
Application of Dental Morphological Characteristics for Medical-Legal Identification: Sexual Diagnosis in a Portuguese Population
*Corresponding Author(s):
Cristiana Palmela PereiraFaculty Of Dentistry, Portuguese Naional Institute Of Legal Medicine And Forensic Sciences, Centre Of Statistics And Applications, University Of Lisbon, Portugal
Tel:+351 218811819,
Email:cristiana.pereira@fmd.ul.pt
Abstract
The main purpose of this work is to investigate the application of dental morphological features as a potential qualitative method of sex classification in medico-legal identification. The classification of 16 non-metric dental traits was performed by ASUDAS (Arizona State University Dental Anthropology System) method. The traits displayed exclusively by the crowns of permanent teeth were analyzed on dental casts of a Portuguese population sample consisting of 53 male and 57 female individuals.
No significant differences (p<0.05) were found between males and females for all investigated traits, except for the dental tubercle of tooth 13, for the metaconule of tooth 27 and the canine distal accessory ridge of teeth 33 and 43. Based on the method applied, tooth 43 has proven to be the most dimorphic among the Portuguese population. Further, a gender classification model using significant different traits between both sexes of the teeth 13, 27 and 43 was developed, and it proved to have a reasonable gender classification reliability (with 76.4 percent of correct classification, Cox & Snell’s pseudo R2 equals to 0.344 and Nagelkerke’s pseudo R2 equals to 0.458) although with a significant probability of misclassification.
Keywords
INTRODUCTION
Diagnosis of gender is a significant step in reconstructive postmortem profiling of medico-legal personal identification [1].
Sex classification is a complicated procedure, especially in scenarios of severe mutilation or advanced decomposition, where bodies are damaged beyond recognition and also in cases of non-adult skulls whose sexual characteristics have not yet been developed. In these circumstances, teeth can play an important role as a source of sex information [2,3].
In humans, although only two biological sexes exist, sex differentiation through morphological methods is difficult [2,4,5]. However, the examination of skeleton and dental morphological features represents a method that may help to build the biological profile of an unknown individual, serving as alternative and complementary elements useful in sex discrimination [6-10].
Even though dental sexual dimorphism has been mostly studied by means of odontometric analysis, tooth morphology also represents a valid qualitative method for its study [11-14].
The investigations in dental morphology context seek to observe record, analyze and understand the behavior of the expression (frequency, variability and degree of expression) of the coronary and radicular morphological characteristics of human teeth. These dental traits can take different phenotypic presentations of dental enamel, whose expression is thought to be regulated by the genome of an individual and population during odontogenes is; these can appear in forms of positive structures (such as cusps) or negative (as grooves and depressions). These distinct features or traits have the potential to be present or not in a specific location, occur in one or several individuals within the same population group and, when present, exhibit variable degrees of expression while having both a conservative and a dynamic evolution [15-19].
So far, there are over 100 coronary and radicular dental traits recognized in the human dentition. The excelling analysis method of these features reported in the literature is the ASUDAS method developed at the Department of Anthropology of the Arizona State University. This observational method provides a scoring system to classify the degree of expression of dental morphological variants allowing to evaluate them beyond the dichotomy presence or absence [13,20].
OBJECTIVES
The main purpose of this experimental study was to test the possibility of a correct sexual diagnosis using a qualitative method, namely through the analysis of dental morphological traits.
This identification procedure of forensic character was carried out on a sample of the Portuguese population gathered at the School of Dentistry, University of Lisbon.
About 16 Non-metric Dental Crown Traits (NDCT) were investigated on 24 permanent teeth resulting in multiple experimental hypotheses (66) to be tested.
MATERIALS AND METHODS
The sample used in this study consisted of dental casts selected from 110 patients of the Orthodontics Post-Graduate Specialization Course, after following the ethical approval by the Ethics Committee for Health of the School of Dentistry, University of Lisbon.
The participants were of Portuguese nationality and Portuguese origin (data obtained from individual records of the clinical process) and had Caucasian population affinity (obtained through the cover photo of the clinical process). Also the presence of permanent teeth (except third molars) as well as the absence of teeth affected by congenital anomalies were inclusion criteria for this study. No restriction of age was applied.
