
Role of Echocardiography in Diagnosis Arrhytmogenic Right Ventricle Dysplasia: A Rare Case
*Corresponding Author(s):
Prima AlmaziniDepartment Of Cardiology And Vascular Medicine, Faculty Of Medicine, University Of Indonesia, Jakarta, Indonesia
Tel:62 21 5684085,
Email:dr.primz@gmail.com
Abstract
Background: Arrhythmogenic right ventricular dysplasia/ cardiomyopathy (ARVD/C) is an inherited cardiomyopathy, clinically characterized by fibrous and fatty replacement of right ventricle and life-threatening ventricular arrhythmias. Heart failure, arrhythmia, and sudden cardiac death are most severe clinical manifestations of ARVD/C. Early and accurate diagnosis of ARVD/C is crucial to prevent lethal ventricular arrhythmias that can occur in the very early stages of ARVD/C before overt structural alterations appear.
Aim: The aim of this case report is to illustrate the role of echocardiography in diagnosis of ARVD/C
Case summary: In this report, a 46 year-old-man was admitted at National Cardiovascular Center Harapan Kita (NCCHK) with the presenting complains of syncope due to ventricular arrhytmia. Patient underwent electrocardiography, echocardiography, and magnetic resonance imaging (MRI) examinations. Electrocardiogram showed epsilon waves and inverted T wave at lead V1 and V2. Echocardiography showed regional aneurysm and decrease of right ventricle function. MRI showed aneurysm and dyskinesia of right ventricle free wall with fibrous and fatty replacement at both ventricles. Following the imaging results and clinical findings, patient fulfilled the 2010 International Task Force of Cardiomyopathy criteria for ARVD/C.
Conclusion: We report a man with clinical presentation arrhythmia who fulfilled the 2010 International Task Force of Cardiomyopathy criteria for ARVD/C. Echocardiography is the first-line method for evaluating patients with suspected ARVD/C in conjunction with other modalities such as electrocardiography and MRI. Echocardiography is a non-invasive imaging tool that has an established role in diagnosis, follow up, and prognosis of ARVD/C patients.
Keywords
Arrhythmia; ARVD/C; Diagnosis; Echocardiography
INTRODUCTION
Arrhythmogenic right ventricular dysplasia (ARVD) is a myocardial disease which is inherited genetically, characterized by replacement of myocardial tissue with fatty and fibrous tissue, involving mainly the right ventricle, from the epicardium and extending to transmural [1].
ARVD disease is inherited with autosomal dominant pattern. Mutations in genes encode desmosomes (plakobin, desmoplakin) which are the proteins that bind cells one to another, found in almost half of ARVD patients[2,3].
The clinical presentation of ARVD patients varies greatly. Heart failure, ventricular arrhythmias and sudden cardiac death are the most severe clinical manifestations of ARVD patients. Early diagnosis is crucial, considering the prognostic implications of the disease and in order to prevent sudden cardiac death. Mortality on patients who have been diagnosed with ARVD and have used implantable cardioverter defibrillator (ICD) is relatively low [4].The diagnosis of ARVD is based on the presence of major and minor criteria from Revised Task Force criteria 2010 [5].
CASE DESCRIPTION
A 46-year-old male patient, admitted to the emergency room (ER) with complaint sudden darkness of his eyesight since 2 hours before come to the hospital. It accompanied by dizziness, but faint denied. Palpitation, chest pain, shortness of breath, headache, fever and limb weakness were denied. The patient have experienced syncope 3 times when he was in elementary school and junior high school. History of sudden death in his family was denied. There were no history of hypertension, diabetes mellitus, smoking, and dyslipidemia.
During observation in the ER, patient was suffering cardiac arrest, got defibrillation 2 times and cardio pulmonary resuscitation (CPR) for 15 minutes, then became return of spontaneous circulation (ROSC). The patient was intubated and transferred to the Intensive Care Unit (ICU).
The patient was compos mentis with an otherwise normal physical examination. Vital signs were blood pressure 91/68 mmHg, heart rate 103 bpm, and respiratory rate 18 times/minute, saturation 98% (room temperature).
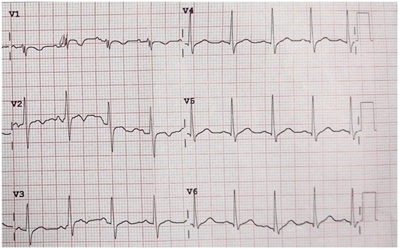 Figure 1: Epsilon wave at V1.
Figure 1: Epsilon wave at V1.
Electrocardiogram showed sinus tachycardia, inverted T wave at V1-V2, and epsilon wave at V1 (Figure 1). Chest x-ray examination showed cardiothoracic ratio was 57% and an upward apex, (Figure 2).
