
Anterior Angle Closure Glaucoma Induced by Topiramate and Follow Up by Anterior Chamber Angle Optical Coherence Tomography - A Case Report
*Corresponding Author(s):
Sergio Antonio Arrascue LimoDepartment Of Ophthalmology, Vision 20/20 Clinic, Lambayeque, Peru
Tel:+51 946059651,
Email:sarrascue.oftalmo@gmail.com
Abstract
We describe a case of a 26 year old female who developed bilateral angle closure glaucoma associated with oral topiramate therapy and her follow up by Anterior Chamber Angle Optical Coherence Tomography (ACA-OCT) during her treatment. We observed a partial resolution of symptoms and corneal edema after cessation of topiramate and initiation of topical and oral antiglaucomatose drugs, but, an anatomic resolution determined by the aperture of the anterior chamber angle by ACA-OCT was only visualized after topical cycloplegic drugs were started.
Intraocular pressure normalized during the first week after the therapy started, aperture of the anterior chamber angle was observed 1 day after cycloplegic drugs therapy started, visual acuity normalized 20 day after the cessation of topiramate and two weeks after the cessation of topical atropine.
Topiramate, a sulfa-derivative antiepileptic medication may cause idiosyncratic ciliochoroidal detachments and ciliary body edema leading to anterior displacement of the lens-iris diaphragm, lens thickening, and acute angle-closure glaucoma. The clinical history is important for the identification of this pathology in order to start the treatment based in cessation of the oral topiramate, antiglaucomatose drugs and topical atropine.
CASE REPORT
She had been taking oral topiramate, 50 mg qid, for 7 days for treatment of depression. She has a history of refractive myopia in both eyes; there was no family history of glaucoma or ocular hypertension in her closest relatives.
At the initial exam, the best corrected visual acuity was +1.00 LogMAR (20/200 Snellen) in both eyes. The slit lamp evaluation showed a hyperemic conjunctiva, bilateral micro-cystic corneal edema, shallow anterior chambers (dangerously narrow by Van Herick clinical classification), with bombe anterior displacement of the iris. Medium mydriatic pupils with poor photo-reaction activity, clear lenses and good ocular motility. The Intraocular Pressure (IOP) measured with Goldman applanation tonometer was 35 mmHg in both eyes. Given suspicion that the acute angle closure glaucoma was due to the oral topiramate, this drug was stopped. Treatment with oral carbonic anhydrase inhibitors (acetazolamide 250 mg qid) and topic dorzolamide 2%, timolol 0.5%, brimonidine tartrate 0.2% was started.
Repeat exam 24 hours after treatment showed improved corneal clarity and edema. IOP was measured to be 24 mmHg in both eyes. However the shallow anterior chamber with bombe iris remained (Figure 1). Topical Atropine Sulphate 1% qid was added to the treatment.
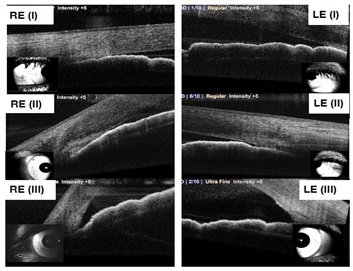 Figure 1: Shows the temporal ACA-OCT in right eye (RE) and left eye (LE) at the beginning of the Bilateral TPM induced AACG (I), 24 hours after antiglaucomatose therapy and cessation of topiramate (II) and 24 hours after beginning of topical atropine therapy (III).
Figure 1: Shows the temporal ACA-OCT in right eye (RE) and left eye (LE) at the beginning of the Bilateral TPM induced AACG (I), 24 hours after antiglaucomatose therapy and cessation of topiramate (II) and 24 hours after beginning of topical atropine therapy (III).At 48 hours after initial treatment, IOP was 17 mmHg in both eyes and open Anterior Chamber Angles (ACA) were seen on slit lamp evaluation. She continued to receive topical treatment for a week and presented for follow up every 48 hours, ACA Optical Coherence Tomography (OCT) follow up was maintained withaNidekRS-3000 Advance Spectral Domain OCT equipment (Figure 1).
A week after the treatment was started, there was clinical improvement, IOP was 15 mmHg, and there was open ACA confirmed by OCT and clinical evaluation. We decided to stop topical atropine and oral acetazolamide.
On the last evaluation, 20 days after symptoms started, the IOP was measured to be 10 mmHg IOP; best corrected visual acuity was 0 LogMAR (20/20 Snellen) with a -5.25 D myopia refraction, which was her initial refraction according to medical records.
There was no glaucomatous damage observed in the SITA 24-2 visual fields test (HAAG-STREIT Octopus 600 perimeter), neither at the macular map OCT of the ganglion cell layer or at the optic disk map of the nerve fiber layer in both eyes after the resolution of the ocular hypertensive episode.
DISCUSSION
Acute Angle Closure Glaucoma (AACG) is a devastating condition that presents with eye pain, blurred vision, headache, nausea and vomiting, and is one of the most dangerous adverse effects of topiramate for the eyes. Optic nerve damage with visual field defects and vision impairment may happen if elevated Intraocular Pressure (IOP) is persistent [9,10]. Fortunately, in our case, there was no permanent damage to the optic structures due to hypertensive ocular episode noted on follow up.
The mechanisms of drug-induced angle closure are pupillary block. As the dilated pupil constricts, angle crowding results due to pupillary dilation and idiosyncratic drug reactions that change the iridocorneal angle by ciliochoroidal effusion [9].