For the observation of 16 NDCT in the permanent dentition (Tables 1 and 2) this study used the Arizona State University Dental Anthropology System (ASUDAS).Each of the 110 cases was assigned a corresponding code number to the clinical process, not only for the observer to be unaware of the sex of the participants during observations (single-blinded), as well as to facilitate further processing data and in order not to disclose identifying information.
| Nonmetric dental crown trait | Teeth |
|
Labial convexity Shoveling Tuberculum dentale |
12, 11, 21, 22 13, 12, 11, 21, 22, 23 13, 12, 11, 21, 22, 23 |
|
Canine mesial ridge Canine distal accessory ridge |
13, 23 13, 23 |
|
Metacone Hypocone |
17, 16, 26, 27 17, 16, 26, 27 |
|
Cusp 5 (Metaconule) Carabelli’s Trait |
17, 16, 26, 27 17, 16, 26, 27 |
| Parastyle | 17, 16, 26, 27 |
Table 1: Dental morphological traits observed in permanent dentition - Upper arch.
| Non-metric dental crown trait | Teeth |
|
Shoveling Canine distal accessory ridge |
41, 42, 31, 32 43, 33 |
|
Premolar lingual cusp variation Anterior Fovea |
45, 44, 34, 35 46, 36 |
|
Deflecting wrinkle Cusp 5 (Hypoconulid) |
46, 36 47, 46, 36, 37 |
|
Cusp 6 (Entoconulid) |
47, 46, 36, 37 |
| Cusp 7 (Metaconulid) | 47, 46, 36, 37 |
Table 2: Dental morphological traits observed in permanent dentition - Lower arch.
The observation sessions took place in a room with natural light and were macroscopically performed without magnification lens (Figure 1).The study counted only with the participation of one observer and was performed over approximately 2 months. Inorder to minimize potential eye strain of the viewer that would compromise the following observations, small breaks (10 minutes) were taken between each characteristic assessed during the data collection.
 Figure 1: UI2 shovel plaque for scoring upper lateral incisors and upper canines. Each of the ASUDAS standard reference plaques gives a physical representation of minimal and maximal characteristic expression and various gradations between these two points.
Figure 1: UI2 shovel plaque for scoring upper lateral incisors and upper canines. Each of the ASUDAS standard reference plaques gives a physical representation of minimal and maximal characteristic expression and various gradations between these two points.
All NDCT were classified by its degree of expression and data were registred in a form developed for this purpose. Permanent teeth were named according to the FDI system.
All data resulting from observations were collected by the same observer. Thus, it was intended to introduce the least possible bias in a ssessments; simultaneously introducing interobserver variability was excluded. Each observation session of the dental morphological traits followed the same methodological criteria set for its completion.
However, it was necessary to estimate the intraobserver error. Thus, the observations were repeated at 10% of the sample after a time interval of more than1 month in order to suppress the memory effect.
The data obtained from the NDCT observation from each of the 110 cases were first entered into a template on Excel® sheet and statistical analyses was performed using the IBM® SPSS Statistics® Software, version 22.0. The 16 NDCT combined with the permanent teeth observed (all except third molars, often un erupted or extracted, and upper premolars, frequently extracted in orthodontic cases and due to the fact that the NDCT proposed by the ASUDAS for these teeth were considered irrelevant to include in the study) resulted in 66 independent variables (placed as experimental hypothesis) plus the variable sex.
Descriptive statistics was presented (distribution of the sample population by sex, distribution of all of teeth examined, frequencies of all independent variables and descriptive analysis of each variable by gender) and non-parametric tests were applied (Mann-Whitney U and Wilcoxon). The contingency coefficient was determined and the kappa coefficient was assessed. The conventional 5% level (p<0.05) was considered to be statistically significant.
In addition, abinomial logistic regression allowed modeling the probability of success for the reference category female, based on the development of a model for gender classification with 3 selected variables which showed significant differences among both sexes.
