the joint may be due direct invasion of the synovium, e.g., Poncet's arthritis [8,11]. It may also affect nonweight-bearing joints such as the wrist, elbow, and the small joints of the hands. Articular disease often starts as a synovitis progressing to periarticular demineralization, marginal erosions, and lastly, joint destruction [13]. The time period from synovitis to joint destruction can be rapid, particularly in weight-bearing joints. When tubercular tenosynovitis and arthritis are complicated by a secondary infection such as Staphylococcus aureus presents, they result in accelerated joint destruction associated with severe systemic features [14]. Joint tissue necrosis secondary to other diseases, as in the cases of osteonecrosis of the joints due to sickle cell disease and chondrocalcinosis may predispose an individual to tubercular infection [15,16].
Patients generally have mild local and constitutional symptoms, frequently leading to significant delays in diagnosis. The diagnosis of tubercular tenosynovitis and arthritis is also frequently delayed due to its varied clinical presentation and frequent lack of radiographic findings and constitutional features [7,17]. The delay in diagnosis and treatment, may result in additional bone or joint destruction, particularly in patients with TB tenosynovitis and arthritis due to infection caused by mycobacterial species [7,18]. Therefore, early diagnosis and treatment are essential. We therefore performed a review of these topics based on the recent literatures.
DATA COLLECTION
We initially collected all of the articles that were published from January 1990 through March 2014, which described subjects affected by TB tenosynovitis and arthritis. These articles were obtained by searching MEDLINE (National Library of Medicine, Bethesda, Maryland, USA) using the key words "osteoarticular tuberculosis" [OR] "tuberculosis arthritis" [OR] "diagnosis of TB arthritis" [OR] "management of TB arthritis". All of the articles are included. Manuscripts without an abstract (which were assumed to not be original), and opinion articles were excluded from the review. After selecting the articles, the relevant information was extracted and classified according to TB arthritis diagnosis, TB arthritis management, the country of study, and the study design.
Literature searches were performed in July 2014 and August 2014. Using the search terms previously described, a total of 161 documents were retrieved from MEDLINE. After screening the articles, a total of 103 articles were considered to be relevant. The countries were primarily from India, the United States, Turkey, Korea, Taiwan, Germany and other countries. The setting that produced the most original information was India, with 35% of the articles. After analyzing the abstracts, we found that 83% of the studies were case reports, 10% were retrospective, and 7% referenced other designs.
DIAGNOSIS OF TB INFECTION
Tuberculin skin test
| Methods |
| Tuberculin Skin Test (TST) |
| Interferon Gamma Release Assays (IGRAs) |
| Enzyme-Linked Immunosorbent Assays (ELISA) |
| Bacteriology |
| Radiology |
| Computed Tomography (CT) |
| Magnetic Resonance Imaging (MRI) |
| Polymerase Chain Reaction (PCR) |
| Synovial fluid examination |
| Synovial biopsy |
Table 1: Summarizing the methods to diagnose tuberculosis arthritis.
Interferon Gamma Release Assays (IGRAs)
In the general population, IGRAs appear to be more powerful than the TST for diagnosing active TB or LTBI [34]. T-SPOT, QuantiFERON-TB and the TST have good diagnostic value for chronic inflammatory arthritis, but indeterminate results may complicate their use [35].
Enzyme-Linked Immunosorbent Assays (ELISA)
Diagnosis of tuberculosis tenosynovitis and arthritis
The most common symptom is local pain and swelling followed by restriction of movement of affected area. There is wasting of the regional muscle, and deformity may occur. Less commonly, a painless cold abscess has been reported as the only clinical presentation. The involvement of multiple sites is observed in 5-30% cases of tubercular arthritis [41,42]. Reactivation of tubercular arthritis after treatment occurs in 17-34% of individuals and is most commonly observed in the hip joint [8,43].