Bilateral Acute Angle Closure Glaucoma (AACG) with myopic shift is a rare entity that is usually seen after anterior rotation of ciliary processes and anterior shifting of the iris-lens diaphragm. This can be noted after uveal effusion syndrome as an adverse effect of Topiramate therapy but also can be noted after severe panretinal photocoagulation, Vogt-Koyanagi-Harada syndrome, aqueous misdirection syndrome and induced by other drugs (oral sulfa-based drugs, acetazolamide, cotrimoxazole, topical pilocarpine) [11-13].
We describe a case of sudden onset of blurred vision, headache, photophobia and ocular pain due to angle-closure glaucoma as a rare adverse effect following topiramate use. We suspected her symptoms were due to topiramate given the symptomatology onset and short time from exposure to topiramate.
Serial ACA-OCT was captured demonstrating the gradual resolution of the angle narrowing that was reached only after the topical cycloplegic therapy was started (Figure 2). There are several lines of evidence indicating that cycloplegics are effective in improving cilichoroidal effusion and reducing IOP since they cause retraction of the ciliary processes and induce an anatomic deeper anterior chamber [14].
The clinical and medical history is important for the identification of this pathology in order to start the proper treatment, including cessation of the oral topiramate, starting antiglaucomatose drugs and topical atropine. Kawali et al., recommend in their case report, not to only focus on the narrow angle, but also on the hypertensive and inflammatory state of the eye because starting a topical cholinergic agonist drug like pilocarpine, in order to generate a miotic effect, could result in more inflammation and aggravation of the pathology [15].
A difficulty in our case was the identification of the ACA exact meridian for the proper follow up with the ACA OCT, even we carefully measured the nasal and temporal ACA meridians with a corneal marker, it may not be exactly the same measured ACA angles, however, the evolution of the anterior chamber angles apertures was really notorious as the resolution of the hypertensive episode. We could recommend for more accurate measurements, the use of an anterior chamber tracking device, this was not available in our OCT software.
CONCLUSION
REFERENCES
- Banta JT, Hoffman K, Budenz DL, Ceballos E, Greenfield DS (2001) Presumed topiramate-induced bilateral acute angle-closure glaucoma. Am J Ophthalmol 132: 112-114.
- Senthil S, Garudadri C, Rao HBL, Maheshwari R (2010) Bilateral simultaneous acute angle closure caused by sulphonamide derivatives: A case series. Indian J Ophthalmol 58: 248-252.
- Goldberg JL, Lau AG, Fan B, Ford L, Greenberg HE (2016) Is uveitis associated with topiramate use? A cumulative review. Clin Ophthalmol 10: 1467-1470.
- Hesami O, Hosseini SS, Kazemi N, Hosseini-Zijoud SM, Moghaddam NB, et al. (2016) Evaluation of Ocular Side Effects in the Patients on Topiramate Therapy for Control of Migrainous Headache. J Clin Diagn Res 10: 1-4.
- Rosenberg K, Maguire J, Benevento J (2017) Topiramate-induced macular neurosensory retinal detachment. Am J Ophthalmol Case Rep 7: 31-37.
- Mahendradas P, Parab S, Sasikumar R, Kawali A, Shetty BK (2018) Topiramate-induced acute angle closure with severe panuveitis: A challenging case report. Indian J Ophthalmol 66: 1342-1344.
- Fraunfelder FW, Fraunfelder FT, Keates EU (2004) Topiramate-associated acute, bilateral, secondary angle-closure glaucoma. Ophthalmology 111: 109-111.
- Etminan M, Maberley D, Mikelberg FS (2012) Use of topiramate and risk of glaucoma: a case-control study. Am J Ophthalmol 153: 827-830.
- Cruciani F, Lorenzatti M, Nazzarro V, Abdolrahimzadeh S (2009) Bilateral acute angle closure glaucoma and myopia induced by topiramate. Clin Ter 160: 215-216.
- Liu Y, Rhee DJ (2013) Acute bilateral angle closure. JAMA Ophthalmol 131: 1231-1232.
- Blumenthal DT (2004) Acute myopia and angle-closure glaucoma induced by topiramate. Neurology 63: 762.
- Craig JE, Ong TJ, Louis DL, Wells JM (2004) Mechanism of topiramate-induced acute-onset myopia and angle closure glaucoma. Am J Ophthalmol 137: 193-195.
- Mazumdar S, Tripathy K, Sarma B, Agarwal N (2019) Acquired myopia followed by acquired hyperopia due to serous neurosensory retinal detachment following topiramate intake. Eur J Ophthalmol 29: 21-24.
- Abtahi MA, Abtahi SH, Fazel F, Roomizadeh P, Etemadifar M, et al. (2012) Topiramate and the vision: a systematic review. Clin Ophthalmol 6: 117-131.
- Kawali A, Ayyar A, Ahuja A, Abdul KM (2016) Topiramate-induced hypertensive uveitis and evolving Fuchs' uveitis: A case report. J Clin Ophthalmol Res 4: 46-48.
Citation: Limo SAA, Limo CEA (2019) Anterior Angle Closure Glaucoma Induced by Topiramate and Follow Up by Anterior Chamber Angle Optical Coherence Tomography - A Case Report. J Ophthalmic Clin Res 6: 058.
Copyright: © 2019 Sergio Antonio Arrascue Limo, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