RESULTS
Descriptive statistics
Intra-observer error:
There were no significant differences observed between the two measurements except for the variables presented in table 3. Although only 3 variables had significant disagreement in scoring the NDCT, their individual values obtained for the kappa coefficient were far from a perfect level of concordance (1).
| Independent variable | Tooth |
Wilcoxonp <0.05 |
Kappa coefficient value |
Strength of agreement (Altman, 1991) |
|
Labial convexity Anterior Fovea |
12* 36 |
0.034 0.034 |
0.146 0.314 |
Poor Fair |
| Anterior Fovea | 46 | 0.034 | 0.302 | Fair |
Sexual dimorphism and bilateral symmetry
| Independent variable | Tooth | Mann-Whitney | Contingency coefficient | |
| p<0.05 | ||||
| Value | p<0.05 | |||
| Tuberculum dentale | 13 | 0.048 | 0.403 | 0.006 |
| Tuberculum dentale | 23 | 0.206 | 0.413 | 0.007 |
| Cusp 5 (Metaconule) | 17 | 0.056 | 0.299 | 0.148 |
| Cusp 5 (Metaconule) | 27 | 0.032 | 0.254 | 0.372 |
| Canine distal accessory ridge | 33 | 0.022 | 0.333 | 0.089 |
| Canine distal accessory ridge | 43 | 0.008 | 0.343 | 0.023 |
Let us notice that table 4 contains symmetrical teeth. This may occur because the symmetrical teeth may include the same kind of information about the individual’s gender. Therefore, the analysis of dental bilateral symmetry was executed based on a non-parametric test matched by pairs of antimere teeth (Wilcoxon) for the variables with significant differences between male and female. As evidenced by the results (Table 5), statistically none of the pairs were significantly different at the 5% level. Thus, in the gender classification model discussed in the next subsection it will only be used one tooth of each pair of symmetry (the most significant between the two), in order to obtain a more parsimonious model.
| NDCT | Pair | Wilcoxon |
| p<0.05 | ||
|
Tuberculum dentale Cusp 5 (Metaconule) Canine distal accessory ridge |
13-23 17-27 33-43 |
0.551 0.961 0.174 |
Table 5: Bilateral symmetry of the pairs of antimere teeth.
Gender classification analysis
Through a binomial logistic regression and by taking the variables of the permanent canines (above mentioned in table 4) with significant differences between both sexes (without using symmetric teeth), the developed model uses the information from three teeth: the tuberculum dentale of tooth 13, the cusp 5 (Metaconule) of tooth 27 and the canine distal accessory ridge of tooth 43 (Figure 2).
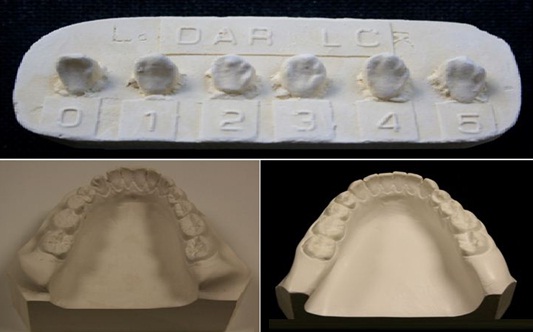 Figure 2: DAR LC plaque for scoring Canine distal accessory ridge on lowers canines. Each of the ASUDAS standard reference plaques gives a physical representation of minimal and maximal characteristic expression and various gradations between these two points for each gender.
Figure 2: DAR LC plaque for scoring Canine distal accessory ridge on lowers canines. Each of the ASUDAS standard reference plaques gives a physical representation of minimal and maximal characteristic expression and various gradations between these two points for each gender.However, the Pseudo R square proved to be low, the Cox and Snell’s pseudo R2 value obtained was 0.334 and the Nagelkerke’s pseudo R2 equals to 0.458, thus the model did not show great potential to gender classification although it predicted 76.4 percentage correct. Nevertheless, 24.6 percent of the females and 22.6 percent of the males are misclassified.