Joint swelling and evidence of effusion, periarticular abscess and chronic sinus formation occur late. Systemic symptoms of fever, weight loss, and night sweats may or may not be present during active TB tenosynovitis and arthritis. Less than 50% of individuals with tubercular tenosynovitis and arthritis have active pulmonary TB at the time of diagnosis [44]. Patients with TB may have hypersensitivity phenomena such as erythema nodosum, episcleritis, uveitis and Poncet's arthritis.
Clinically, TB tenosynovitis and arthritis has been classified into 5 stages [45-47]. Stage I, or the synovitis stage, presents with soft tissue swelling, no bony lesions, and localized osteoporosis, and the outcome after treatment is excellent. Stage II is early arthritis with marginal erosions (one or more erosions or lytic lesions in the bone; discrete diminution of the joint space), and the outcome is good, with only mild stiffness [6]. Stage III is advanced arthritis with subperichondrial cyst formation and loss of joint space; the outcome is fair, with notable loss of motion [6]. Stage IV is more advanced arthritis with joint destruction and no motion at the joint after treatment. Stage V is ankylosis of the joint.
Bacteriology: A confirmation of Acid Fast Bacillus (AFB) from any body fluid or tissue is the gold standard for the diagnosis of tuberculosis. Several studies have reported bacteriological positivity rates as high as 33% even for primary disease states, such as hilar adenopathy [48]. Therefore, every attempt must be made to bacteriologically prove the diagnosis in every case of suspected tuberculosis. Whatever method a clinician uses, he/she needs to collect at least two, preferably three, samples. A Ziehl-Neelsen (ZN) stain can reveal AFB only if the sample contains greater than 10,000 bacilli per mL. Different culture methods, such as Lowenstein-Jensen medium and radiometric (Bactec 12B liquid medium) and non-radiometric (Bactec MGIT 960 system) culture, can be used to confirm the diagnosis in the paucibacillary state [49]. The newer methods are capable of providing faster results and may be used if available. Mycobacterial culture assumes special significance in cases of suspected drug resistance [49].
There is a push to switch from (ZN to auramine microscopy. Despite World Health Organization guidelines that one staining method is sufficient, in some countries national guidelines prescribe that auramine-positive samples should be confirmed by ZN [50]. A positive auramine result followed by a negative ZN result could not be used to exclude TB or to indicate the presence of NTM species. Confirming auramine-positive samples using ZN provided no clinically informative information and was a waste of resources.
Based on the results of bacteriological methods, Multidrug Resistant (MDR) tuberculosis is defined as tuberculosis resistant to at least both isonizid and rifampicin [51]. Extensively Drug-Resistant (XDR) tuberculosis is defined as tuberculosis resistant to isoniazid and refampicin; and any fluoroquinolone; and at least one of the three second-line injectable drug (capreomycin, kanamycin, and amikacin) [51]. Recently, WHO has revised the case definitions of TB and drug-resistant TB so that also the results of WHO-endorsed new molecular and nonmolecular methods can be used to define a case. Case definitions were further revised to avoid judgmental language, so the terms "defaulter" and "TB suspect" have been replaced by "lost to follow-up" and "presumptive TB", respectively [51].
The main Drug Susceptibility Testing (DST) methods are the absolute-concentration method and the Proportion Method (PM) on Lowenstein-Jenson (L-J) medium, but both methods take some weeks for the results. Automation of culture using BACTEC MGIT 960 (M960) system is being widely implemented [52].
Accurate and rapid detection of Tuberculosis (TB) and drug resistance are critical for improving patient care and decreasing the spread of TB. The Xpert MTB/RIF assay, which enables simultaneous detection of Mycobacterium Tuberculosis (MTB) and Rifampicin (RIF) resistance, was endorsed by WHO in 2010 [53]. Xpert may also be valuable as an add-on test following microscopy for patients who have previously been found to be smear-negative. An Xpert result that is positive for rifampicin resistance should be carefully interpreted and take into consideration the risk of MDR-TB in a given patient and the expected prevalence of MDR-TB in a given setting.