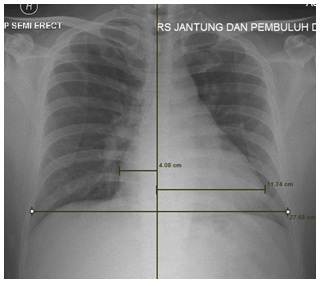 Figure 2: Chest radiograph showed cardiomegaly.
Figure 2: Chest radiograph showed cardiomegaly.
Laboratory tests showed an increase of high-sensitive Troponin T (Hs-trop T) 156 ng/mL and normal electrolyte levels. While in the ER, the patient was diagnosed with a history of cardiac arrest e.c.Ventricle Tachycardia (VT), Suspect ARVD. The patients were given bisoprolol 1×5 mg in the ER then was transferred to the intermediate ward (IW). At the IW, the patient had no complaint, blood pressure 109/60 mmHg, heart rate 79 bpm. Bisoprolol 1×5 mg was replaced by metoprolol 2×100 mg.
The patient underwent echocardiography. The results showed normal LV size and function, TAPSE 1.1 cm, right ventricular dilatation, right ventricle (RV) free wall hypokinetic, parasternal right ventricular outflow tract (RVOT) diameter at short axis (PSAX) of 36 mm, diameter RVOT parasternal long axis (PLAX) of 39 mm, basal right ventricular diameter of 3.8 cm, mid ventricular right diameter of 4.4 cm, long RV basal apex of 6.2 cm. The results met the criteria of major ARVD based on Revised Task Force criteria 2010. The patient was then transferred to ward(Figure 3).
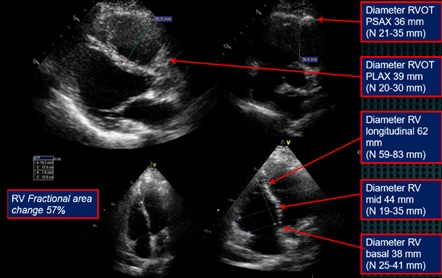 Figure 3:Echocardiography findings.
Figure 3:Echocardiography findings.
At the ward, the patient had no complaint. Blood pressure 107/71 mmHg, heart rate 67 bpm. Patient underwent magnetic resonance imaging (MRI). MRI results showed there were right ventricular wall aneurysm and protrusion of right ventricular wall in systolic phase (Figure 4). In the T1 sequence, there was mild dilatation of RVOT diameter compared to left ventricle outflow tract (LVOT) diameter. Aneurysm dilatation in the RVOT area with fat infiltration. There was significant fat infiltration along the right ventricular wall. In the delayed enhancement sequence there were fibrosis in the endocardium of right ventricular wall. Fibrosis were also seen in the anterior and anteroseptal walls of left ventricle. The fibrosis was not associated with coronary heart disease nor myocarditis. The conclusion of the findings supported ARVD diagnosis with involvement of left ventricular.
Before discharge, patient had no complaints. The patient was discharged at the 6thday of hospitalization. Therapy at discharge was metoprolol 2×100 mg. Patient was scheduled for electrophysiological examination and implantation of ICD. Patient suggested to bring his family members for screening of ARVD.
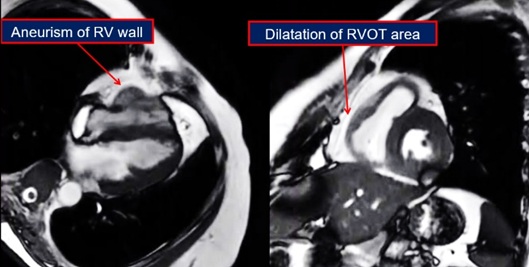 Figure 4: MRI findings.
Figure 4: MRI findings.
DISCUSSION
The patients of ARVD usually has VT as a first sign of the disease. This tachycardia is not only observed in ARVD, it may be an idiopathic origin and can be seen in patients with CAD, right or biventricular cardiomyopathy, sarcoidosis and congenital heart disease. Clinical symptoms, history, and imaging exams usually can distinguish ARVD with other diseases.
Clinical symptom
ARVD can cause heart failure, arrhythmias and sudden death. Symptoms usually begin in the 2nd or 3rd decade. Because the spectrum is broad, each person has different clinical feature and the diagnosis become difficult. Early diagnosis and immediate treatment are not only can save life but also improve the quality of life[6].