In short, even though the model display significant differences between genders, the used information does enable a reliable gender classification model, with low probability of misclassification (the sensitivity and the specificity of the model are around 75 percent) (Table 6).
| Predicted | |||
| Gender | Percentage | ||
| Observed | Male | Female | Correct |
| Male | 41 | 12 | 77.4 |
| Female | 14 | 43 | 75.4 |
| Overall Percentage | 76.4 | ||
DISCUSSION
Integrity of the experimental methodology
The experimental procedure consisted of an observational non-destructive qualitative method, easy to perform without leading to tissue damage or using an invasive technique, thus ensuring the physical integrity of the dental pieces [21], in this case the plaster models.
The random sample counted with a total of 110 cases with a minimum of 50 individuals for each group (sex). However, with little time and more limited resources, a hundred individuals are considered a viable sample to provide a good characterization of the population [13].
The cases were selected between the existing plaster models of the orthodontics department of the Faculty of Dentistry from University of Lisbon according to the inclusion criteria, thus absent of a true randomization.
The ideal sample size of a study is determined by the level of significance, the power and the magnitude of the differences to be detected between groups (male and female). By setting up a statistical significance level of 5%, a smaller sample size leads to a reduced statistical power. The main consequence of this reduction is the loss of ability to detect statistically significant differences between groups. For this, the author is aware that data collection should be planed to take sized nM (male) and nF (female) sub samples such that, with a preassigned probability, differences greater than a threshold (bound, “B”) could be detected. Therefore, the half width of the confidence interval (making a Gaussian approximation, legitimate at t(γ) with γ≥30),
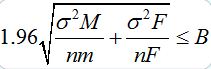
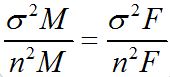
Plaster models have advantages for the study of dental morphology, they may be obtained from current populations, as was the case in this study, and they allow to define the sample size and age groups depending on the intended goals without being dependent on factors such as post mortem tooth loss. The main disadvantage is clearly the restricted morphological observation of only one part of the teeth, meaning the crowns [22]. Furthermore, a significant fraction of the sample may be lost, for example due to phenomena such as dental tooth wear, carries, congenital anomalies, the presence of dental fillings, dental trauma fractures, dental extractions, dental agenesis and also by influence of environmental factors [23]. Yet the study of dental morphological characteristics in skeletonized remains has the great advantage to include the study of root morphology in the research protocol, even in the absence of dental structures. However, 100 human skeletons do not provide a sample of 100 morphological features to be observed [22].
In the population sample, 22% of the observations failed in classifying the degree of expression of the morphological characteristics determined as variables, mainly due to individuals with absent teeth, the presence of partially erupted teeth and restorations. The first two mentioned reasons refer to the fact that many plaster models belonged to individuals under the age of 12 years with mixed dentition. Accordingly, the selection of morphological characteristics to be assessed in the experimental design could have been more embracing by including also the analysis of dental morphology in primary teeth given the fact that they count as precursors in the development of the permanent ones, representing a possible omen of morphological features later displayed by the permanent dentition [22]. Restorations were observed mostly in teeth with a more complex occlusal anatomy (molars), especially in the first permanent molars probably because these had been for the longest in the oral cavity. Furthermore, the younger population currently requires a greater number of dental treatments and has a higher caries experience than previous generations [24]. Considering that the population of this sample was selected from individuals who attend or attended the orthodontics department, several plaster models showed teeth with orthodontic bands or brackets, fixed orthodontic appliance, fixed containment device and dental crowding. There were also cases where the plaster models were not in perfect condition and, in certain cases, the morphological characteristics were not noticeable as a consequence of dental wear. All these conditions together ultimately reduced the potential of the dental morphology of the sample.
The observation in plaster models (the ones included in the study securely placed in an individual box) or directly in teeth require conditions such as an adequate and well-lit environment, preferably using a magnifying lens [22]. This tool was not used and all observations were made macroscopically by a single observer implying a detailed and deep examination of the characteristics from all possible angles, to prevent more subtle features to be unnoticed. Analyzing dental morphological characteristics directly has its drawbacks (as material defects), but the reflection of light by the enamel makes the observation of traits less obvious and more difficult. Scott increases that the analysis of tooth morphology in an intra-oral environment in living individuals is not ideal for two reasons: the reflection of light is exacerbated by the presence of saliva and various observation perspectives become limited in the oral cavity [22].