Radiology: Radiographic features are usually noted 2 to 5 months after disease onset [7]. The classical triad of radiologic characteristics of TB tenosynovitis and arthritis is juxta-articular osteoporosis, peripheral osseous erosion and gradual narrowing of the intra-articular space (Figure 1) [6,54,55]. In contrast to rheumatoid arthritis, the joint space is relatively preserved in early TB arthritis. In children, an enlargement of the epiphysis may be observed. A bone scan shows increased uptake, but bone scan findings are non-pathognomonic.
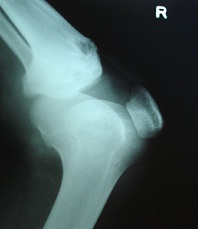 Figure 1: Radiograph of tuberculosis tenosynovitis and arthritis at tibial tuberosity.
Figure 1: Radiograph of tuberculosis tenosynovitis and arthritis at tibial tuberosity.
Imaging: Computed Tomography (CT) and Magnetic Resonance Imaging (MRI) are helpful in further defining the disease (Figure 2) [6,56]. MRI defines soft tissues better, while CT is good for bony lesions. The MRI features of tubercular tenosynovitis and arthritis include synovitis, effusion, central and peripheral erosions, active and chronic pannus, abscess, bone chips, and hypo-intense synovium. MRI is the investigation of choice to reveal both the extent and severity of damage [16]. MRI is also nonspecific but better depicts the extent of the lesion when compared with X-rays. These imaging features in an appropriate clinical setting may aid the diagnosis of tubercular tenosynovitis and arthritis [55]. Positron Emission Tomography (PET) using 2-[18F]-Fluoro-2-Deoxy-D-Glucose (FDG) has been used to diagnose in breast, colorectal, head and neck, lung, lymphoma, melanoma, oesophageal and thyroid cancers [57]. However, other management decisions require further research to show the impact of FDG-PET on patient management or added value in the diagnostic pathway for tuberculosis arthritis.
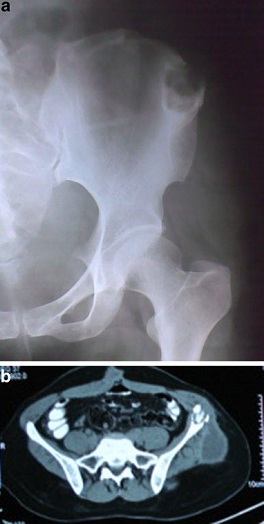 Figure 2: Comparison of imaging of radiography and Computed Tomography (CT).
Figure 2: Comparison of imaging of radiography and Computed Tomography (CT).
PCR testing: Nucleic acid amplification tests using Polymerase Chain Reaction (PCR) cannot differentiate living bacilli from dead bacilli. Thus, these tests continue to give positive results even after successful treatment. PCR is positive in 95% to 100% of culture-positive cases and in 50% to 60% of culture-negative cases [58]. In one study, a multiplex real-time PCR with species-specific primers based on senX3-regX3+IS711 sequences proved to be sensitive and specific and useful for the highly efficient detection of M. tuberculosis and Brucella spp. in different clinical samples [59]. Over the past several decades, the diagnostic methods for M. tuberculosis have improved, and nucleic acid amplification techniques now allow rapid and sensitive detection in clinical settings [60].
Synovial fluid examination: Synovial fluid is usually nonhemorrhagic and turbid, with moderate elevation of the white blood cell count, ranging between 10,000 and 20,000 cells/mL with a predominance of polymorphonuclear leukocytes. Culture for M. tuberculosis should also be planned. Joint fluid aspiration from the affected joint for standard/routine investigation and TB culture is recommended, when possible, for at-risk patients, even if previous cultures have been negative. Synovial fluid culture is positive in approximately 20-40% of cases [61,62]. PCR analysis in synovial fluid, tissue samples, bone marrow aspirate, and peripheral blood is faster and more specific but less sensitive and less widely available [62,63].
Synovial biopsy: The gold standard for the diagnosis of tubercular arthritis is synovial biopsy, with positive results in 80% of cases [64]. It shows caseating granulomas, lymphocyte infiltration, and giant cells with caseation, which are very characteristic of tubercular arthritis.