The most often clinical presentation of ARVD patients are heart failure, arrhythmias, sudden death. Patients can be asymptomatic or come with presentation of severe right heart failure. The spectrum of arrhythmia is very wide, from premature ventricular complexes to ventricular tachycardia and ventricular fibrillation. The premature ventricular contraction usually has the right axis and can be multifocal. The incidence of sudden death is significantly high, explaining how importance this disease. ARVD is responsible for 17%-20% of sudden death in young adults. The sudden death rate is around 1% per year. There is usually a history of syncope in many cases of sudden death[7].
Diagnostic approach
History of palpitations especially at a young age and history sudden death in families at a young age, both increase suspicion of ARVD. Physical examination is usually normal in 50% of patients with ARVD. A murmur is rarely found. The diagnosis of ARVD is often made after work-up of tachycardia in healthy adult. 50% to 90% of ARVD patients have special characteristic of ECG at rest. This characteristics includes the T wave inverted on anterior precordial leads (V1 to V6), epsilon wave, or Ventricular Tachycardia (VT). Epsilon wave is a small deflection after the QRS complex (Figure 5). This wave most commonly seen on leads V1 to V3. Every potential in leads V1 to V3 that exceed the QRS duration on V6 leads over 25 milliseconds, must be considered an epsilon wave.
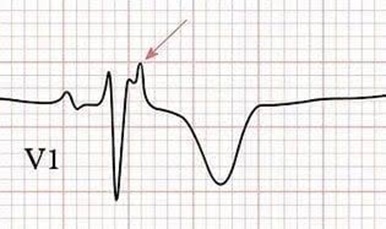 Figure 5: Epsilon wave.
Figure 5: Epsilon wave.
The preferred method for establishing a diagnosis of ARVD is histological examination. Unfortunately, the sensitivity of this examination is very low. After the suspicion of ARVD arise, consultation to cardiologist is recommended so that further tests can be carried out for exclude other diagnoses. Examinations include holter, exercise training, x-ray chest, echocardiography and MRI. Invasive examinations include right ventricular angiography, contrast echocardiography, electrophysiology, and endomyocardial biopsy.
Holter and exercise training are usually normal. Furthermore, exercise training test can trigger VT. Chest radiography is usually normal except if there is extensive ventricular enlargement. Contrast echocardiography and right ventricular angiography can identify ventricular aneurysm and area of dyskinesia but requires invasive techniques.
 Figure 6: 2010 Revised Task Force criteria[5].
Figure 6: 2010 Revised Task Force criteria[5].
Revised task force criteria
ARVD was diagnosed clinically based on structural, histological, electrocardiographic, arrhythmia, and family history. All criteria are divided into major and minor categories based on its specificity. The 1994 criteria focused on manifestation of abnormalities in the right ventricle without or with left ventricular involvement.
The diagnostic criteria was revised in 2010, two major or 1 major and 2 minor are needed to established diagnosis. This guideline defines "borderline" as 1 major and 1 minor or 3 minor and defines "possible” as 1 major or 2 minor (Figure 6). Major and minor criteria are obtained from six class categories, 1-regional/global functional and structural changes, 2-tissue characteristic, 3-repolarization abnormality, 4-conduction/depolarization abnormality, 5-arrhythmia, 6-family history [5].
Functional or structural changes criteria can be assessed with 2D echocardiography, MRI and right ventricular angiography. The tissue characteristic can be obtained from the biopsy. Repolarization and depolarization/conduction abnormalities can be observed from ECG and signal-averaged ECG (SAECG). ARVD diagnosis criteria are the integration of several information from disease history, physical examination, ECG, SAECG, echocardiography, 24-hour holter, cardiac MRI, CT angiography, endomyocardial biopsy, and electrophysiology.
In this patients there are (1) structural abnormalities and regional right ventricular dysfunction, RVOT dimension ≥ 32 mm in PLAX view and ≥ 36 mm in PSAX view from echocardiography, (2) right ventricular regional dyskinesia and reduction in ejection fraction > 40% up to ≤ 45% from MRI, (3) repolarization abnormalities, there was inverted T waves in leads V1 and V2 without complete right bundle branch block on electrocardiography (4) depolarization abnormalities, there was epsilon wave at V1.
Based on 2010 Revised Task Force criteria, the definitive diagnosis of ARVD in this patient was made because it met 2 major criteria and 2 minor criteria. Furthermore, ARVD in this patient has involved the left ventricle. This is accordance with literature that ARVD can involve not only the right ventricle but also left ventricle.
Role of echocardiography
Echocardiographic parameters are part of the diagnostic criteria in 2010 Revised Task Force criteria and consists of visual analysis of wall movement abnormalities, RVOT dimensions, and FAC. Akinetic, dyskinetic or aneurysmatic of right ventricular wall are movement disorder that are included in the criteria. Hypokinesis is not included in the 2010 Revised Task Force criteria. Regional hypokinesia is common in healthy individuals [5].