Data obtained by dental qualitative assessment sin grades have a great inter observer error [25]. In this study the data were all registered by the same investigator-the author. Dental qualitative assessments were established by criteria strictly defined before the study, through a careful definition of the variables. Using only one investigator was advantageous in order to limit the introduction of various non quantifiable or controllable factors.
DISCUSSION OF THE RESULTS
Sexual dimorphism: Through analysis of teeth, it is possible to study the sexual dimorphism of an individual from the patterns of dental development and eruption, the expression of the amelogen in protein, dental morphology and dental dimensions [13]. Once formed, teeth do not undergo changes in morphology like bones, but non-metric dental crown traits which are phenotypic forms of the enamel (expressed and regulated by the human genome of each individual) can disappear due to dental wear and as a consequence of certain oral pathologies [15,23,26].
In this study, the characteristics which showed significant differences between male and female were in tooth 13 (tuberculum dentale), tooth 27 (cusp 5/metaconule) and in teeth 33 and 43 (canine distal accessory ridge). However, Rocha et al., [27] achieved significant results for the existence of sexual dimorphism for the shoveling characteristic in the maxillary central incisors, using the ASUDAS method in the permanent dentition in a sample of 42 Colombian women and 42 Colombian men.
Scott and Turner fall within the group of researchers who claim that dental morphological characteristics are equally expressed between both sexes and that the sexual dimorphism exhibited by them is low or nonexistent. In studies of Goyes et al., [8] and Díaz et al., [28], both applied the ASUDAS method in the permanent dentition in different samples of the Colombian population, whose results demonstrated the absence of sexual dimorphism for the tuberculum dentale of the maxillary canines and the distal accessory ridge of the mandibular canines in the first study and, for cusp 5/metaconule for the second study mentioned. Such contradictory facts can reside on the assumption that in both cases the number of the sample was below the population of the present study, or that dental morphological characteristics are specific to a given population, or even because of the existing geographical distance separating population groups. Genetic and environmental factors may also be an explanation for these facts, although it is suggested that tooth morphology, in comparison with tooth size, is less susceptible to external effects [23,29].
As observed in the current study, other researchers concluded that there is no evidence of sexual dimorphism in the meta cone, hypo cone, Carabelli’s trait, parastyle, canine mesial ridge, labial convexity, shoveling, deflecting wrinkle, cusp 6/entoconulid and cusp 7/metaconulid when applying the ASUDAS method [15,28,30-32]. Reality behind these findings may be, again, due to a low number of individuals in each subpopulation group (gender), including the present study. A possible systematic reduction of sexual dimorphism throughout human evolution, combined with a gradual process of simplification of dental morphology, may have lead to trends increasingly monomorphic [9]. There may be also an association with a line of evolution of the species in the way that currently, a phenomenon related to the gracilization of males phenotypic characteristics, tend to occur [25]. However, according to Moreno-Gómez, contemporary human beings are dimorphic, although less than other hominids, contributing dental morphological traits (especially those of the canines) to this fact.
Dental sexual dimorphism studied by means of odontometric analysis have shown statistically significant differences in the permanent dentition for the canines, especially the mandibular ones [25,33-39]. Although the present study has employed a qualitative method, results are in agreement with this evidence. Based on the results obtained for the canine distal accessory ridge, teeth 33 and 43 were the most dimorphic in the population sample. Also Dumancic et al., [40], who devoted a study exclusively to the canine distal accessory ridge applying the ASUDAS method in a Croatian population, showed that the mandibular canine is a better sexual indicator than the maxillary canine in the permanent dentition. According to Scott [22], the canine distal accessory ridge is considered the NDCT with more dimorphic among the human dentition, displaying frequencies and forms of more pronounced expression in males, as verified by the results of the present study.