Management of TB arthritis
The mainstay treatment of TB arthritis is appropriate anti-TB drug therapy (Table 2). Early antimicrobial therapy can result in near-complete resolution and preservation of function. In TB arthritis without pulmonary involvement, the risk of transmission to contacts is minimal and thus constitutes little threat to public health. Antimicrobial therapy in general should be of at least 6-9 months duration but should be continued longer in children and immunocompromised hosts [9,12,13]. The basic principles that underlie the treatment of pulmonary tuberculosis also apply to extra-pulmonary forms of the disease (Table 2) [54]. Although many fewer studies have examined the treatment of extra-pulmonary tuberculosis compared with pulmonary disease, increasing evidence, including some randomized controlled trials, suggests that the 6- to 9-month regimens (2 months of Isoniazid (INH) and Rifampicin (RIF), Pyrazinamide (PZA), and Ethambutol (EMB) followed by 4-7 months of INH and RIF) are recommended as initial therapy unless the organisms are known to be or strongly suspected of being resistant to the first-line drugs. If PZA cannot be used in the initial phase, the continuation phase must be increased to 7 months, as described for pulmonary tuberculosis. Several studies have examined the treatment of bone and joint tuberculosis and have shown that the 6-9-month regimens containing RIF are at least as effective as the 18-month regimens that do not contain RIF [68,70,71]. Myelopathy with or without functional impairment most often responds to chemotherapy. In two Medical Research Council studies conducted in Korea, 24 of 30 patients in one study [70] and 74 of 85 patients in an earlier study [72] had complete resolution of myelopathy or complete functional recovery when treated medically. The recommended dosages are listed in Table 3 [23,54].
| Category of treatment | Category of TB cases | Anti-TB drug regimens | |
| Intensive phase | Continuation phase | ||
| I | New Patient Regimen New smear-positive PTB Smear-negative PTB with extensive Parenchymal involvement Severe forms of EPTB other than TB meningitis |
2HRZE | 4HR |
| II | New Patient Regimen Smear-negative PTB without extensive P Parenchymal involvement Less severe forms of EPTB (e.g., TB cervical adenitis) |
2HRZ | 4HR |
| III | New Patient Regimen TB meningitis |
2HRZSa | 4HR |
| IV | Retreatment regimen Previously treated smear-positive PTB (relapse, treatment after interruption or treatment failure) If low risk for MDR-TB or risk unknown, continue with retreatment regimen If high risk for MDR-TB, use MDR-TB regimen below |
2HRZES/1HRZE | 5HRE |
| V | MDR Regimen MDR-TB |
Individualized regimens | |
E: Ethambutol; EPTB: Extra-Pulmonary Tuberculosis; H: Isoniazid; HIV: Human Immunodeficiency Virus; MDR-TB: Multi-Drug Resistant Tuberculosis; PTB: Pulmonary Tuberculosis; R: Rifampicin; S: Streptomycin; Z: Pyrazinamide; 2HRZ 4HR: Denotes a Two-month Intensive Phase of Daily Isoniazid, Rifampicin, and Pyrazinamide followed by a Four-month Continuation Phase of Daily Isoniazid and Rifampicin
| Recommended | Dose per day | |
| Adults | Children | |
| Drug | Dose and range (mg/kg body weight) |
Dose and range (mg/kg body weight) |
| Isoniazid | 5 (4-6) | 12 (10-15) |
| Rifampicin | 10 (8-12) | 15 (10-20) |
| Pyrazinamide | 25 (20-30) | 35 (30-40) |
| Ethambutol | 15 (15-20) | 20 (15-25) |
| Streptomycin | 15 (12-18) | 15 (12-18) |
Table 3: Recommended doses of first-line anti-tuberculosis drugs for adults [42,60].
CONCLUSIONS
TB is still an important public health problem throughout the world. TB arthritis accounts for approximately 1-3% of all cases of tuberculosis and for approximately 10-11% of extra-pulmonary cases. Nonspinal skeletal TB is rare and insidious in onset and is often difficult to diagnose. Early diagnosis and specific and adequate treatment can improve the possibility of maintaining good joint function.