An abnormality of regional walls must be obtained to determine the major or minor criteria besides RVOT dilatation and or diastolic dysfunction. Therefore, the optimal visual image analysis of right ventricular wall movements is crucial and recommended to visualize the entire right ventricular region during examination [8].
Echocardiography with contrast can be used to improve visualization right ventricular wall border and accuracy of ventricular wall movement analysis when the image quality is suboptimal. Echocardiographic parameters was chosen as one of diagnostic criteria because of its high specificity (76 - 95%).
The abnormal findings that do not meet the 2010 Revised Task Force criteria can either be examined further with MRI or with repeat echocardiography after 1 year. This recommendation is based on daily practice and not based on literature [9]. In addition to the diagnostic role of echocardiography in ARVD, prognostic parameters are also has been studied. Both TAPSE and RV-FAC decreases are associated with events major cardiac disease in ARVD. Both parameters are easy to obtain and add risk stratification of ARVD patients. Decreased of left ventricle ejection fraction has been reported as the strongest independent predictor of clinical outcomes[9].
The re-echocardiographic evaluation interval in ARVD patients is unknown. One study recommends echocardiographic examination if clinical changes occurred (such as heart failure, syncope, and arrhythmia). In stable patients without clinical changes, serial echocardiographic every 4-5 years can be carried out.
CONCLUSION
A male patient was reported with a clinical presentation of arrhythmias. ECG, echocardiography, and MRI examinations were conducted and met the diagnostic criteria of ARVD based on 2010 Revised Task Force criteria. Noninvasive imaging of right ventricular using echocardiography plays an important role as a first-line diagnostic tool for establishing the diagnosis of ARVD, together with other examinations such as ECG and cardiac MRI. Echocardiography can be used for diagnostic, follow-up, and providing useful information regarding prognostic in ARVD patients.
REFERENCES
- Basso C, Corrado D, Marcus FI, Nava A, Thiene G (2009) Arrhythmogenic right ventricular cardiomyopathy. The Lancet 373:1289-1300.
- Cox MG, van der Zwaag PA, van der Werf C, van der Smagt JJ, Noorman M, et al. (2011) Arrhythmogenic right ventricular dysplasia/cardiomyopathy: Pathogenic desmosome mutations in index-patients predict outcome of family screening: Dutch arrhythmogenic right ventricular dysplasia /cardiomyopathy genotype-phenotype follow-up study. Circulation 123:2690-2700.
- Fressart V, Duthoit G, Donal E, Probst V, Deharo JC, et al. (2010)Desmosomal gene analysis in arrhythmogenic right ventricular dysplasia /cardiomyopathy: Spectrum of mutations and clinical impact in practice. Europace12: 861-868.
- Dalal D, Nasir K, Bomma C, Prakasa K, Tandri H, et al. (2005) Arrhythmogenic right ventricular dysplasia: A United states experience. Circulation 112:3823-3832.
- Marcus FI, McKenna WJ, Sherrill D, Basso C, Bauce B, et al. (2010) Diagnosis of arrhythmogenic right ventricular cardiomyopathy/dysplasia: Proposed modification of the task force criteria. Circulation 121:1533-1541.
- McKenna WJ, Thiene G, Nava A, Fontaliran F, Blomstrom-Lundqvist C, et al. (1994) Diagnosis of arrhythmogenic right ventricular dysplasia/ cardiomyopathy. Task Force of the Working Group Myocardial and Pericardial Disease of the European Society of Cardiology and of the Scientific Council on Cardiomyopathies of the International Society and Federation of Cardiology. Br Heart J 71: 215-218.
- Ulucam MZ (2013) Confusing aspects of arrhythmogenic right ventricular dysplasia: current clinical review. World J Cardiovasc Dis 3: 154-158.
- Rudski LG, Lai WW, Afilalo J, Hua L, Handschumacher MD, et al. (2010) Guidelines for the echocardiographic assessment of the right heart in adults: a report from the American Society of Echocardiography is endorsed by the European Association of Echocardiography, a registered branch of the European Society of Cardiology, and the Canadian Society of Echocardiography. J Am SocEchocardiog23: 685-713.
- Mast TP, Teske AJ, Doevendans PA, Cramer MJ (2016) Current and future role of echocardiography in ARVD/C. Cardiol J 22: 362-374.
Citation: Almazini P, Sukmawan R, Rudiktyo E,Ariani R,AryoBRM, et al. (2019) Role of Echocardiography in Diagnosis Arrhytmogenic Right Ventricle Dysplasia: A Rare Case. J Emerg Med Trauma Surg Care 6: 032.
Copyright: © 2019 Prima Almazini, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