From a macro evolutionary point of view, it is known that canine teeth are less influenced by environmental factors and have greater stability in their morphogenetic field, hence the possible explanation for the higher degree of sexual dimorphism [7,13]. Nevertheless, further investigations are needed to clarify which genetic factors and sex hormones are directly involved in the process of morphogenesis, in order to better understand the presence/absence of various dental morphological characteristics in each sex, as well as the more pronounced forms of expression observed in males.
Dental symmetry: The pairs of antimere teeth (13-23/tuberculum dentale, 17-27 /metaconule and 33-43/canine distal accessory ridge) did not show bilateral asymmetry in the dental morphological characteristics between the right and the left side of the dental arches.
These results corroborate those of other studies in which there was a symmetric expression of the dental morphological characters [6,15,27,41]. In all, the ASUDAS method was employed in the permanent dentition. However, the dental morphological characteristics investigated were not the same, as well as the studied population. Thus, and in spite of the current study did not focus on the same dental morphological traits, it seems that a general symmetry of pairs of homologous teeth exist. The explanation more plausible for the observed bilateral symmetry is related to the gene influence that acts identically on opposite sides of the dental arch [42].
Accordingly, and with regard to the goals of this work, there is no gain in using two symmetrical teeth in the gender classification model since they contain similar information.
ASUDAS method reliability in sex classification: The attempt to model the probability of success for the female sex, by taking dental morphological characteristics of permanent teeth (13, 27 and 43) to elaborate a model, was innovative. But, despite having obtained more than 75% correct classifications, this did not fully fit the desired goal by presenting a significant probability of misclassification (23.6%).
CONCLUSION
The results proved the existence, albeit not as significant as it was intended, of sexual dimorphism in dental morphological characteristics of permanent teeth in a sample of the Portuguese population. These not only represent sexually dimorphic phenotypic structures, as may be specific to a particular population. The distal accessory ridge of the mandibular canines, proved to be the NDCT with more evidence of sexual dimorphism, showing frequencies and more pronounced forms of expression in males. Based on the method applied, tooth 43 has proven to be the most dimorphic and best sexual indicator among the Portuguese population through the NDCT canine distal accessory ridge.
However, the ASUDAS method has showed a not significant reliability to be used as a means of sexual diagnosis alone. The classification model elaborated with the dental morphological crown characteristics of teeth 13, 27 and 43 proved to be reasonable (76.4%) to estimate successfully for female (23.6% of misclassification)
Future work should include larger samples and comparative studies between distinct population groups to provide a greater reliability of this qualitative method of sex diagnosis, for the purpose of forensic application.
ACKNOWLEDGMENTS
Fundação Nacional para a Ciência e Tecnologia, Portugal - FCT under the project (PEst-OE/MAT/UI0006/2014), based on the research project for the integrated master thesis in Dental Medicine from the first author
REFERENCES
- Zorba E, Moraitis K, Manolis SK (2011) Sexual dimorphism in permanent teeth of modern Greeks. Forensic Sci Int 210: 74-81.
- Rösing FW, Graw M, Marré B, Ritz-Timme S, Rothschild MA, et al. (2007) Recommendations for the forensic diagnosis of sex and age from skeletons. Homo 58: 75-89.
- Pereira C (2012) Medicina Dentária Forense. Lidel, Lisbon, Portugal.
- Iscan MY (2005) Forensic anthropology of sex and body size. Forensic Sci Int 147: 107-112.
- Fortes de Oliveira O, Lima Ribeiro Tinoco R, Daruge Júnior E, Silveira Dias Terada AS, Alves da Silva RH, et al. (2012) Sexual dimorphism in Brazilian human skulls: discriminant function analysis. J Forensic Odontostomatol 30: 26-33.
- Girón G, Gómez P, Morales L, León M, Moreno F (2009) Morphologic and Metric Dental Crown Traits of Upper and Lower Premolars in Scholars of Cali, Colombia. Int J Morphol 27: 913-925.
- Goyes J, Guerrero L, Narváez N, Moreno F (2011) Non-metric Dental Traits of Deciduous and Permanent Canines in a Group of Mixed Ethnicity Population in Cali, Colombia. Revista Colombiana de Investigación en Odontología 2: 1-13.