There is usually a long delay in diagnosis, due partly to its tendency to mimic other diseases as a result of its varied clinical presentation and radiographic appearance [3,67,73]. TB arthritis should be considered in patients who present with indolent symptoms of chronic tenosynovitis [18]. Early diagnosis of arthritis due to TB is essential to preserve the articular cartilage and joint space. The mainstay of treatment is multidrug anti-TB therapy (for 12-18 months) and active-assisted nonweight-bearing exercises of the involved joint throughout the period of healing. Operative intervention (synovectomy and debridement) is required when the patient is not responding after 4-5 months of anti-TB therapy.
REFERENCES
- World Health Organization (2012) Global tuberculosis report 2012. Geneva, Switzerland: WHO.
- Glaziou P, Falzon D, Floyd K, Raviglione M (2013) Global epidemiology of tuberculosis. Semin Respir Crit Care Med 34: 3-16.
- Norbis L, Miotto P, Alagna R, Cirillo DM (2013) Tuberculosis: lights and shadows in the current diagnostic landscape. New Microbiol 36: 111-120.
- Malaviya AN, Kotwal PP (2003) Arthritis associated with tuberculosis. Best Pract Res Clin Rheumatol 17: 319-343.
- Iademarco MF, Castro KG (2003) Epidemiology of tuberculosis. Semin Respir Infect 18: 225-240.
- Narang S (2012) Tuberculosis of the entheses. Int Orthop 36: 2373-2378.
- Samuel S, Boopalan PR, Alexander M, Ismavel R, Varghese VD, et al. (2011) Tuberculosis of and around the ankle. J Foot Ankle Surg 50: 466-472.
- Abdulaziz S, Almoallim H, Ibrahim A, Samannodi M, Shabrawishi M, et al. (2012) Poncet's disease (reactive arthritis associated with tuberculosis): retrospective case series and review of literature. Clin Rheumatol 31: 1521-1528.
- Tuli SM (2002) General principles of osteoarticular tuberculosis. Clin Orthop Relat Res: 11-19.
- Scanzello CR, Goldring SR (2012) The role of synovitis in osteoarthritis pathogenesis. Bone 51: 249-257.
- Poncet A (1897) De la polyarthrites tuberculeuse deformante ou pseudorheumatisme chronique tuberculeux. Congres Francaise de chirurgerie 1: 732.
- Sequeira W, Co H, Block JA (2000) Osteoarticular tuberculosis: current diagnosis and treatment. Am J Ther 7: 393-398.
- Furia JP, Box GG, Lintner DM (1996) Tuberculous arthritis of the knee presenting as a meniscal tear. Am J Orthop (Belle Mead NJ) 25: 138-142.
- Al-Shaikh R, Goodman SB (2003) Delayed-onset Mycobacterium tuberculosis infection with staphylococcal superinfection after total knee replacement. Am J Orthop (Belle Mead NJ) 32: 302-305.
- Varango G, Bamba I, Kodo M, Dao A, Lambin Y (1998) Osteonecrosis of the hip in sickle-cell disease associated with tuberculous arthritis. A review of 15 cases. Int Orthop 22: 384-389.
- Pointud P, Prudat M, Laluque S, Amouroux J (1993) [Tuberculous arthritis and chondrocalcinosis. Apropos of 2 cases]. Rev Rhum Ed Fr 60: 617-620.
- Walker GF (1968) Failure of early recognition of skeletal tuberculosis. Br Med J 1: 682-683.
- Hsiao CH, Cheng A, Huang YT, Liao CH, Hsueh PR (2013) Clinical and pathological characteristics of mycobacterial tenosynovitis and arthritis. Infection 41: 457-464.
- Chan PC, Chang LY, Wu YC, Lu CY, Kuo HS, et al. (2008) Age-specific cut-offs for the tuberculin skin test to detect latent tuberculosis in BCG-vaccinated children. Int J Tuberc Lung Dis 12: 1401-1406.
- Wang PD (2009) Assessment of the need for universal BCG vaccination of children in Taipei. Public Health 123: 74-77.
- Menzies D (2000) What does tuberculin reactivity after bacille Calmette-Guérin vaccination tell us? Clin Infect Dis 3: 71-74.
- Araujo Z, de Waard JH, de Larrea CF, Borges R, Convit J (2008) The effect of Bacille Calmette-Guérin vaccine on tuberculin reactivity in indigenous children from communities with high prevalence of tuberculosis. Vaccine 26: 5575-5581.
- Chen SC, Chen KL, Chen KH, Chien ST, Chen KT (2013) Updated diagnosis and treatment of childhood tuberculosis. World J Pediatr 9: 9-16.
- Dhanasekaran S, Jenum S, Stavrum R, Wiker HG, Kenneth J, et al. (2014) Effect of non-tuberculous Mycobacteria on host biomarkers potentially relevant for tuberculosis management. PLoS Negl Trop Dis 8: 3243.
- Arend SM, Thijsen SF, Leyten EM, Bouwman JJ, Franken WP, et al. (2007) Comparison of two interferon-gamma assays and tuberculin skin test for tracing tuberculosis contacts. Am J Respir Crit Care Med 175: 618-627.
- Ewer K, Deeks J, Alvarez L, Bryant G, Waller S, et al. (2003) Comparison of T-cell-based assay with tuberculin skin test for diagnosis of Mycobacterium tuberculosis infection in a school tuberculosis outbreak. Lancet 361: 1168-1173.
- Brock I, Weldingh K, Lillebaek T, Follmann F, Andersen P (2004) Comparison of tuberculin skin test and new specific blood test in tuberculosis contacts. Am J Respir Crit Care Med 170: 65-69.
- Mori T, Sakatani M, Yamagishi F, Takashima T, Kawabe Y, et al. (2004) Specific detection of tuberculosis infection: an interferon-gamma-based assay using new antigens. Am J Respir Crit Care Med 170: 59-64.
- Mazurek GH, Villarino ME, CDC (2003) Guidelines for using the QuantiFERON-TB test for diagnosing latent Mycobacterium tuberculosis infection: Centers for Disease Control and Prevention. MMWR Recomm Rep 52: 15-18.
- Mazurek GH, Jereb J, Lobue P, Iademarco MF, Metchock B, et al. (2005) Guidelines for using the QuantiFERON-TB Gold test for detecting Mycobacterium tuberculosis infection, United States. MMWR Recomm Rep 54: 49-55.
- Detjen AK, Keil T, Roll S, Hauer B, Mauch H, et al. (2007) Interferon-gamma release assays improve the diagnosis of tuberculosis and nontuberculous mycobacterial disease in children in a country with a low incidence of tuberculosis. Clin Infect Dis 45: 322-328.
- Catanzaro A, Daley C (2013) A summary of the Third Global Interferon-? Release Assay Symposium. Infect Control Hosp Epidemiol 34: 619-624.
- Pai M, Zwerling A, Menzies D (2008) Systematic review: T-cell-based assays for the diagnosis of latent tuberculosis infection: an update. Ann Intern Med 149: 177-184.
- Pai M, Riley LW, Colford JM Jr (2004) Interferon-gamma assays in the immunodiagnosis of tuberculosis: a systematic review. Lancet Infect Dis 4: 761-776.
- Costantino F, de Carvalho Bittencourt M, Rat AC, Loeuille D, Dintinger H (2013) Screening for Latent Tuberculosis Infection in Patients with Chronic Inflammatory Arthritis: Discrepancies Between Tuberculin Skin Test and Interferon-? Release Assay Results. J Rheumatol 40: 1986-1993.
- Perkins MD, Roscigno G, Zumla A (2006) Progress towards improved tuberculosis diagnostics for developing countries. Lancet 367: 942-943.
- Daniel TM (1988) Antibody and antigen detection for the immunodiagnosis of tuberculosis: why not? What more is needed? Where do we stand today? J Infect Dis 158: 678-680.
- Gennaro ML (2000) Immunologic diagnosis of tuberculosis. Clin Infect Dis 3: S243-246.