- Monali C, Pritam P, Tapan M, Kajal D (2011) Gender determination: a View of Forensic Odontologist. Indian Journal of Forensic Medicine and Pathology 4: 147-151.
- Thapar R, Angadi PV, Hallikerimath S, Kale AD (2012) Sex assessment using odontometry and cranial anthropometry: evaluation in an Indian sample. Forensic Sci Med Pathol 8: 94-100.
- Rai B, Kaur J (2013) Sex Determination Methods in Forensic Odontology. In: Rai B, Kaur J (eds.). Evidence-Based Forensic Dentistry. Springer Berlin Heidelberg, Germany. Pg no: 73-79.
- Kondo S, Townsend GC, Yamada H (2005) Sexual dimorphism of cusp dimensions in human maxillary molars. Am J Phys Anthropol 128: 870-877.
- Radlanski RJ, Renz H, Hopfenmüller W (2012) Sexual dimorphism in teeth? Clinical relevance. Clin Oral Investig 16: 395-399.
- Moreno-Gómez F (2013) Sexual Dimorphism in Human Teeth from Dental Morphology and Dimensions: A Dental Anthropology Viewpoint. In: Hiroshi Moriyama (ed.). Sexual Dimorphism. Intech open science open minds, Croatia. Pg no: 97-124.
- Viciano J, López-Lázaro S, Alemán I (2013) Sex estimation based on deciduous and permanent dentition in a contemporary Spanish population. Am J Phys Anthropol 152: 31-43.
- Aguirre L, Castillo D, Solarte D, Moreno F (2006) Frequency and Variability of Five Non-Metric Dental Crown Traits in the Primary and Permanent Dentitions of a Racially Mixed Population from Cali, Colombia. Dental Anthropology 19: 39-48.
- Hanihara T (2008) Morphological variation of major human populations based on nonmetric dental traits. Am J Phys Anthropol 136: 169-182.
- Kimura R, Yamaguchi T, Takeda M, Kondo O, Toma T, et al. (2009) A common variation in EDAR is a genetic determinant of shovel-shaped incisors. Am J Hum Genet 85: 528-535.
- Pereira C, Antunes MT, Pestana D, Mendonça MCd, Santos JC (2012) An Unidentified Skeletal Assemblage from a Post-1755 Mass Grave of Lisbon: Dental Morphology and Population Affinity. Bulletin of the International Association for Paleodontology 6: 12-26.
- Carter K, Worthington S, Smith TM (2014) News and views: non-metric dental traits and hominin phylogeny. J Hum Evol 69: 123-128.
- Turner GC, Nichol CR, Scott GR (1991) Scoring Procedures for Key Morphological Traits of the Permanent Dentition: The Arizona State University Dental Anthropology System. In: Kelley MA, Larsen CS (eds.). Advances in Dental Anthropology. Wiley-Liss, New York, USA. Pg no: 13-31.
- Cunha C (2012) Caracterização da Morfologia Dentária no Médio Guadiana no Neolítico Final-Calcolítico. Fundamentação para o Mapeamento Morfológico das Populações Locais na Pré-História Recente. In: António Valera (ed.). Apontamentos de Arqueologia e Património. Núcleo de Investigação Arqueológica-NIA, Lisbon, Portugal. Pg no: 49-55.
- Scott GR (2008) Dental Morphology. In: Katzenberg MA, Saunders SR (eds.). Biological Anthropology of the Human Skeleton. (2nd edn), New Jersey, John Wiley & Sons. Pg no: 265-298.
- Scott GR, Turner CG (1988) Dental Anthropology. Annu Rev Anthropol 17: 99-126.
- Sweet D (2010) Forensic dental identification. Forensic Sci Int 201: 3-4.
- Pereira C, Bernardo M, Pestana D, Santos JC, Mendonça MC (2010) Contribution of teeth in human forensic identification - discriminant function sexing odontometrical techniques in Portuguese population. J Forensic Leg Med 17: 105-110.