- Simonney N, Molina JM, Molimard M, Oksenhendler E, Perronne C, et al. (1995) Analysis of the immunological humoral response to Mycobacterium tuberculosis glycolipid antigens (DAT, PGLTb1) for diagnosis of tuberculosis in HIV-seropositive and -seronegative patients. Eur J Clin Microbiol Infect Dis 14: 883-891.
- Papa F, Cruaud P, David HL (1989) Antigenicity and specificity of selected glycolipid fractions from Mycobacterium tuberculosis. Res Microbiol 140: 569-578.
- Hanza M (1994) Joint and spine tuberculosis. Rev Rheum 60:83-86.
- Valdazo JP, Perez-Ruiz F, Albarracin A, Sanchez-Nievas G, Perez-Benegas J, et al. (1990) Tuberculous arthritis. Report of a case with multiple joint involvement and periarticular tuberculous abscesses. J Rheumatol 17: 399-401.
- Enarson DA, Fujii M, Nakielna EM, Grzybowski S (1979) Bone and joint tuberculosis: a continuing problem. Can Med Assoc J 120: 139-145.
- Krama SB, Lee SHS, Abramson SB (2004) Nonvertibral infections of musculoskeletal tuberculosis. In: Rom WN, Garay SM (eds.). Tuberculosis. (2nd edn). Lippincott, William & Wilkins, 577-591.
- Martini M, Ouahes M (1988) Bone and joint tuberculosis: a review of 652 cases. Orthopedics 11: 861-866.
- Spiegel DA, Singh GK, Banskota, Ashok K (2005) Tuberculosis of the musculoskeletal system. Tech Orthop 20: 167-178.
- Wang MN, Chen WM, Lee KS, Chin LS, Lo WH (1999) Tuberculous osteomyelitis in young children. J Pediatr Orthop 19: 151-155.
- Singh M, Moosa NV, Kumar L, Sharma M (2000) Role of gastric lavage and broncho-alveolar lavage in the bacteriological diagnosis of childhood pulmonary tuberculosis. Indian Pediatr 37: 947-951.
- Siddiqi S, Ahmed A, Asif S, Behera D, Javaid M, et al. (2012) Direct drug susceptibility testing of Mycobacterium tuberculosis for rapid detection of multidrug resistance using the Bactec MGIT 960 system: a multicenter study. J Clin Microbiol 50: 435-440.
- den Hertog AL, Daher S, Straetemans M, Scholing M, Anthony RM (2013) No added value of performing Ziehl-Neelsen on auramine-positive samples for tuberculosis diagnostics. Int J Tuberc Lung Dis 17: 1094-1099.
- World Health Organization (2013) Definitions and reporting framework for tuberculosis - 2013 revision. Geneva, Switzerland.
- Zhao P, Fang F, Yu Q, Guo J, Zhang JH, et al. (2014) Evaluation of BACTEC MGIT 960 system for testing susceptibility of Mycobacterium tuberculosis to first-line drugs in China. PLoS One 9: e99659.
- Lawn SD, Mwaba P, Bates M, Piatek A, Alexander H, et al. (2013) Advances in tuberculosis diagnostics: the Xpert MTB/RIF assay and future prospects for a point-of-care test. Lancet Infect Dis 13: 349-361.
- WHO (2010) Treatment of Tuberculosis: Guidelines - 4th edition. World Health Organization, Geneva, Switzerland.
- Sawlani V, Chandra T, Mishra RN, Aggarwal A, Jain UK, et al. (2003) MRI features of tuberculosis of peripheral joints. Clin Radiol 58: 755-762.
- Gehlot PS, Chaturvedi S, Kashyap R, Singh V (2012) Pott's Spine: Retrospective Analysis of MRI Scans of 70 Cases. J Clin Diagn Res 6: 1534-1538.
- Facey K, Bradbury I, Laking G, Payne E (2007) Overview of the clinical effectiveness of positron emission tomography imaging in selected cancers. Health Technol Assess 11: iii-iv, xi-267.
- Papaventsis D, Ioannidis P, Karabela S, Nikolaou S, Syridou G, et al. (2012) Impact of the Gen-Probe Amplified MTD® Test on tuberculosis diagnosis in children. Int J Tuberc Lung Dis 16: 384-390.
- Sanjuan-Jimenez R, Morata P, Bermúdez P, Bravo MJ, Colmenero JD (2013) Comparative Clinical Study of Different Multiplex Real Time PCR Strategies for the Simultaneous Differential Diagnosis between Extrapulmonary Tuberculosis and Focal Complications of Brucellosis. PLoS Negl Trop Dis 7: e2593.
- Kim JH, Kim YJ, Ki CS, Kim JY, Lee NY (2011) Evaluation of Cobas TaqMan MTB PCR for detection of Mycobacterium tuberculosis. J Clin Microbiol 49: 173-176.
- Mattei R, Savarino A, Fabbri M, Moneta S, Tortoli E (2009) Use of the BacT/Alert MB mycobacterial blood culture system for detection of mycobacteria in sterile body fluids other than blood. J Clin Microbiol 47: 711-714.
- Chakravorty S, Tyagi JS (2005) Novel multipurpose methodology for detection of mycobacteria in pulmonary and extrapulmonary specimens by smear microscopy, culture, and PCR. J Clin Microbiol 43: 2697-2702.
- Verettas D, Kazakos C, Dermon A, Petrov H, Galanis V (2003) Polymerase chain reaction for the detection of Mycobacterium tuberculosis in synovial fluid, tissue samples, bone marrow aspirate and peripheral blood. Acta Orthop Belg 69: 396-399.
- Arthanari S, Yusuf S, Nisar M (2008) Tuberculosis of the knee complicating seronegative arthritis. J Rheumatol 35: 1227-1228.
- Al-Qattan MM, Al-Namla A, Al-Thunayan A, Al-Omawi M (2011) Tuberculosis of the hand. J Hand Surg Am 36: 1413-1421.
- Chen SH, Lee CH, Wong T, Feng HS (2013) Long-term retrospective analysis of surgical treatment for irretrievable tuberculosis of the ankle. Foot Ankle Int 34: 372-379.
- Lawn SD, Zumla AI (2011) Tuberculosis. Lancet 378: 57-72.
- [No authors listed] (1999) Five-year assessment of controlled trials of short-course chemotherapy regimens of 6, 9 or 18 months' duration for spinal tuberculosis in patients ambulatory from the start or undergoing radical surgery. Fourteenth report of the Medical Research Council Working Party on Tuberculosis of the Spine. Int Orthop 23: 73-81.
- Anurag D Bhargava, Anand N Malaviya, Ashok Kumar (1998) Tuberculous rheumatism (poncet's disease)-a case series*. Ind J Tub 45: 215.
- [No authors listed] (1993) Controlled trial of short-course regimens of chemotherapy in ambulatory treatmen of spinal tuberculosis: results at three years of a study in Korea. Twelfth report of the Medical Research Council Working Party on Tuberculosis of the Spine. J Bone Joint Surg Br 75: 240-248.
- [No authors listed] (1986) A controlled trial of six-month and nine-month regimens of chemotherapy in patients undergoing radical surgery for tuberculosis of the spine in Hong Kong. Tenth report of the Medical Research Council Working Party on Tuberculosis of the Spine. Tubercle 67: 243-259.
- Pattisson PR (1986) Pott's paraplegia: an account of the treatment of 89 consecutive patients. Paraplegia 24: 77-91.
- Ellis ME, el-Ramahi KM, al-Dalaan AN (1993) Tuberculosis of peripheral joints: a dilemma in diagnosis. Tuber Lung Dis 74: 399-404.
Citation: Babayeva M, Fuzailov M, Rozenfeld P, Basu P (2014) Marijuana Compounds: A Non-Conventional Therapeutic Approach to Epilepsy in Children. J Emerg Med Trauma Surg Care 1: 002.
Copyright: © 2014 Shou-Chien Chen, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