- Marado L, Campanacho V (2013) Carabelli’s trait: Definition and review of a commonly used dental non-metric variable. Cadernos do GEEvH 2: 24-39.
- Rocha L, Rivas H, Moreno F (2007) Frequency and variability of the dental morphology in African-Colombian children of a school institution of Puerto Tejada, Cauca, Colombia. Colombia Médica Julio- 38: 210-221.
- Díaz E, García L, Hernández M, Palacio L, Ruiz D, et al. (2014) Frequency and variability of dental morphology in deciduous and permanent dentition of a Nasa indigenous group in the municipality of Morales, Cauca, Colombia. Colomb Med (Cali) 45: 15-24.
- Zorba E, Spiliopoulou C, Moraitis K (2013) Evaluation of the accuracy of different molar teeth measurements in assessing sex. Forensic Sci Med Pathol 9: 13-23.
- Moreno F, Moreno SM, Díaz CA, Bustos EA, Rodríguez JV (2004) Prevalencia y variabilidad de ocho rasgos morfológicos dentales en jóvenes de tres colegios de Cali. Colombia Médica 35: 16-23.
- Delgado-Burbano (2007) Variacíon dental no-métrica y el tráfico de esclavos por el atlántico: la ascendencia biológica y los orígenes geográficos de una población afro-colombiana. Revista Española de Antropología Física 27: 13-32.
- Aragón N, Bastidas C, Bedón LK, Duque P, Sánchez M, et al. (2008) Rasgos morfológicos dentales coronales en denticíon temporal y permanente: Distancia biológica entre tres grupos indígenas del Amazonas Colombiano. Revista Odontológica Mexicana 12: 13-28.
- Boaz K, Gupta C (2009) Dimorphism in human maxillary and mandibular canines in establishment of gender. J Forensic Dental Sciences 1: 42-44.
- Kapila R, Nagesh KS, Iyengar AR, Mehkri S (2011) Sexual Dimorphism in Human Mandibular Canines: A Radiomorphometric Study in South Indian Population. J Dent Res Dent Clin Dent Prospect 5: 51-54.
- Sonika V, Harshaminder K, Madhushankari GS, Sri Kennath JA (2011) Sexual dimorphism in the permanent maxillary first molar: a study of the Haryana population (India). J Forensic Odontostomatol 29: 37-43.
- Parekh DH, Patel SV, Zalawadia AZ, Patel SM (2012) Odontometric Study of Maxillary Canine Teeth to Establish Sexual Dimorphism in Gujarat Population. Int J Biol Med Res 3: 1935-1937.
- Angadi PV, Hemani S, Prabhu S, Acharya AB (2013) Analyses of odontometric sexual dimorphism and sex assessment accuracy on a large sample. J Forensic Leg Med 20: 673-677.
- Ayoub F, Shamseddine L, Rifai M, Cassia A, Diab R, et al. (2014) Mandibular Canine Dimorphism in Establishing Sex Identity in the Lebanese Population. Int J Dent 2014: 235204.
- Chukwujekwu IE, Ezejindu DN, Moses FC (2014) Odontometric Study of Mandibular Canine Teeth Dimorphism in Establishing Sex Identity in South-east Nigeria. Int J Med Med Sci 1: 38-41.
- Dumancic J, Pavicin IS, Brkic H (2013) Sexual Dimorphism in Canine Morphology. J Forensic Odontostomatology 31: 44.
- Marcovich I, Prado E, Díaz P, Ortiz Y, Martínez C, et al. (2012) Dental Morphology Analysis of Afro-Colombian School Children from Villa Rica, Cauca, Colombia. Rev Fac Odontol Univ Antioq 24: 37-61.
- Wetherell J, Winning T, Townsend G (2004) Localized Asymmetry in Human Dental Crown Form- an Interesting Case. Dental Anthropology 17: 18-23.
Citation: Abrantes C, Santos R, Pestana D, Pereira CP (2015) Application of Dental Morphological Characteristics for Medical-Legal Identification: Sexual Diagnosis in a Portuguese Population. J Forensic Legal Investig Sci 1: 001.
Copyright: © 2015 Catarina Abrantes, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

